Arrange the acids shown from lowest pk, to highest pKa by clicking and dragging them into...
Fantastic news! We've Found the answer you've been seeking!
Question:
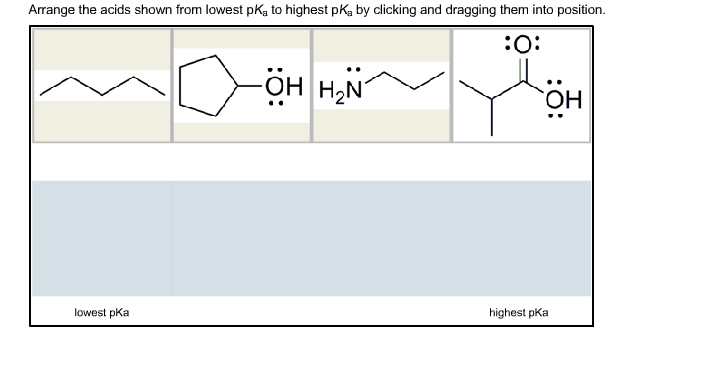
Transcribed Image Text:
Arrange the acids shown from lowest pk, to highest pKa by clicking and dragging them into position. :O: lowest pka OH H₂N ОН OH highest pka Arrange the acids shown from lowest pk, to highest pKa by clicking and dragging them into position. :O: lowest pka OH H₂N ОН OH highest pka
Expert Answer:
Answer rating: 100% (QA)
Answer THE ACID WHICH IS STRONGER HAS LOWEST PKa value due to resonance sta... View the full answer

Related Book For 

Organic Chemistry structure and function
ISBN: 978-1429204941
6th edition
Authors: K. Peter C. Vollhardt, Neil E. Schore
Posted Date:
Students also viewed these chemistry questions
-
Arrange the following from lowest to highest ionization energy: O, O2, O2-, O2+. Explain your answer.
-
The block is deformed into the position shown by the dashed lines. Determine the average normal strain along line AB. 30 mm 30 mm 70 mm 15 mm 55 mm --B': 100 mm 110 mm 70 mm 130 mml
-
The square deforms into the position shown by the dashed lines. Determine the shear strain at each of its corners, A, B, C, and D, relative to the x, y axes. Side D' B' remains horizontal. 3 mm D' D...
-
The current zero-coupon yield curve of semi-annually compounded rates for risk-free bonds is as follows: 1.0 Years 1.5 Years 2.0 Years 2.5 Years 3.0 Years 9.00% 10.20% 3.00% 4.00% 6.20% MATURITY 0.5...
-
Interpret the following GLM results. Following from an example in the chapter, Performance is the performance rating for a business unit manager. Sales are a measure of the average sales for that...
-
On August 1, 2020, Mark Diamond began a tour company in the Northwest Territories called Millennium Arctic Tours. The following occurred during the first month of operations: Required 1. Prepare...
-
The following accounts and December 31, 2006, balances of New York Optical Corporation are arranged in no particular order. Requirements 1. Prepare the company's classified balance sheet in account...
-
The following data relate to notes receivable and interest for Owens Co., a financial services company. (All notes are dated as of the day they are received.) Mar. 8. Received a $33,000, 5%, 60-day...
-
Given the function sin x + cos y = 2y, find all critical points. Then use both the first and second derivative tests to classify these critical points as relative maxima or minima. (8 points)
-
The Foxridge Investment Group buys and sells rental income properties in southwest Virginia. Bill Hunter, president of Foxridge, has asked for your assistance in analyzing a small apartment building...
-
Discuss the negative impacts of social media on users or what type of negative impact is less likely talked about and what three scholarly articles ?
-
The following information was disclosed during the audit of Zheng Inc. 1. 2. On January 1, 2014, equipment costing 600,000,000 is purchased. For financial reporting purposes, the company uses...
-
Samson Corporation issued a 4-year, 75,000, zero-interest-bearing note to Brown Company on January 1, 2015, and received cash of 47,664. The implicit interest rate is 12%. Prepare Samsons journal...
-
On January 1, 2015, Adams Corporation signed a 5-year, non-cancelable lease for a machine. The terms of the lease called for Adams to make annual payments of \($9\),968 at the beginning of each year,...
-
Hayes Co. reported the following pretax financial income (loss) for the years 20132017. Pretax financial income (loss) and taxable income (loss) were the same for all years involved. The enacted tax...
-
On July 1, 2016, Fontaine Company purchased for cash 40% of the outstanding ordinary shares of Knoblett Company. Both Fontaine Company and Knoblett Company have a December 31 year-end. Knoblett...
-
Predict the spring constant for a helical coil spring whose coil has an outside diameter of 20.6 mm, wire diameter of 2.1 mm, a count of 35.5 active coils and a modulus of 77.2 GPa. Report the result...
-
The Cholesterol Level data sets give cholesterol levels of heart attack patients. Cholesterol measures are taken 2, 4, and 14 days aft er a patient has suffered a heart attack. Is there a significant...
-
(2S, 3S)-3-Hydroxyleucine is an amino acid (Chapter 26) that is a key component in the structures of many "depsipeptide" antibiotics, such as sanjoining (margin), (a) Find the part of the sanjoinine...
-
(a) Write the expected product of the reaction of 3-octyne with Na in liquid NH 3 . (b) When the same reaction is carried out with cyclooctyne (Problem 33b), the product is cis-cyclooctene, not...
-
For each reaction in Problem 29, write out a detailed step-by-step mechanism?
-
Derive the frequency equation for the longitudinal vibration of the systems shown in Fig. 8.28. M p. A, E.1 p.A. E,I M k p. A. E,I ell M k (a) (b) FIGURE 8.28 Bar with different end conditions. (c)
-
Fill in the Blank. The ____________ energy of a beam is denoted by \(\frac{1}{2} \int_{0}^{l} E I\left(\frac{\partial^{2} w}{\partial x^{2}} ight)^{2} d x\).
-
A thin bar of length \(l\) and mass \(m\) is clamped at one end and free at the other. What mass \(M\) must be attached to the free end in order to decrease the fundamental frequency of longitudinal...

Study smarter with the SolutionInn App


