An analyst prepares a stock Cr(III)nitrate standard (SSS) by dissolving 1.0000g of dry chromium nitrate into...
Fantastic news! We've Found the answer you've been seeking!
Question:
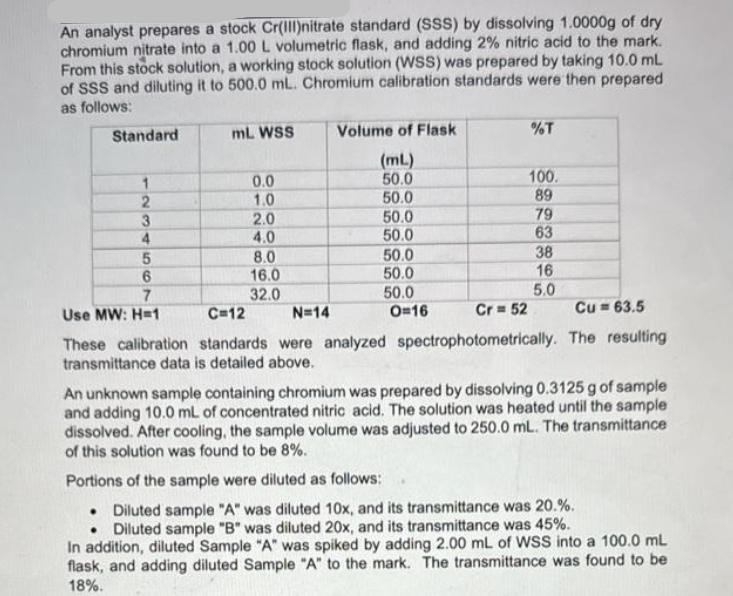

Transcribed Image Text:
An analyst prepares a stock Cr(III)nitrate standard (SSS) by dissolving 1.0000g of dry chromium nitrate into a 1.00 L volumetric flask, and adding 2% nitric acid to the mark. From this stock solution, a working stock solution (WSS) was prepared by taking 10.0 mL of SSS and diluting it to 500.0 mL. Chromium calibration standards were then prepared as follows: Standard mL. WSS Volume of Flask %T (mL) 1 0.0 50.0 100. 23450 1.0 50.0 89 2.0 50.0 79 4.0 50.0 63 8.0 50.0 38 6 16.0 50.0 16 7 32.0 50.0 5.0 Use MW: H=1 C=12 N=14 O=16 Cr=52 Cu 63.5 These calibration standards were analyzed spectrophotometrically. The resulting transmittance data is detailed above. An unknown sample containing chromium was prepared by dissolving 0.3125 g of sample and adding 10.0 mL of concentrated nitric acid. The solution was heated until the sample dissolved. After cooling, the sample volume was adjusted to 250.0 mL. The transmittance of this solution was found to be 8%. Portions of the sample were diluted as follows: Diluted sample "A" was diluted 10x, and its transmittance was 20.%. Diluted sample "B" was diluted 20x, and its transmittance was 45%. In addition, diluted Sample "A" was spiked by adding 2.00 mL of WSS into a 100.0 mL flask, and adding diluted Sample "A" to the mark. The transmittance was found to be 18%. a. Using this data above, make a properly documented Beer Lambert Plot using Excel. Be sure to provide a trendline, formula and R2 value. What is the linear working range of your calibration curve in concentration? (5 marks) b. Determine the amount of chromium in the unknown solid in ppm and % w/w. (8 marks) c. Did you experience matrix effects? Show all your math.(4 marks) d. Do you have confidence in the results? Explain with detail. (4 marks) An analyst prepares a stock Cr(III)nitrate standard (SSS) by dissolving 1.0000g of dry chromium nitrate into a 1.00 L volumetric flask, and adding 2% nitric acid to the mark. From this stock solution, a working stock solution (WSS) was prepared by taking 10.0 mL of SSS and diluting it to 500.0 mL. Chromium calibration standards were then prepared as follows: Standard mL. WSS Volume of Flask %T (mL) 1 0.0 50.0 100. 23450 1.0 50.0 89 2.0 50.0 79 4.0 50.0 63 8.0 50.0 38 6 16.0 50.0 16 7 32.0 50.0 5.0 Use MW: H=1 C=12 N=14 O=16 Cr=52 Cu 63.5 These calibration standards were analyzed spectrophotometrically. The resulting transmittance data is detailed above. An unknown sample containing chromium was prepared by dissolving 0.3125 g of sample and adding 10.0 mL of concentrated nitric acid. The solution was heated until the sample dissolved. After cooling, the sample volume was adjusted to 250.0 mL. The transmittance of this solution was found to be 8%. Portions of the sample were diluted as follows: Diluted sample "A" was diluted 10x, and its transmittance was 20.%. Diluted sample "B" was diluted 20x, and its transmittance was 45%. In addition, diluted Sample "A" was spiked by adding 2.00 mL of WSS into a 100.0 mL flask, and adding diluted Sample "A" to the mark. The transmittance was found to be 18%. a. Using this data above, make a properly documented Beer Lambert Plot using Excel. Be sure to provide a trendline, formula and R2 value. What is the linear working range of your calibration curve in concentration? (5 marks) b. Determine the amount of chromium in the unknown solid in ppm and % w/w. (8 marks) c. Did you experience matrix effects? Show all your math.(4 marks) d. Do you have confidence in the results? Explain with detail. (4 marks)
Expert Answer:

