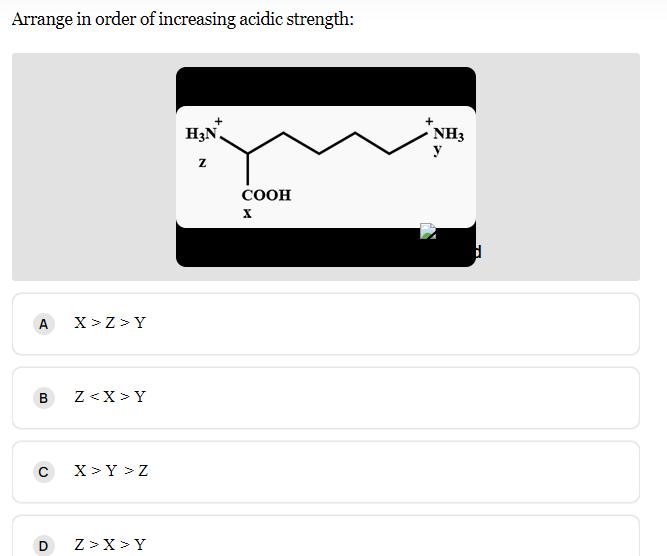
Arrange in order of increasing acidic strength: A X>Z>Y B Z Y C X>Y > Z...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
Arrange in order of increasing acidic strength: A X>Z>Y B Z<X>Y C X>Y > Z D Z>X>Y H3N. Z COOH X NH3 y d Arrange in order of increasing acidic strength: A X>Z>Y B Z<X>Y C X>Y > Z D Z>X>Y H3N. Z COOH X NH3 y d
Expert Answer:
Related Book For 

Posted Date:
Students also viewed these accounting questions
-
Arrange each group of compounds in order of increasing acidity. (a) Phenol, ethanol, acetic acid (b) P-toluenesulfonic acid, acetic acid, chloroacetic acid (c) Benzoic acid, o-nitrobenzoic acid,...
-
(a) Assuming standard conditions, arrange the following in order of increasing strength as oxidizing agents in acidic solution: Cr2O72-, H2O2, Cu2+, Cl2, O2. (b) Arrange the following in order of...
-
Arrange the following compounds in order of increasing boiling point. Explain your answer in terms of the intermolecular forces in each compound. (a) (b) (c) (d)
-
This section contains 6 questions. Questions 83 to 88 have four statements (A, B C and D) given in Column I and four statements (p, q, r and s) in Column II. Any given statement in Column I can have...
-
The following data summarizes the per-day cost of crashing the activities in the problem described in the previous question. a. Create an LP model to determine the least costly way of crashing the...
-
Show the calculation of the following financing ratios: (1) The debt to equity ratio, (2) The times interest earned ratio.
-
Which country was the first to require the audit of corporations, England or the United States?
-
Lawsons Department Store faces a buying decision for a seasonal product for which demand can be high, medium, or low. The purchaser for Lawsons can order 1, 2, or 3 lots of the product before the...
-
Bond J is a 3 percent coupon bond. Bond K is a 9 percent coupon bond. Both bonds have 7 years to maturity, make semiannual payments, and have a YTM of 6 percent. If interest rates suddenly rise by 5...
-
I. THE CASE 0 n February 15, 2019, the Chief Operating Officer of Larson Industries, Pete Moreen, announced a major step forward for the company: Today, we have entered into an agreement to sell our...
-
Cost and sales information for the most recent fiscal year are shown below. WALLACE RIVER COMPANY Cost and Sales Information For the Year Ended December 31 Purchases of raw materials Raw materials...
-
Contribution Margin Income Statement Sales (440,000 units) Variable costs Direct materials Direct labor Overhead Contribution margin Fixed costs Fixed overhead Fixed general and administrative $...
-
At Clarkson Corp., the assembly department's April static overhead budget shows total overhead costs of $89,000. $19,000 of those costs are variable, and the rest are fixed. Direct labor hours is the...
-
3. What will be the radiation intensity from 2590 GBq CO-60 source (SEC above) be at a distance of 5 meters? How many TVL-s is needed to reduce its intensity to 0.025 mSv/h? Hint: 1st Solve for...
-
On Jan - 1 , Our Business the Sharks Corp Issued 7 , 0 0 0 Bonds at 1 0 3 . 5 0 % Each Bond had a Face Value of $ 1 , 0 0 0 . The Maturity Date is 2 0 years from Now The Face Rate of Interest is 4 ....
-
A manufacturer completed a business income work sheet for the recent policy period based on operating 6 months per year (after plant summer shutdown and usual employee paid holidays). The annual...
-
Explain the importance of ethical and multicultural competency to the practice of professional psychology. Identify and explain personal limitations you realized as a result of the Quality and...
-
Saccharin is an artificial sweetener that is used in diet beverages. In order for it to be metabolized by the body, it must pass into cells. Below are shown the two forms of saccharin. Saccharin has...
-
Which of the following compounds are chiral? Draw each compound in its most symmetric conformation, star (*) any asymmetric carbon atoms, and draw any mirror planes. Label any meso compounds. You may...
-
Give logical fragmentation reactions to account for the following ions observed in these mass spectra. (a) N-octane: 114, 85, 71, 57 (b) Methylcyclohexane: 98, 83 (c) 2-methylpent-2-ene: 84, 69 (d)...
-
Deuterium (D) is the isotope of hydrogen of mass number 2, with a proton and a neutron in its nucleus. The chemistry of deuterium is nearly identical to the chemistry of hydrogen, except that the C(D...
-
You have been asked to value the practice of Dr. Vong, a pediatrician in your town, and are provided with the following facts: The practice generated $800,000 in revenues last year, and these...
-
You are trying to decide how much you should bid on a Ken Griffey Jr. rookie baseball card in good condition on eBay. You notice that there have been eight transactions involving Ken Griffey Jr....
-
A company is considering delaying a project with after-tax cash flows of \($25\) million but that costs \($300\) million to take on (the life of the project is 20 years, and the cost of capital is...

Study smarter with the SolutionInn App



