A piston-cylinder device with a spring connected to the piston contains molecular nitrogen (N2) at an...
Fantastic news! We've Found the answer you've been seeking!
Question:


Transcribed Image Text:
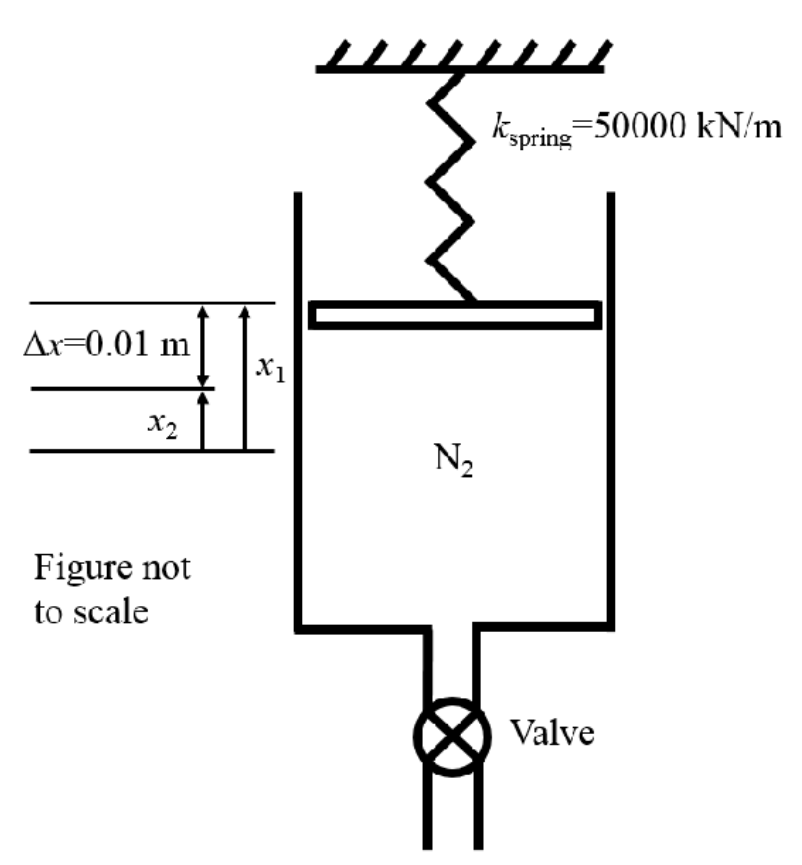
A piston-cylinder device with a spring connected to the piston contains molecular nitrogen (N2) at an initial temperature of 350 K and has an initial internal volume of 2.00 m. At the initial state, the linear spring with spring constant 50,000 kN/m applies a force to the piston such that the initial pressure of the N2 is 3000 kPa. The piston has a cross-sectional area of 1.5 m and the piston has negligible mass. At t=0 a discharge process is initiated by opening a valve such that N flows from the piston cylinder device until the piston is lowered by 0.01 m, at which point the discharge process is stopped by closing the valve. During the discharge process heat is transferred to the nitrogen in the piston-cylinder device such the process occurs in an isothermal manner. Assume that the N2 behaves as an ideal gas, if necessary the constant specific heat assumption may be employed. Ax=0.01 m m X2 X1 N Figure not to scale Kspring 50000 kN/m Valve a) b) c) d) e) f) State any assumptions necessary to solve the problem and draw system boundary for first law analysis indicating all heat transfer and work interactions. (2 Marks) Hint: for parts b), c) and d) utilise F = kx and P=F/A Determine the amount the spring is compressed at the initial state to achieve the initial pressure (in m). (1 Mark) Determine the pressure of the N at the end of the discharge process (in kPa). (1 mark) k Show that the boundary work done by linear spring can be given by W = 1/(x-x), b.spring 2 where x and x2 are the distances at the initial and final state from the spring zero datum respectively. Hence determine the work done by the spring during the discharge process (in kJ). (3 Marks) Determine the initial and final mass of N in the piston cylinder device (in kg). (2 Marks) Determine the heat transferred to the N during the discharge process (in kJ). (6 marks) A piston-cylinder device with a spring connected to the piston contains molecular nitrogen (N2) at an initial temperature of 350 K and has an initial internal volume of 2.00 m. At the initial state, the linear spring with spring constant 50,000 kN/m applies a force to the piston such that the initial pressure of the N2 is 3000 kPa. The piston has a cross-sectional area of 1.5 m and the piston has negligible mass. At t=0 a discharge process is initiated by opening a valve such that N flows from the piston cylinder device until the piston is lowered by 0.01 m, at which point the discharge process is stopped by closing the valve. During the discharge process heat is transferred to the nitrogen in the piston-cylinder device such the process occurs in an isothermal manner. Assume that the N2 behaves as an ideal gas, if necessary the constant specific heat assumption may be employed. Ax=0.01 m m X2 X1 N Figure not to scale Kspring 50000 kN/m Valve a) b) c) d) e) f) State any assumptions necessary to solve the problem and draw system boundary for first law analysis indicating all heat transfer and work interactions. (2 Marks) Hint: for parts b), c) and d) utilise F = kx and P=F/A Determine the amount the spring is compressed at the initial state to achieve the initial pressure (in m). (1 Mark) Determine the pressure of the N at the end of the discharge process (in kPa). (1 mark) k Show that the boundary work done by linear spring can be given by W = 1/(x-x), b.spring 2 where x and x2 are the distances at the initial and final state from the spring zero datum respectively. Hence determine the work done by the spring during the discharge process (in kJ). (3 Marks) Determine the initial and final mass of N in the piston cylinder device (in kg). (2 Marks) Determine the heat transferred to the N during the discharge process (in kJ). (6 marks)
Expert Answer:

