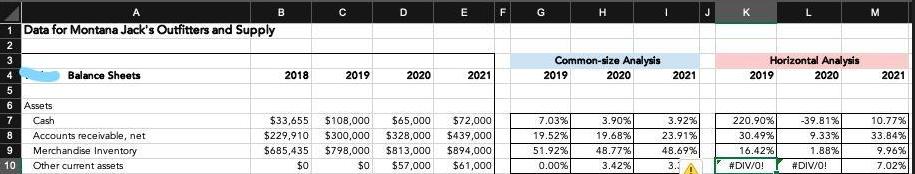
Calculate horizontal analysis change in percentage when the prior year has a zero balance 1 Data for
Fantastic news! We've Found the answer you've been seeking!
Question:
Calculate horizontal analysis change in percentage when the prior year has a zero balance
Transcribed Image Text:
1 Data for Montana Jack's Outfitters and Supply 3 4 Balance Sheets 5 6 Assets 7 Cash 8 Accounts receivable, net 9 Merchandise Inventory 10 Other current assets B 2018 O 2019 D 2020 E 2021 $33,655 $108,000 $65,000 $72,000 $229,910 $300,000 $328,000 $439,000 $685,435 $798,000 $813,000 $894,000 $0 $0 $57,000 $61,000 F Common-size Analysis 2019 H 7.03% 19.52% 51.92% 0.00% 2020 3.90% 19.68% 48.77% 3.42% 2021 Horizontal Analysis 2020 3.92% 23.91% 2019 220.90% 30.49% 48.69% 16.42% 3. #DIV/0! -39.81% 9.33% 1.88% #DIV/0! M 2021 10.77% 33.84% 9.96% 7.02% 1 Data for Montana Jack's Outfitters and Supply 3 4 Balance Sheets 5 6 Assets 7 Cash 8 Accounts receivable, net 9 Merchandise Inventory 10 Other current assets B 2018 O 2019 D 2020 E 2021 $33,655 $108,000 $65,000 $72,000 $229,910 $300,000 $328,000 $439,000 $685,435 $798,000 $813,000 $894,000 $0 $0 $57,000 $61,000 F Common-size Analysis 2019 H 7.03% 19.52% 51.92% 0.00% 2020 3.90% 19.68% 48.77% 3.42% 2021 Horizontal Analysis 2020 3.92% 23.91% 2019 220.90% 30.49% 48.69% 16.42% 3. #DIV/0! -39.81% 9.33% 1.88% #DIV/0! M 2021 10.77% 33.84% 9.96% 7.02%
Expert Answer:
Related Book For 

Income Tax Fundamentals 2013
ISBN: 9781285586618
31st Edition
Authors: Gerald E. Whittenburg, Martha Altus Buller, Steven L Gill
Posted Date:
Students also viewed these accounting questions
-
The following additional information is available for the Dr. Ivan and Irene Incisor family from Chapters 1-5. Ivan's grandfather died and left a portfolio of municipal bonds. In 2012, they pay Ivan...
-
A researcher wanted to find out if there was difference between older movie goers and younger movie goers with respect to their estimates of a successful actors income. The researcher first...
-
A survey of information systems managers was used to predict the yearly salary of beginning programmer/analysts in a metropolitan area. Managers specified their standard salary for a beginning...
-
10 1 Journalize the following transactions for Bigelow Company for the month of October. 31 Oct Stockholders invest cash in the company in exchange for common stock. 5 Oct The company buys a delivery...
-
A flat uniform circular disk has a mass of 3.00 kg and a radius of 70.0 cm. It is suspended in a horizontal plane by a vertical wire attached to its center. If the disk is rotated 2.50 rad about the...
-
The demand function of a good is Q = 100 P + 2Y + 1/2A where Q, P, Y and A denote quantity demanded, price, income and advertising expenditure, respectively. (a) Calculate the demand when P = 10, Y...
-
Because a client does not maintain perpetual records, an auditor elects to use mean- per-unit estimation to determine whether ending inventory is fairly stated. The follow- ing data are available:...
-
Bowery Company manufactures a single product that sells for $100 per unit. The company projects sales of 400 units per month. Projected costs follow. 1. Prepare a contribution margin income statement...
-
How has face book influenced political candidate's electoral success? What is the relationship between social media technology called face book and electoral success?
-
7. Copper Mines, Inc (CMI) . purchased property for the purpose of extracting copper ore. CMl paid $3,500,000 for the property plus $100,000 closing costs. CMl estimates it will be able to extract...
-
At the 0.05 level of significance, is there evidence of a difference in the mean rating of the pens following exposure to five advertisements?
-
Anna is running to the right, as shown in Figure Q3.19. Balls 1 and 2 are thrown toward her by friends standing on the ground. According to Anna, both balls are approaching her at the same speed....
-
A, B, and C are three similar plants under the same management who wants to merge them for better operation. The details are as under: You have to find out: (i) the capacity of the merged plant for...
-
Small Ltd. has been offered a choice to buy one out of two machines, A and B. You are required to compute: (a) Break-even point for each of the machines. (b) The level of sales at which both machines...
-
ABC Company Ltd. expects the following sales by months in units for the first six months of next year. The company has a policy of maintaining an inventory equal to budgeted sales for the following...
-
The Royal Industries Ltd. has prepared its annual sales forecast, expecting to achieve sales of 30,00,000 next year. The controller is uncertain about the pattern of sales to be expected by month and...
-
Janko Wellspring Incorporated has a pump with a book value of $29,000 and a four-year remaining life. A new, more efficient pump is available at a cost of $50,000. Janko can receive $8,500 for...
-
The company manufactures three products: wooden chairs, tables and dressers. AFC started off as a 'Mom & Pop' shop but has grown rapidly. AFC uses one assembly line to build all three products,...
-
Joan is a self-employed attorney in New York City. Joan took a trip to San Diego, CA, primarily for business, to consult with a client and take a short vacation. On the trip, Joan incurred the...
-
Deborah purchases a new $30,000 car in 2012 to use exclusively in her business. If Deborah does not elect to expense or take bonus depreciation in 2012 and holds the car until it is fully...
-
Jan has two jobs during 2012. One employer withheld and paid FICA taxes on $66,600 of Jan's salary, and the other employer withheld and paid FICA taxes on $44,400 in salary paid to Jan. Calculate the...
-
For an ideal solution, isotherm on an enthalpy-concentration diagram will be (a) Parabola (b) Hyperbola (c) Sine curve (d) Straight line
-
For a reversible process, change in entropy of the system (a) Approaches to zero (b) Increases (c) Decreases (d) Remains constant
-
For a multi-component system, the chemical potential is equivalent to (a) Molar free energy (b) Molar concentration difference (c) Molar free energy change (d) Partial molar free energy

Study smarter with the SolutionInn App