Related Book For 

Posted Date:
Students also viewed these chemical engineering questions
-
The following additional information is available for the Dr. Ivan and Irene Incisor family from Chapters 1-5. Ivan's grandfather died and left a portfolio of municipal bonds. In 2012, they pay Ivan...
-
CANMNMM January of this year. (a) Each item will be held in a record. Describe all the data structures that must refer to these records to implement the required functionality. Describe all the...
-
What are the possible coping strategies that could help freelancers to overcome financial challenges and improve their financial well-being? Is there any significant impact in the financial...
-
Give 2 reasons why US national health reform became such heated debate for decades.
-
What are three methods for estimating stand-alone selling prices of goods and services that normally are not sold separately?
-
A cylinder having the dimensions shown is made from magnesium Am 1004-T61. If it is struck by a rigid block having a weight of \(800 \mathrm{lb}\) and traveling at \(2 \mathrm{ft} / \mathrm{s}\),...
-
Springfield Corporation operates on a calendar-year basis. It begins the annual budgeting process in late August, when the president establishes targets for total sales dollars and net operating...
-
A 2 m rope can support a 500 kg object hanging stationary before snapping. You decide to use it to spin a 100 kg mass in a vertical circle. a. How fast can you spin the object before the rope snaps?...
-
1. (4) A blood pressure of 110 mm Hg is measured using a cuff over the wrist which is positioned 30 cm above heart level. According to Bernoulli's equation(given below), what is the blood pressure at...
-
Bill, aged 7, has severe recurrent verbal and/or physical temper outbursts that are grossly out of proportion in intensity or duration to the situation or provocation and that are not developmentally...
-
2. A car starts from rest and travels along a straight road (x(0) = v(0) = 0). Its engine provides a constant acceleration of 20 ft/s. The combination of air and road resistance provides a...
-
An air-filled capacitor consists of two parallel plates, each with an area of 7.60 cm, separated by a distance of 2.00 mm. If a 16.0 V potential difference is applied to these plates, calculate the...
-
a) Determine the rate on the FRA(6x9) contract if the 6-month and 9-month LIBOR rates are 2.00% and 2.10%, respectively. Use day-counts of 182 and 272 for the two LIBOR rates b) Determine the dollar...
-
10. An unpowered projectile is fired vertically upwards from the surface of the Moon. To what height above the surface will the projectile rise if it leaves the surface at 1500 m/s?**
-
A rocket launches upwards, accelerating at 5 m/s. If Its initial velocity is 0 m/s, how high does the rocket reach after 10 seconds?
-
For a fixed number of processes, SJF scheduling algorithm: O a. Minimize average waiting time. O b. Only Minimize average waiting time and Minimize average turn-around time O. Only Maximizes CPU...
-
A random sample of 10 houses heated with natural gas in a particular area, is selected, and the amount of gas (in therms) used during the month of January is determined for each house. The resulting...
-
Consider the compounds butanoic acid, pentanal, M-hexane, and 1-pentanol. The boiling points of these compounds (in no specific order) are 69C, I03C, 137C, and 164C. March the boiling points to the...
-
Calculate the sodium ion concentration when 70.0 mL of 3.0 M sodium carbonate is added to 30.0 mL of 1.0 M sodium bicarbonate.
-
Calculate the wavelength of light emitted in each of the following spectral transitions in the hydrogen atom. What type of electromagnetic radiation is emitted in each transition? a. \(n=3...
-
Fill in the Blank. __________ equation involves time-dependent coefficients.
-
Fill in the Blank. When finite amplitudes of motion are involved, __________ analysis becomes necessary.
-
Fill in the Blank. __________ principle is not applicable in nonlinear analysis.

Study smarter with the SolutionInn App


