Assign formal charges to the atoms in these three structures (only necessary for atoms with nonzero...
Fantastic news! We've Found the answer you've been seeking!
Question:
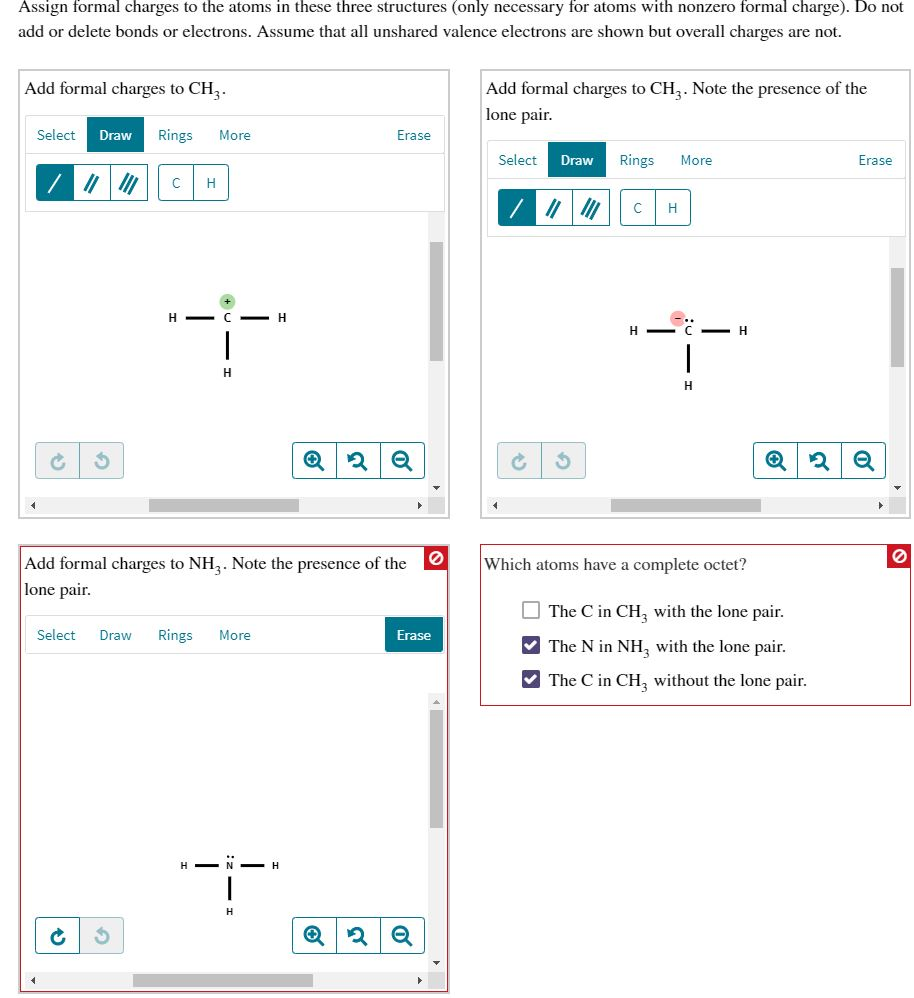
Transcribed Image Text:
Assign formal charges to the atoms in these three structures (only necessary for atoms with nonzero formal charge). Do not add or delete bonds or electrons. Assume that all unshared valence electrons are shown but overall charges are not. Add formal charges to CH₂. Select Draw /// Ć 3 Rings More C H H- C-H 44- H Select Draw Rings More Add formal charges to NH3. Note the presence of the lone pair. iדי Erase -H ✪ 2 Q 6 Erase Q 2 Q Add formal charges to CH3. Note the presence of the lone pair. Select Ć Draw Rings 1101 3 C H- H More H H Which atoms have a complete octet? Erase Q2 Q The C in CH₂ with the lone pair. The N in NH, with the lone pair. ✔The C in CH3 without the lone pair. Assign formal charges to the atoms in these three structures (only necessary for atoms with nonzero formal charge). Do not add or delete bonds or electrons. Assume that all unshared valence electrons are shown but overall charges are not. Add formal charges to CH₂. Select Draw /// Ć 3 Rings More C H H- C-H 44- H Select Draw Rings More Add formal charges to NH3. Note the presence of the lone pair. iדי Erase -H ✪ 2 Q 6 Erase Q 2 Q Add formal charges to CH3. Note the presence of the lone pair. Select Ć Draw Rings 1101 3 C H- H More H H Which atoms have a complete octet? Erase Q2 Q The C in CH₂ with the lone pair. The N in NH, with the lone pair. ✔The C in CH3 without the lone pair.
Expert Answer:
Answer rating: 100% (QA)
9 Date foumal charge 1 H b H h C HNH 1 80 N H Saathi vale... View the full answer

