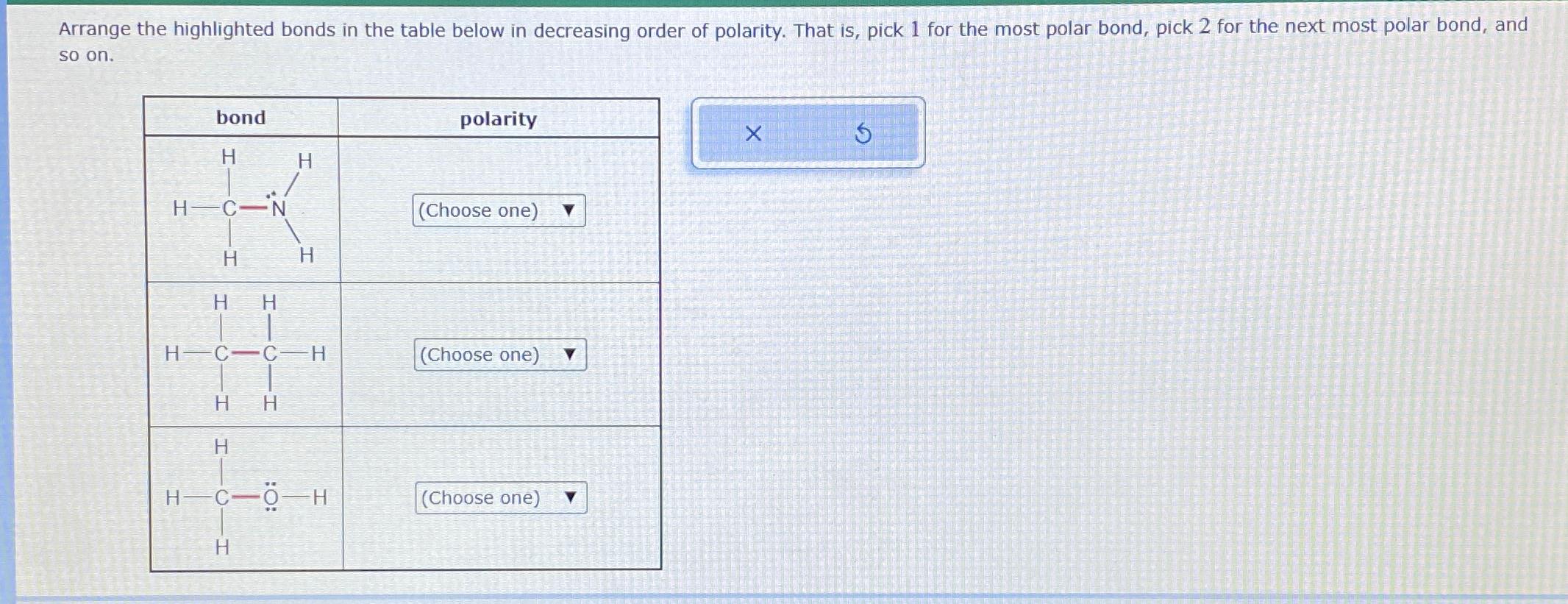
Arrange the highlighted bonds in the table below in decreasing order of polarity. That is, pick...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
Arrange the highlighted bonds in the table below in decreasing order of polarity. That is, pick 1 for the most polar bond, pick 2 for the next most polar bond, and so on. bond H H-C- H H H H H H C C-H H H H H HC-H polarity (Choose one) (Choose one) (Choose one) X Arrange the highlighted bonds in the table below in decreasing order of polarity. That is, pick 1 for the most polar bond, pick 2 for the next most polar bond, and so on. bond H H-C- H H H H H H C C-H H H H H HC-H polarity (Choose one) (Choose one) (Choose one) X
Expert Answer:
Answer rating: 100% (QA)
The image presented shows a question asking to arrange the highlighted bonds in decreasing order of ... View the full answer