Related Book For 

Thermodynamics An Interactive Approach
ISBN: 978-0130351173
1st edition
Authors: Subrata Bhattacharjee
Posted Date:
Students also viewed these mechanical engineering questions
-
a. Example 13-1: Batch Reactor with an Exothermic Reaction Wolfram 1. Adiabatic Case: Use Wolfram to see whether you can find a trajectory that is ready to ignite and whose trajectory looks like a...
-
R. Bright, an electrical goods wholesaler, has three departments: (a) Music, (b) TV and (c) Kitchen. The following is a summary of Brights sales invoices during the week 1 to 7 February 2013. (a)...
-
Frankum Company has issued three different bonds during 2019. Interest is payable annually on each of these bonds. 1. On January 1, 2019, 1,000, 8%, 5-year, $1,000 bonds dated January 1, 2019, were...
-
For the network of Fig. 5.156: a. Determine re. b. Find Zi and Z0. c. Calculate Av. d. Repeat parts (b) and (c) with r0 = 20 Ω. 020 V 2.2 k 390 k 9-140 20 100 k Vi Z,
-
A neighborhood fuel cell power plant is to be designed for an electric power output of \(2000 \mathrm{~kW}\) with liquid-water product. Estimate the flow rates of hydrogen and oxygen during peak...
-
The Glory Mountain State Ski Are expects to attract 292,500 skier days during the coming ski season. A skier day represents one skier at the mountain for one day. In addition to a $ 2,000,000 per...
-
Three friends decide to start a web development business together. They have been working separately as independent consultants for several years, but they decide they would benefit greatly from...
-
Define size and height (see Exercise 8.10) for rooted ternery trees (RTT) and rooted trees (RT). (a) Prove: For any rooted ternary tree (RTT) 7, size(7) (3height(7)+1-1)/3. Find such a bound for the...
-
The risk-free interest rate is 3% annualized and continuously compounded. Assume the market is frictionless. When you plot the forward curves below, you may choose to either (i) use the analytical...
-
For the given open-loop transfer function, design a PD controller using root-locus (angle and magnitude conditions) to shape the closed-loop step response with a settling time of 1 second and a 10%...
-
For the given four-bar linkage mechanism, Perform position, speed, and acceleration analysis. Draw the speed polygon and acceleration polygon. 0 226 355 15 466 40 20- 10 0 Linkage 02 = 100 0 10 3 4...
-
When the buyer for the activewear department started to develop the merchandise plan for the spring season, she reviewed the six-month plan in Figure 35. The numbers that were achieved represent the...
-
1. Let X[0.. n-1] be an array of characters. A subsequence of X is a subset of not-necessarily- consecutive characters (X[i], X[i].....X[ik]) such that i
-
Solve foa B. 30 =C D= 81 A 22 6
-
Distinguish between the work performed by public accountants and the work performed by accountants in commerce and industry and in not-for-profit organisations.
-
One kmol of H2O is heated to 3000 K at a pressure of 1 atm. Determine the equilibrium composition of H2O (in kmols) assuming that only H2O , OH, O2 and O are present.
-
A power plant operates on a regenerative vapor power cycle with one closed feed water heater. Steam enters the first turbine stage at 12 MPa, 520oC and expands to 1200 kPa, where some of the steam is...
-
A gas turbine power plant operates on a simple Brayton cycle with air as the working fluid. Air enters the turbine at 800 kPa and 1200 K, and it leaves the turbine at 100 kPa and 750 K. Heat is...
-
A system, consisting of \(2 \mathrm{~mol}\) of \(\mathrm{N}_{2}, 5 \mathrm{~mol}\) of \(\mathrm{H}_{2}\) and \(2 \mathrm{~mol}\) of \(\mathrm{NH}_{3}\) initially, is undergoing the following...
-
The following reaction takes place in a system consisting of \(3 \mathrm{~mol} \mathrm{CH}_{4}, 5 \mathrm{~mol} \mathrm{H}_{2} \mathrm{O}\), \(1 \mathrm{~mol} \mathrm{CO}\) and \(4 \mathrm{~mol}...
-
The study of thermodynamics enables us to understand (a) Whether the transformation of energy is feasible or not (b) To what extent the transformation will take place (c) In which direction the...

Study smarter with the SolutionInn App


