Aphoton of wavelength . when absorbed by a 2-D rigid rotor. takes it from first excited...
Fantastic news! We've Found the answer you've been seeking!
Question:
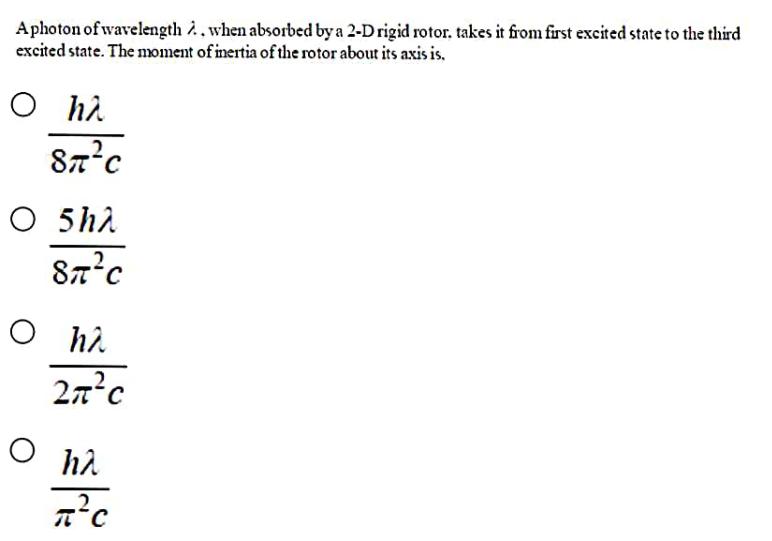
Transcribed Image Text:
Aphoton of wavelength . when absorbed by a 2-D rigid rotor. takes it from first excited state to the third excited state. The moment of inertia of the rotor about its axis is. O h 87C O 5 h 8C O ha 27C O ha 2 Aphoton of wavelength . when absorbed by a 2-D rigid rotor. takes it from first excited state to the third excited state. The moment of inertia of the rotor about its axis is. O h 87C O 5 h 8C O ha 27C O ha 2
Expert Answer:
Related Book For 

Posted Date:
Students also viewed these physics questions
-
The treatment of a particle in a one-dimensional box can be extended to a two-dimensional box of dimensions Lx and Ly yielding the following expression for energy: The two quantum numbers...
-
The ionization energy of a certain element is 412 kJ/mol. When the atoms of this element are in the first excited state, however, the ionization energy is only 126 kJ/mol. Based on this information,...
-
The following additional information is available for the Dr. Ivan and Irene Incisor family from Chapters 1-5. Ivan's grandfather died and left a portfolio of municipal bonds. In 2012, they pay Ivan...
-
Jack Williams works in a very active department called purchasing. He works with store managers, marketing, and supply companies. The people in his department try to have the right product, in the...
-
In 1980, empirical studies indicated that the sever was the bottleneck in a remote file system. Assume a stateless file server such as Sun NFS using a packet-level protocol such as raw IP....
-
OBB Personenverkehr (OBB), the Austrian state-owned railway, carries about 235 million passengers a year on routes within wholly owned by OBB Holding Group, which in turn, is wholly owned by the...
-
On November 15, 2010, Chandlers Department Store purchased $5,100 of inventory on account from one of its suppliers. The terms were 3/15, n/45, FOB shipping point. On November 18 Chandlers Department...
-
A Bode diagram for a process, valve, and sensor is shown in figure.(a) Determine an approximate transfer function for this system.(b) Suppose that a proportional controller is used and that a value...
-
Grey Company would like to increase its operational efficiency. For the first quarter of operations during the current year, the following data were reported: Inspection time Process time Move time...
-
When developing a marketing strategy using new digital media, a marketer must be aware of the strengths and weaknesses of these new media. Digital media are relatively new to the field of marketing...
-
a) A vector field = 3xyz, + x za, + (x'y 2z)a, is given. For this vector field, i) Find Divergence ii) Check for solenoidality ii) Investigate rotationality b) Fort he path = 2a, + ag + , in the...
-
List and describe three legal requirements that you must adhere to at work. Describe how you adhere to these at work. List and describe three ethical requirements that you must adhere to at work....
-
I would like for each of you to explore the topic of your Cultural Competence Assignment with your peers. Please provide a small overview of the population you chose, why this population was...
-
INSTRUCTIONS research one aspect/topic of occupational health and safety law. Once completed, make an essay. The written report: Your report should be within three to ten pages. Your report should...
-
What is managed health care? What are the three types of managed health care plans? What are the advantages and disadvantages of each? Describe the basic intention of the Patient Protection and...
-
December 31, 2022: Reported $60,000 net income for the year and closed net income, other comp income and dividend accounts. December 31, 2022 Prepare the entry to close net income. Account Retained...
-
On January 1, Year 1, Lester Brown Inc. (LBI) leased a new excavator from Double J Corp. The fair market value of the excavator on January 1, Year 1, is $490,000. Relevant details regarding the lease...
-
State whether each statement is true or false. If false, give a reason. {purple, green, yellow} = {green, pink, yellow}
-
Which of the compounds in Exercise 13 exhibit cis-trans isomerism?
-
The equilibrium constant is 0.0900 at 25oC for the reaction H2O(g) + Cl2O(g) 2HOCl(g) For which of the following sets of conditions is the system at equilibrium? For those which are not at...
-
Sketch the galvanic cells based on the following half-reactions. Calculate Ïo, show the direction of electron flow and the direction of ion migration through the salt bridge, identify the...
-
Fill in the Blank. In the finite element method, \(a(n)\) ___________ solution is assumed within each element.
-
The stiffness matrix of a bar element is given by a. \(\frac{E A}{l}\left[\begin{array}{ll}1 & 1 \\ 1 & 1\end{array} ight]\) b. \(\frac{E A}{l}\left[\begin{array}{rr}1 & -1 \\ -1 & 1\end{array}...
-
What is the basis for the derivation of transformation matrices?

Study smarter with the SolutionInn App