Related Book For 

Income Tax Fundamentals 2013
ISBN: 9781285586618
31st Edition
Authors: Gerald E. Whittenburg, Martha Altus Buller, Steven L Gill
Posted Date:
Students also viewed these chemical engineering questions
-
Find general solutions of the systems in Problems 1 through 22. In Problems 1 through 6, use a computer system or graphing calculator to construct a direction field and typical solution curves for...
-
Marketing, Celebrity Endorsement, and Market Share - are there relationships in the BSG among these three factors? If so, what are they? Is there something missing from this list that also influences...
-
For the following exercises, suppose a weight is attached to a spring and bobs up and down, exhibiting symmetry. At time = 0, what is the displacement of the weight? y 5- 4- 3 2+ 1- -1- -2- -3- -4- ...
-
What is the difference between an optimistic approach and a pessimistic approach to decision making under assumed uncertainty?
-
To the degree job growth (and increased car sales that come from more competitive labor costs) is based on two tier-wage structures, how sustainable is this approach? Consider, for example, that Fiat...
-
What is cash flow from operating activities? Give examples.
-
Which of the following is an activity not usually associated with forensic accounting and fraud examination consulting and litigation support? 1. A. Assessing fraud risk associated with internal...
-
OGrady Apparel Company was founded nearly 160 years ago when an Irish merchant named Garrett OGrady landed in Los Angeles with an inventory of heavy canvas, which he hoped to sell for tents and wagon...
-
An l-regular graph, l 1, is a simple graph where all of its vertices have degree l. The union of two simple graphs G G U G2 = (V U V2, E1 U E2). = (V1, E1) and G2 = (V2, E2), is the simple graph Two...
-
Fourth-Sixth-Twelfth Bank currently pays an annuall dividend of $1.40 per share. After extensive analysis, you forecast that the Bank's stock will continue to pay the $1.40 dividend for four years....
-
1. Consider the following statements and select the correct option regarding simple epithelium? Statement 1: Cuboidal epithelium help in secretion and absorption. Statement 2: Squamous epithelium...
-
Direct financing allows investors to easily: a. Diversify their investment portfolio b. Lower search and transaction costs c. Expand their portfolios d. Match amounts and maturity of...
-
Here is a Java programming question: Note: Use s.hasNextLine for multiple lines and terminate the input with "end" Don't use string.split or arrays, just use flow controls, loops and iterations. The...
-
What is negative carry? What does this mean and why is it important to a hedging strategy?
-
Write a formal business letter, to be sent out to one of your stakeholders congratulating them on completing 10 years of business together. the business letter must have a practical application, so...
-
Mortgage-backed securities are believed to have contributed to the recent subprime mortgage crisis. Assess the fairness of this statement, providing a rationale for your reasoning and a...
-
Select a peer-reviewed article that is focused on one of the following subject areas: Port Security The vulnerability of the Maritime Transportation Security (MTS) Vulnerability to world shipping...
-
To balance the chemical equation SiH3 + O2 SiO2 + HO, you could introduce coefficients a, b, c, d and write aSiH3 + bO2 cSiO + dHO then write linear equations for each element. The equation for Si...
-
Ken (age 31) and Amy (age 28) Booth have brought you the following information regarding their income, expenses, and withholding for the year. They are unsure which of these items must be used to...
-
Sally and Charles Heck received the following dividends and interest during 2012: Assuming the Hecks file a joint tax return, complete Schedule B of Form 1040 (on page 2-33) for them for the 2012 tax...
-
Tom has a successful business with $100,000 of income in 2012. He purchases one new asset in 2012, a new machine which is 7-year MACRS property and costs $25,000. If you are Tom's tax advisor, how...
-
A thermally isolated system at constant pressure consists of \(10 \mathrm{~kg}\) of air at a temperature of \(1000 \mathrm{~K}\) and \(10 \mathrm{~kg}\) of water at \(300 \mathrm{~K}\), connected...
-
An incompressible liquid of specific volume \(v_{l}\), is in equilibrium with its own vapour and an inert gas in a closed vessel. The vapour obeys the law \[p(v-b)=\Re T\] Show that \[\ln...
-
Show that if a liquid is in equilibrium with its own vapour and an inert gas in a closed vessel, then \[\frac{\mathrm{d} p_{v}}{\mathrm{~d} p}=\frac{ho_{v}}{ho_{l}}\] where \(p_{v}\) is the partial...

Study smarter with the SolutionInn App


