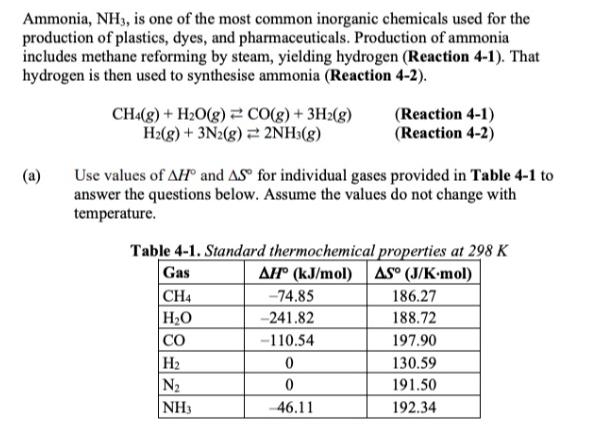
Ammonia, NH3, is one of the most common inorganic chemicals used for the production of plastics,...
Fantastic news! We've Found the answer you've been seeking!
Question:

Transcribed Image Text:
Ammonia, NH3, is one of the most common inorganic chemicals used for the production of plastics, dyes, and pharmaceuticals. Production of ammonia includes methane reforming by steam, yielding hydrogen (Reaction 4-1). That hydrogen is then used to synthesise ammonia (Reaction 4-2). (a) CH4(g) + HO(g) = CO(g) + 3H2(g) H2(g) + 3N2(g) 2NH3(g) Use values of AH and AS for individual gases provided in Table 4-1 to answer the questions below. Assume the values do not change with temperature. (Reaction 4-1) (Reaction 4-2) Table 4-1. Standard thermochemical properties at 298 K Gas AH (kJ/mol) AS (J/K-mol) CH4 HO CO H N NH3 -74.85 -241.82 -110.54 0 0 -46.11 186.27 188.72 197.90 130.59 191.50 192.34 (b) (i) (ii) (iii) (iv) (i) (ii) Calculate the values of A,H for Reactions 4-1 and 4-2. Present these values per mole of H(g). Calculate the standard enthalpy change for the net reaction (i.e., the production of ammonia and carbon monoxide from methane, water vapour, and nitrogen) and state whether the net process is endothermic or exothermic. Determine the temperature at which neither reactants nor products would predominate for EACH reaction. State the range of temperatures where the products would predominate in EACH reaction. Sketch a plot of Gibbs free energy per mol of H versus temperature for Reactions 4-1 and 4-2. Extend the x-axis to 0 K. Label both axes, and show the axes' origins. Label the y-intercept for both lines. Ensure slopes of both lines have the correct sign and magnitude. Explain using thermodynamic concepts why the two steps of ammonia production should be run in separate reactors if the reactions are run at a standard state. Ammonia, NH3, is one of the most common inorganic chemicals used for the production of plastics, dyes, and pharmaceuticals. Production of ammonia includes methane reforming by steam, yielding hydrogen (Reaction 4-1). That hydrogen is then used to synthesise ammonia (Reaction 4-2). (a) CH4(g) + HO(g) = CO(g) + 3H2(g) H2(g) + 3N2(g) 2NH3(g) Use values of AH and AS for individual gases provided in Table 4-1 to answer the questions below. Assume the values do not change with temperature. (Reaction 4-1) (Reaction 4-2) Table 4-1. Standard thermochemical properties at 298 K Gas AH (kJ/mol) AS (J/K-mol) CH4 HO CO H N NH3 -74.85 -241.82 -110.54 0 0 -46.11 186.27 188.72 197.90 130.59 191.50 192.34 (b) (i) (ii) (iii) (iv) (i) (ii) Calculate the values of A,H for Reactions 4-1 and 4-2. Present these values per mole of H(g). Calculate the standard enthalpy change for the net reaction (i.e., the production of ammonia and carbon monoxide from methane, water vapour, and nitrogen) and state whether the net process is endothermic or exothermic. Determine the temperature at which neither reactants nor products would predominate for EACH reaction. State the range of temperatures where the products would predominate in EACH reaction. Sketch a plot of Gibbs free energy per mol of H versus temperature for Reactions 4-1 and 4-2. Extend the x-axis to 0 K. Label both axes, and show the axes' origins. Label the y-intercept for both lines. Ensure slopes of both lines have the correct sign and magnitude. Explain using thermodynamic concepts why the two steps of ammonia production should be run in separate reactors if the reactions are run at a standard state.
Expert Answer:
Answer rating: 100% (QA)
Ammonia Production Calculations a Enthalpy Change Calculations i Enthalpy change per mole of H2g Rea... View the full answer

