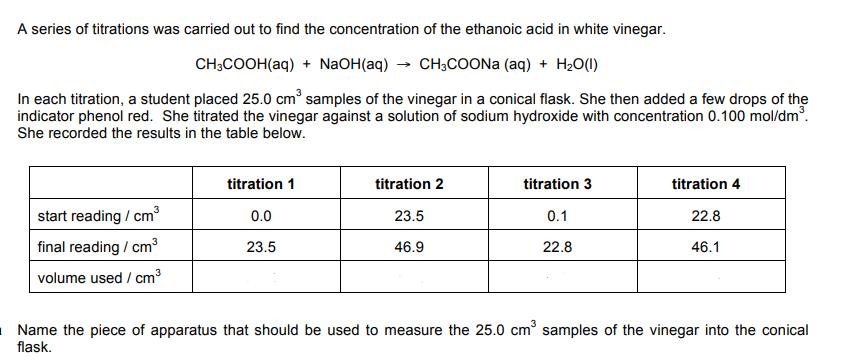
A series of titrations was carried out to find the concentration of the ethanoic acid in...
Fantastic news! We've Found the answer you've been seeking!
Question:

![]()
![]()
![]()
Transcribed Image Text:
A series of titrations was carried out to find the concentration of the ethanoic acid in white vinegar. CH3COOH(aq) + NaOH(aq) → CH3COONa (aq) + H₂O(l) In each titration, a student placed 25.0 cm³ samples of the vinegar in a conical flask. She then added a few drops of the indicator phenol red. She titrated the vinegar against a solution of sodium hydroxide with concentration 0.100 mol/dm³. She recorded the results in the table below. start reading / cm³ final reading / cm³ volume used / cm³ titration 1 0.0 23.5 titration 2 23.5 46.9 titration 3 0.1 22.8 titration 4 22.8 46.1 Name the piece of apparatus that should be used to measure the 25.0 cm³ samples of the vinegar into the conical flask. Name the piece of apparatus that is used for the sodium hydroxide solution. Complete the table to show the volume used in each titration. Calculate the mean volume of sodium hydroxide, leaving out any anomalous results. Find the concentration of the ethanoic acid in mol/dm³. Give your answer to 3 significant figures. Find the concentration of the ethanoic acid in g/dm³. Give your answer to 3 significant figures. Explain why this titration may have been difficult to do with brown vinegar. A series of titrations was carried out to find the concentration of the ethanoic acid in white vinegar. CH3COOH(aq) + NaOH(aq) → CH3COONa (aq) + H₂O(l) In each titration, a student placed 25.0 cm³ samples of the vinegar in a conical flask. She then added a few drops of the indicator phenol red. She titrated the vinegar against a solution of sodium hydroxide with concentration 0.100 mol/dm³. She recorded the results in the table below. start reading / cm³ final reading / cm³ volume used / cm³ titration 1 0.0 23.5 titration 2 23.5 46.9 titration 3 0.1 22.8 titration 4 22.8 46.1 Name the piece of apparatus that should be used to measure the 25.0 cm³ samples of the vinegar into the conical flask. Name the piece of apparatus that is used for the sodium hydroxide solution. Complete the table to show the volume used in each titration. Calculate the mean volume of sodium hydroxide, leaving out any anomalous results. Find the concentration of the ethanoic acid in mol/dm³. Give your answer to 3 significant figures. Find the concentration of the ethanoic acid in g/dm³. Give your answer to 3 significant figures. Explain why this titration may have been difficult to do with brown vinegar.
Expert Answer:
Related Book For 

Complete Business Statistics
ISBN: 9780077239695
7th Edition
Authors: Amir Aczel, Jayavel Sounderpandian
Posted Date:
Students also viewed these chemistry questions
-
A study was carried out to find out whether prices differed, on average, within three possible market conditions: monopoly, limited competition, and strong competition. The results were reported as...
-
A randomized field experiment was carried out to determine how workers respond to wage cuts and the results published in Institute for the Study of Labor: Discussion Paper Series (March 2011). A...
-
A regression analysis was carried out to determine whether wages increase for blue-collar workers depending on the extent to which firms that employ them engage in product exportation. The sample...
-
In Problems 6374, perform the indicated operations and simplify the result. Leave your answer in factored form. 3x x - 1 x - 4 18 x - 2x + 1 2
-
You have a preferred stock which has a call ability feature after 10 years at $115. The dividend annually is $10. Your sister buys the preferred stock from you when yields are 12% after 4 years....
-
The binomial coefficient equals
-
In flange coupling, the weakest element is: (a) Flange (b) Bolt (c) Key (d) Shaft
-
The SAT is the most widely used test in the undergraduate admissions process. Scores on the math portion of the SAT are believed to be normally distributed and range from 200 to 800. A researcher...
-
Calculate the theoretical yield of SnS_2. theoretical yield: 0.768 g The student recovers 0.384 g of SnS_2. Calculate the percent yield of SnS_2 that the student obtained. percent yield: %
-
Pacifico Company, a U.S.-based Importer of beer and wine, purchased 1,500 cases of Oktoberfest-style beer from a German supplier for 375,000 euros. Relevant U.S. dollar exchange rates for the euro...
-
Hutto Corp. has set the following standard direct materials and direct labor costs per unit for the product it manufactures. Direct materials (16 lbs. @ $4 per lb.). $64 48 Direct labor (3 hrs. @ $16...
-
Assume Ellina earns a 4 percent after-tax rate of return and that she owes a friend $1,500. Would she prefer to pay the friend $1,500 today or $1,725 in four years?
-
Why the companies need to consider their social cost and social benefits, explain.
-
Design a business model template as basis for a professional business plan Explain the connection between a business plan and business model. Explain the importance of a business model in a business...
-
Identify the graph of the function. 5) f(x)-(x+3)3-2 A) -6 -6 A) -2,-1, 1,4 B) B) -1, 1,2,4 D) -6 Use a graphing calculator to approximate all real solutions of the equation f(x)=0. 6)...
-
Consider the following logic function: X=A (B+C)+C B 1. (10 pts) First implement this logic using a CMOS circuit by crafting the pulldown network to match the logic expression and utilize a follow-on...
-
One 2 June 2015, the company received their bank statement showing the following transactions: 1/05/15 Opening Balance 7/05/15 Deposit 8/05/15 EFT Electricity 8/05/15 Dishonour Cheque - WK Ltd...
-
Prairie Outfitters, Inc., a retailer, accepts paymnent through credit cards. During August, credit card sales amounted to $12,000. The processor charges a 3% fee. Assuming that the credit card...
-
In a regression analysis, 12 out of 30 residuals are greater than 1.00 in value, and the rest are not. With A denoting a residual greater than 1 and B a residual less than 1, the residuals are as...
-
For problem 8-52, give a 99% confidence interval for the difference between the proportions for viewers of the two shows who watched 1 to 6 commercials. Interpret the results. In problem According to...
-
Recently, Venezuela instituted a new accounting method for its oil revenues. Suppose that a random sample of 100 accounting transactions using the old method reveals 6 errors. Is there evidence of...
-
In 2022, Mark purchased two separate activities. Information regarding these activities for 2022 and 2023 is as follows: The 2022 losses were suspended losses for that year. During 2023, Mark also...
-
Jerry sprayed all of the landscaping around his business with a pesticide in June 2023. Shortly thereafter, all of the trees and shrubs unaccountably died. The FMV and the adjusted basis of the...
-
In 2023, Julie, a single individual, reported the following items of income and deduction: Julie owns 100% and is an active participant in the rental real estate activity. What is her taxable income...

Study smarter with the SolutionInn App



