An analytical chemist is titrating 168.4 mL of a 0.7600M solution of aniline (C,H,NH,) with a...
Fantastic news! We've Found the answer you've been seeking!
Question:
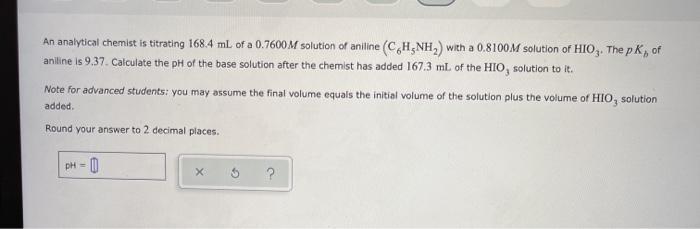
Transcribed Image Text:
An analytical chemist is titrating 168.4 mL of a 0.7600M solution of aniline (C,H,NH,) with a 0.8100M solution of HIO,. The p K, of aniline is 9.37. Calculate the pH of the base solution after the chemist has added 167.3 mL of the HIO, solution to it. Note for advanced students: you may assume the final volume equals the initial volume of the solution plus the volume of HIO, solution added. Round your answer to 2 decimal places. pH = 0 %3! An analytical chemist is titrating 168.4 mL of a 0.7600M solution of aniline (C,H,NH,) with a 0.8100M solution of HIO,. The p K, of aniline is 9.37. Calculate the pH of the base solution after the chemist has added 167.3 mL of the HIO, solution to it. Note for advanced students: you may assume the final volume equals the initial volume of the solution plus the volume of HIO, solution added. Round your answer to 2 decimal places. pH = 0 %3!
Expert Answer:
Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
An analytical chemist is titrating 53.9 mL of a 0.6000 M solution of trimethylamine ((CH,),N) with a 0.7900 M solution of HNO3. The p K, of 3. trimethylamine is 4.19. Calculate the pH of the base...
-
An analytical chemist is titrating 117.3 ml of a 0.2700 M solution of butanoic acid (HC,H,CO,) with a 1.200 M solution of KOH, ThepK, of butanoic acid is 4.82. Calculate the pH of the acid solution...
-
An analytical chemist is titrating 60.6 mL of a 0.5600 M solution of formic acid (H,CO,) with a 0.3100 M solution of KOH. The p K, of formic acid is 3.74. Calculate the pH of the acid solution after...
-
What items should each staff member receive when beginning a veterinary job position?
-
Blue Box is testing a new "half price on Tuesday" policy on DVD rentals at a sample of 10 locations. (a) At = .10, do the data show that the mean number of Tuesday rentals has increased? (b) Is the...
-
What are the general rules for how gains or losses on retirement of plant assets should be reported in income?
-
How many degrees of freedom are there? Exercises 49 refer to the following data: Electric motors are assembled on four different production lines. Random samples of motors are taken from each line...
-
Aaron, a resident of Minnesota, has been a driver for Green Delivery Service for the past six years. For this purpose, he leases a truck from Green, and his compensation is based on a percentage of...
-
A new ALP Government decides to implement more meaningful climate policy. It passes the Safe and Clean Environment Act 2023 (Cth). The legislation regulates a diverse range of activities including...
-
Peanut Company acquired 90 percent of Snoopy Company's outstanding common stock for $270,000 on January 1, 20X8, when the book value of Snoopy's net assets was equal to $300,000. Problem 3-34...
-
What will you do to make your objectives clear and candid during the conversation? How will you manage your emotion during the conversation while still maintaining empathy and trust with the...
-
1. A rationale for your intervention that is grounded in the research literature (-2 pages). This rationale should include a minimum of 5 annotated references from peer reviewed sources. This means...
-
When goods are produced privately, but the cost of their purchase is paid for by the taxpayer or some other third party, a . consumers have a strong incentive to search out those firms offering them...
-
a) Nyamakima group of companies has a sponsored a weight loss programme for mothers aged 18-40 years who are obese. The programme nutritionist has set up minimum daily requirement for several kind of...
-
Manufacturing costs in February 1 beginning inventory: Variable Fixed Cost 4,800 $40.00 $192,000 4,800 15.00 $55.00 72,000 $264,000 Total Manufacturing costs in February: Variable Fixed Total 48,000...
-
In addition to the mentoring and networking, Techstars provided $18,000 of cash to purchase what percent of the common shares? a. If you ignore the value of the non-financial support, Techstars is...
-
Consider a system of coupled ordinary differential equations (ODEs) representing a set of chemical reactions: dx/dt= -0.1x + 0.02y dy/dt= 0.1x - 0.02y - 0.05z dz/dt= 0.05y - 0.1z Write a MATLAB...
-
A number of years ago the United Food and Commercial Workers Union organized 800 workers of the 1035 employees at one of the Wilson Brothers food operations in Toronto, Ontario. The employees include...
-
Arrange the members of each of the following pairs in order of increasing radius and explain the order: a. Sr, Sr2+ b. Br, Br
-
Which has the larger radius, S or S2? Explain.
-
Consider this cell running under standard conditions: Ni(s) |Ni2 + (aq)||Cu + (aq) | Cu(s) a. Is this cell a voltaic or an electrolytic cell? How do you know? b. Does current flow in this cell...
-
A cylindrical tank of methanol has a mass of \(60 \mathrm{~kg}\) and a volume of \(75 \mathrm{~L}\). Determine the methanol's weight, density, and specific gravity. Take the gravitational...
-
A mass of \(0.5-\mathrm{kg}\) of argon is maintained at \(1400 \mathrm{kPa}\) and \(40^{\circ} \mathrm{C}\) in a tank. What is the volume of the tank?
-
The air in an automobile tire with a volume of \(0.015 \mathrm{~m}^{3}\) is at \(30^{\circ} \mathrm{C}\) and \(140 \mathrm{kPa}\) (gage). Determine the amount of air that must be added to raise the...

Study smarter with the SolutionInn App



