Question
Assign formal charges to the atoms in these three structures (only necessary for atoms with nonzero formal charge). Do not add or delete bonds
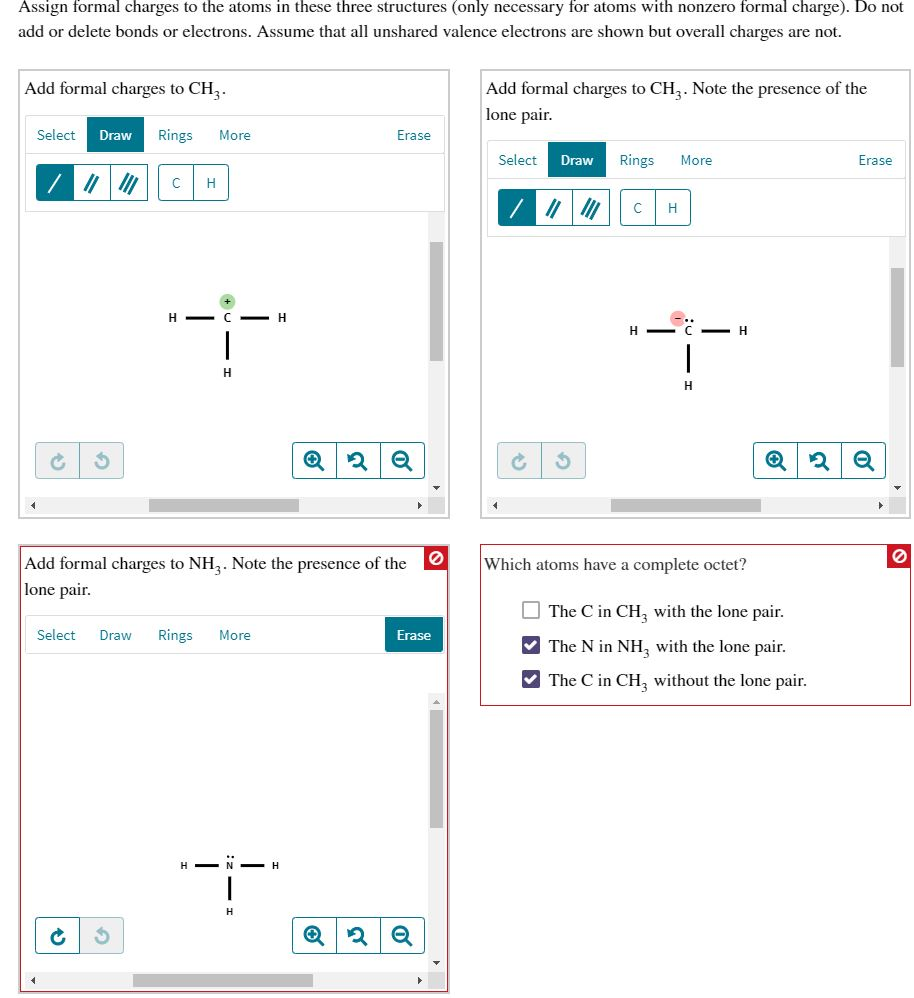
Assign formal charges to the atoms in these three structures (only necessary for atoms with nonzero formal charge). Do not add or delete bonds or electrons. Assume that all unshared valence electrons are shown but overall charges are not. Add formal charges to CH. Select Draw /// 3 Rings More C H H- C-H 44- H Select Draw Rings More Add formal charges to NH3. Note the presence of the lone pair. i Erase -H 2 Q 6 Erase Q 2 Q Add formal charges to CH3. Note the presence of the lone pair. Select Draw Rings 1101 3 C H- H More H H Which atoms have a complete octet? Erase Q2 Q The C in CH with the lone pair. The N in NH, with the lone pair. The C in CH3 without the lone pair.
Step by Step Solution
3.44 Rating (157 Votes )
There are 3 Steps involved in it
Step: 1
9 Date foumal charge 1 H b H h C HNH 1 80 N H Saathi vale...
Get Instant Access to Expert-Tailored Solutions
See step-by-step solutions with expert insights and AI powered tools for academic success
Step: 2

Step: 3

Ace Your Homework with AI
Get the answers you need in no time with our AI-driven, step-by-step assistance
Get StartedRecommended Textbook for
Organic Chemistry
Authors: Graham Solomons, Craig Fryhle, Scott Snyder
11th edition
1118133579, 978-1118133576
Students also viewed these Chemistry questions
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
View Answer in SolutionInn App