Related Book For 

Organic Chemistry
ISBN: 978-1118133576
11th edition
Authors: Graham Solomons, Craig Fryhle, Scott Snyder
Posted Date:
Students also viewed these chemistry questions
-
Assign formal charges to the atoms in the following structures. Which of the two do you think is the more important contributor to the resonance hybrid. (a) H H C=N=N (b) H H C-N=N
-
Assign oxidation numbers to the atoms in H3PO4.
-
Assign oxidation numbers to the atoms in each substance. a. Kr (krypton) b. krypton tetrafluoride (KrF4) c. dioxygen difluoride (O2F2)
-
This exercise shows that the multiplier may be interpreted as a rate of change in general. Assume that the maximum of (x, y) subject to g(x, y) = c occurs at a point P. Then P depends on the value...
-
The Northwoods Manufacturing Company has automated its production facilities dramatically during the last five years, to the extent that the number of direct-labor hours has remained steady while...
-
What is the weight, in N, of an object with a mass of 200 kg at a location where g = 9.6 m/s 2 ?
-
The June 30, 2010, adjusted trial balance of Energized Espresso, Inc., is shown next. Requirements 1. Prepare the June closing entries for Energized Espresso, Inc. 2. Calculate the ending balance in...
-
A study was made by a retail merchant to determine the relation between weekly advertising expenditures and sales. The following data were recorded:(a) Plot a scatter diagram.(b) Find the equation of...
-
1. Duffer Resorts is redoing its golf course at a cost of $2,744,320. It expects to generate cash flows of $1,223,445, $2,007,812, and $3,147,890 over the next three years. If the appropriate...
-
Crane Library, a nonprofit organization, presented the following statement of financial position and statement of activities for its fiscal year ended February 28, 2024. Assets Current Assets Cash...
-
Briefly discuss the types of data that school counselors need to be mindful of in order to promote the academic success of all students Briefly discuss ways in which you plan to enhance your...
-
Lorenzo Company applies overhead to jobs on the basis of direct materials cost. At year - end, the Work in Process Inventory account shows the following. Work in Process Inventory Date Explanation...
-
29. E-Loan, an online lending service, offers a 36-month auto loan at 7.56% compounded monthly to applicants with good credit ratings. If you have a good credit rating and can afford
-
Create a case study where a person has breached at least four (4) articles from the REIC code of Ethics and your employer's code of conduct . Specifically, indicate which four (4) items were breached...
-
Ida Company produces a handcrafted musical instrument called a gamelan that is similar to a xylophone. The gamelans are sold for $ 8 8 0 . Selected data for the company s operations last year follow:...
-
Abbie bought a vacation property for $7,750 down and quarterly mortgage payments of $1224.51 at the end of each quarter for six years. Interest rate is 8.4% p.a. compounded quarterly. What was the...
-
A company that does business in different countries in order to apply the Geert Hofstede's six cross-cultural dimensions and discussing their influence to your company. You should compare the six...
-
Write a declaration for each of the following: a. A line that extends from point (60, 100) to point (30, 90) b. A rectangle that is 20 pixels wide, 100 pixels high, and has its upper-left corner at...
-
Provide a detailed mechanism for each of the following reactions. Include contributing resonance structures and the resonance hybrid for the arenium ion intermediates. (a) (b) (c) HNO HSO NO2 Br Bra,...
-
How would you transform tetradecanal into each of the following? (a) (b) (c) (d) CO2H )--C OH CO2H CO2H CN CO2 NH3
-
Compound A (C9H18O) forms a phenylhydrazone, but it gives a negative Tollens' test. The IR spectrum of A has a strong band near 1710 cm-1. The broadband proton decoupled 13C NMR spectrum of A is...
-
Which of the following best illustrates an error caused by sampling? a. A randomly chosen sample may not be representative of the population as a whole on the characteristic of interest. b. An...
-
The application of statistical sampling techniques is least related to which of the following generally accepted auditing standards? a. The work is to be adequately planned and assistants, if any,...
-
Which of the following is an error caused by sampling? a. Choosing an audit procedure that is inconsistent with the audit objective. b. Choosing a sample size that is too small to achieve the...

Study smarter with the SolutionInn App


