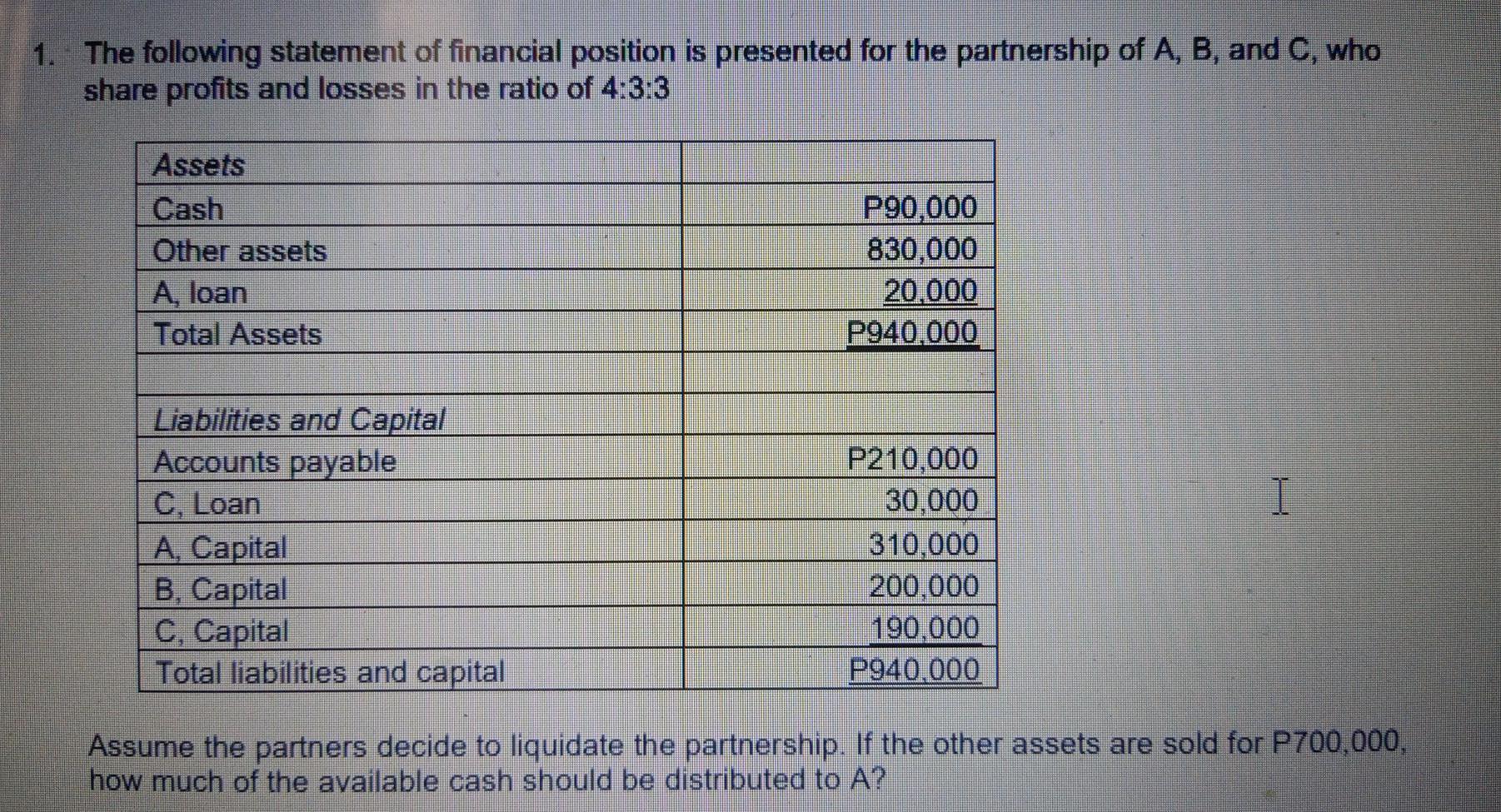
1. The following statement of financial position is presented for the partnership of A, B, and...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
1. The following statement of financial position is presented for the partnership of A, B, and C, who share profits and losses in the ratio of 4:3:3 Assets Cash Other assets A, loan Total Assets Liabilities and Capital Accounts payable C, Loan A, Capital B, Capital C, Capital Total liabilities and capital P90,000 830,000 20,000 P940,000 P210,000 30,000 310,000 200,000 190,000 P940,000 I Assume the partners decide to liquidate the partnership. If the other assets are sold for P700,000, how much of the available cash should be distributed to A? 1. The following statement of financial position is presented for the partnership of A, B, and C, who share profits and losses in the ratio of 4:3:3 Assets Cash Other assets A, loan Total Assets Liabilities and Capital Accounts payable C, Loan A, Capital B, Capital C, Capital Total liabilities and capital P90,000 830,000 20,000 P940,000 P210,000 30,000 310,000 200,000 190,000 P940,000 I Assume the partners decide to liquidate the partnership. If the other assets are sold for P700,000, how much of the available cash should be distributed to A?