Related Book For 

International Marketing And Export Management
ISBN: 9781292016924
8th Edition
Authors: Gerald Albaum , Alexander Josiassen , Edwin Duerr
Posted Date:
Students also viewed these chemical engineering questions
-
Let A, B be sets. Define: (a) the Cartesian product (A B) (b) the set of relations R between A and B (c) the identity relation A on the set A [3 marks] Suppose S, T are relations between A and B, and...
-
Planning is one of the most important management functions in any business. A front office managers first step in planning should involve determine the departments goals. Planning also includes...
-
A spring is at rest in the vertical direction. When a 5 kg mass is placed upon the spring, the length of the spring compresses to 0 . 2 0 meters. The 5 kg mass is removed and replaced by an 8 kg...
-
The balance sheet for Larry's Discount Tire Company shows a book value of stockholders' equity (book value per share * total shares outstanding) of $23,500,000. Furthermore, the firm's income...
-
Three coins are dropped on a table. a. List all possible outcomes in the sample space. b. Find the probability associated with each outcome. c. Let A be the event exactly 2 heads. Find P(A). d. Let B...
-
Briefly explain the manner in which (a) input and output controls and (b) processing controls may be tested when an EDP system is used for cash receipts transactions.
-
The Butterfield Company makes a variety of knives. Each knife is processed on four machines. The processing times required are as follows. Machine capacities (in hours) are 1,500 for machine 1; 1,400...
-
Consider the table below, apply the steps of the banker's algorithm to detect the deadlock. (as we learnt in class) Request matrix, Q Allocation matrix A Available vector V R1 R2 R3 R4 R1 R2 R3 R4 R1...
-
Tires for Less is a franchise of tire stores throughout the greater Northwest. It has projected the following unit sales per tire and costs of tires for the coming year: The company policy is to have...
-
Find an equation of the tangent to the curve at the given point, for the following: x sin y = y cos x at (5, -3) ---
-
Gamma Petroleum owns 100% of the working interest in the Bearcat Field in Wyoming. The field has been producing for 3 years and is expected to produce for 10 more years. Since the state has very...
-
Longhorn Company (70% WI) and Aggie Company (30% WI) own a joint working interest in the Dowling Field. There is a 1/8 royalty owner. The 1/8 royalty is shared proportionally by Longhorn and Aggie....
-
Two leases in far West Texas are being combined to form the West End Unit. REQUIRED: a. Determine the participation factors for each party, assuming the participation factors are based on the acreage...
-
Heagy Oil Company has production on a lease in Louisiana with the following ownership interest: During April, 5,000 (gross) barrels of oil (after correction for temperature, gravity, and BS&W) were...
-
Differentiate between tax evasion and tax avoidance.
-
A gas mixture initially containing 80% CO, 0.4% COS, and 19.6% inert constituents (by mole, mole fraction) is passed over sponge Cu at 1100 K to remove sulfur by the following reaction. COSg) + 2...
-
On January 1, 2017, McIlroy, Inc., acquired a 60 percent interest in the common stock of Stinson, Inc., for $340,200. Stinson's book value on that date consisted of common stock of $100,000 and...
-
Identify the potential barriers (or obstacles) that face companies considering or expanding international marketing operations. Which are the most important and which are less important? Explain.
-
Various classification schemes are sometimes used to given an indication of the potential of a foreign market/country. Explain why these schemes should or should not be used as a basis for deciding...
-
The Woberg Company, located near Aarhus, Denmark, manufactured dishwashers and garbage pulverizers for home use, and cooking equipment and commercial pulverizers for restaurants. A relatively young...
-
True or False. Coulomb damping can be called constant damping.
-
True or False. For an undamped system, the velocity leads the acceleration by \(\pi / 2\).
-
True or False. The motion diminishes to zero in both underdamped and overdamped cases.

Study smarter with the SolutionInn App


