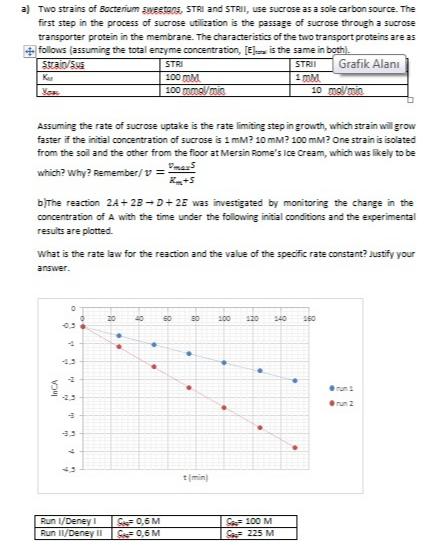
a) Two strains of Bocterium swestons, STRI and STRII, use sucrose as a sole carbon source....
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
a) Two strains of Bocterium swestons, STRI and STRII, use sucrose as a sole carbon source. The first step in the process of sucrose utilization is the passage of sucrose through a sucrose transporter protein in the membrane. The characteristics of the two transport proteins are as + follows (assuming the total enzyme concentration, [E is the same in both). Strain/Sus STRI STRII Grafik Alanı 100 mM. 100 mma/mia Ku 1 mM. 10 mol/min Assuming the rate of sucrose uptake is the rate limiting step in growth, which strain will grow faster if the initial concentration of sucrose is 1 mM? 10 mM? 100 mM? One strain is isolated from the soil and the other from the floor at Mersin Rome's ice Cream, which was likely to be which? why? Remember/v = bithe reaction 24+ 23 - D+ 2E was investigated by monitoring the change in the concentration of A with the time under the following initial conditions and the experimental results are plotted. What is the rate law for the reaction and the value of the specific rate constant? Justify your answer. 20 120 s40 150 40 200 run 23 run 2 Run /Deney I S0,6 M C 0,6 M C 100 M Run II/Deney II S 225 M a) Two strains of Bocterium swestons, STRI and STRII, use sucrose as a sole carbon source. The first step in the process of sucrose utilization is the passage of sucrose through a sucrose transporter protein in the membrane. The characteristics of the two transport proteins are as + follows (assuming the total enzyme concentration, [E is the same in both). Strain/Sus STRI STRII Grafik Alanı 100 mM. 100 mma/mia Ku 1 mM. 10 mol/min Assuming the rate of sucrose uptake is the rate limiting step in growth, which strain will grow faster if the initial concentration of sucrose is 1 mM? 10 mM? 100 mM? One strain is isolated from the soil and the other from the floor at Mersin Rome's ice Cream, which was likely to be which? why? Remember/v = bithe reaction 24+ 23 - D+ 2E was investigated by monitoring the change in the concentration of A with the time under the following initial conditions and the experimental results are plotted. What is the rate law for the reaction and the value of the specific rate constant? Justify your answer. 20 120 s40 150 40 200 run 23 run 2 Run /Deney I S0,6 M C 0,6 M C 100 M Run II/Deney II S 225 M
Expert Answer:
Answer rating: 100% (QA)
a From given Km and Vmax data and micheal menton kinetic equation we know Km is ... View the full answer

Related Book For 

Intermediate Accounting Reporting and Analysis
ISBN: 978-1285453828
2nd edition
Authors: James M. Wahlen, Jefferson P. Jones, Donald Pagach
Posted Date:
Students also viewed these electrical engineering questions
-
The following reaction is the first step in the industrial synthesis of acetone and phenol (C6H5OH). AIBN (2,29-azobisisobutyronitrile) initiates radical reactions by breaking down upon heating to...
-
Why does the first step in the strategic market planning process involve a situation analysis?
-
Raphael is a professor who teaches physics at a university where he is paid a yearly salary of $200,000. He plans to take the next year off to write a book, so he won't earn any money next year. He...
-
As manager of a local pizza parlor, you want to develop a balanced scorecard so you can more effectively monitor the restaurants performance. Required a. Propose at least two goals for each...
-
Obtain the exponential Fourier series expansion of the half-wave rectified sinusoidal current of Fig. 17.82. ATA i(t) sin t
-
Diane's Dairy Sales & Delivery finished its first year of operations on December 31, 2014. After adjusting journal entries, the company presented the following adjusted trial balance. Using this...
-
How can quota sampling being applied through a website like www.linkedin.com?
-
Pink Martini Company has gathered the following information. Units in beginning work in process ............20,000 Units started into production .............72,000 Units in ending work in process...
-
A new medical practice purchases computer equipment that cost $ 1 5 , 0 0 0 , to be used for medical billing. In addition, the practice purchases billing software that cost $ 5 , 0 0 0 . Both the...
-
[The following information applies to the questions displayed below.) Assume Down, Incorporated, was organized on May 1 to compete with Despair, Incorporated-a company that sells de- motivational...
-
(x) The figure above shows one of the possible energy eigenfunctions y(x) for a particle bouncing freely back and forth along the x-axis between impenetrable walls located at x = -a and x = +a. The...
-
A eukaryotic protein-encoding gene contains two introns and three exons: exon 1intron 1exon 2intron 2exon 3. The 5 splice site at the boundary between exon 2 and intron 2 has been eliminated by a...
-
Lets suppose a mutation removes the ICR next to the Igf2 gene. If this mutation is inherited from the mother, will the Igf2 gene (from the mother) be silenced or expressed? Explain.
-
What is the difference between an autonomous element and a nonautonomous element? Is it possible for nonautonomous elements to move? If yes, explain how.
-
Researchers have determined that environmental agents that do not cause gene mutations can contribute to cancer. Explain how. Would these epigenetic changes be passed to offspring?
-
In rabbits, the color of body fat is controlled by a single gene with two alleles, designated Y and y. The outcome of this trait is affected by the diet of the rabbit. When raised on a standard...
-
In 20X2, Jacque's Gardening Supply acquired Glenn's Agricultural Center and recorded goodwill in the amount of $450,000. At the end of the 20X4 year, the net assets (including goodwill) of Glenn's...
-
A police officer pulls you over and asks to search your vehicle because he suspects you have illegal drugs inside your car. Since he doesn't have reasonable suspicion to search your car, legally he...
-
In 2017, Tarlo Company agrees to construct a highway for Brice County over a 3 year period ( 2017 through 2019). The contract price is $ 1,200,000, and the construction costs ( both actual and...
-
Assume prices were stable during the period. The following values were obtained from the inventory records of Harris Company which has a fiscal year ending on December 31: Inventory, January 1, 2016,...
-
Each unit of Black Corporations inventory has a net realizable value of $300, a normal profit margin of $35, and a current replacement cost of $250. Determine the amount per unit that should be used...
-
Factor (smart beta) strategy ETFs are least likely to be used by investors: A. to modify portfolio risk. B. for tactical trading purposes. C. to seek outperformance versus a benchmark.
-
Which of the following statements regarding applications of ETFs in portfolio management is correct? A. Equity ETFs tend to be more active than fixed-income ETFs. B. The range of risk exposures...
-
Which of Stosurs statements regarding ETF mechanics is correct? A. Statement 1 B. Statement 2 C. Statement 3 Howie Rutledge is a senior portfolio strategist for an endowment fund. Rutledge meets with...

Study smarter with the SolutionInn App