Expert Answer:
Related Book For 

Financial Accounting for Decision Makers
ISBN: 978-0273763451
6th Edition
Authors: Peter Atrill, Eddie McLaney
Posted Date:
Students also viewed these accounting questions
-
Blackwell Automotive' s balance sheet at the end of its most recent fiscal year shows the following information: Blackwell Automotive Balance Sheet as of March 31, 2013 In addition, it was reported...
-
Forecast DP's 2020 income statement and balance sheet using the following relations ($ millions). Assume total revenues grow by 13% in 2020. All other percentages (other than sales growth and...
-
1. What is the pH of a solution that has an [H*] concentration of: a. 6.50 x 10-0 mol/L b. 1.00 x 10 mol/L 2. What is the [H*] concentration of a solution with pH of: a. 3.82 b. 11.11 3. Calculate...
-
What type of isomers are exhibited by [Fe(en) 3 ]Cl 2 (en = ethane-1,2-diamine)? no isomers are possible. cis and trans isomers fac and mer isomers optical isomers
-
a. Calculate K1 at 25C for phosphoric acid: b. Which thermodynamic factor is the most significant in accounting for the fact that phosphoric acid is a weak acid? Why? H3PO4(aq)--H"(aq) + H,PO4-(aq)...
-
Do there exist square matrices without eigenvalues?
-
The following MINITAB output presents a 95% confidence interval for the mean ozone level on days when the relative humidity is 60%, and a 95% prediction interval for the ozone level on a particular...
-
Use the data in Problem P11-48A to prepare the Crowley Cosmetics statement of retained earnings for the year ended December 31, 2012. Use the Statement of Retained Earnings for Maxim, Inc., in the...
-
Sprint buys a 1.60 $/ (strike) call on 300,000 for 0.10$/. Suppose that the exchange rate turns out to be 1.75 $/ at expiration. What is Sprint's overall profit or loss on the transaction?
-
Your marketing research firm is preparing the final written report on a research project commissioned by a major manufacturer of lawn mowers. One objective of the project was to investigate seasonal...
-
As sales manager, Joe Batista was given the following static budget report for selling expenses in the Clothing Department of Coronado Company for the month of October, CORONADO COMPANY Clothing...
-
Assume a 10-year Treasury bond has a coupon rate of 3.2 % and par value of $1000, and yield to maturity of 2%. Is this bond selling at a premium or discount? Explain why?
-
At the weekly farmers market in Atlanta, Sean, a local corn farmer, brings a truckload of fresh corn and sells it at a booth. It is more efficient for Sean to produce a large quantity of corn, but...
-
What are the advantages and disadvantages of organizing a business as a corporation? 2. How does a partnership differ from a limited liability company? 3. Why do corporates file for bankruptcy? 4....
-
Consider the following project's after-tax cash flow and the expected annual general inflation rate during the project period. End of Year Expected Cash Flow General Inflation 0 23 (in Actual $)...
-
Identify the primary audience for and mission of any organization or brand in Canada. 2. Find that organization on as many social platforms as you can and link to all their accounts. 3. Consider AT A...
-
3. (30 Pts) A hot water storage system is being designed in which a large thin-walled tank is warmed by a heat transfer fluid flowing through a jacket around the side walls of the tank. The system...
-
A police officer pulls you over and asks to search your vehicle because he suspects you have illegal drugs inside your car. Since he doesn't have reasonable suspicion to search your car, legally he...
-
What are the main methods of creative accounting?
-
The following is the statement of financial position of Davids business at 1 January of last year. The following is a summary of the transactions that took place during the year: 1 Inventories were...
-
What is an associate company and what are the consequences for accounting of one company being the associate company of a group of companies?
-
Through the implementation of efficient and effective management, New Belgium Brewing (NBB) has grown from operating out of the founder's basement to having two state-of-the-art facilities and over...
-
Define management and indicate what its principal purposes are and why managers are essential to organizations.
-
What are the resources of the organization? How are these used in management decision making?

Study smarter with the SolutionInn App



