Calculate the fraction of the molecules in a gas that are moving with translational kinetic energies...
Fantastic news! We've Found the answer you've been seeking!
Question:
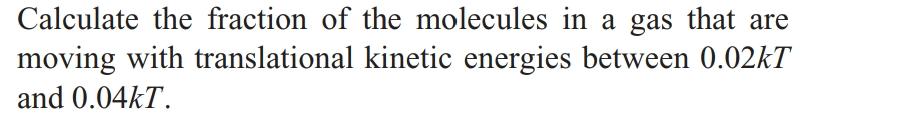
Transcribed Image Text:
Calculate the fraction of the molecules in a gas that are moving with translational kinetic energies between 0.02kT and 0.04kT. Calculate the fraction of the molecules in a gas that are moving with translational kinetic energies between 0.02kT and 0.04kT.
Expert Answer:
Related Book For 

Posted Date:
Students also viewed these physics questions
-
In one mole of a gas at a temperature of 650 K (kT = 8.97 10-21 J = 0.0560 eV), calculate the number of molecules with energies between 0.0105 eV and 0.0135 eV. (b) In this gas, calculate the...
-
Calculate the fractions of molecules in a gas that have a speed in a range t. v at the speed ne relative to those in the same range at c itself? This calculation can be used to estimate the fraction...
-
The kinetic theory of gases states that the kinetic energy of a gas is directly proportional to the temperature of the gas. A relationship between the microscopic properties of the gas molecules and...
-
The following condensed balance sheet is for the partnership of Miller, Tyson, and Watson, who share profits and losses in the ratio of 6:2:2, respectively: For how much money must the other assets...
-
A person stands hands at his side, on platform that is rotating at a rate of 1.30 rev/s. If he raises his arms to a horizontal position Fig. 8-48, the speed of rotation decreases to 0.80 rev/s. (a)...
-
Write an application that allows a user to enter the names and birthdates of up to 10 friends. Continue to prompt the user for names and birthdates until the user enters the sentinel value ZZZ for a...
-
The United States is the largest producer of corn in the world. Each year, a corn farmer must decide what type of seed to use and what the end market for the corn crop will be. In approximately a...
-
The following facts pertain to a noncancelable lease agreement between Earth Leasing Corporation and New Moon Company, a lessee. Inception date October 1, 2014 Annual lease payment due at the...
-
A single die is rolled one time. Find the probability of rolling a number at most 4 or a prime number. O 4/6 2/6 3/6 0.5/6
-
1. Consider the problem of returning change to a customer requiring the smallest number of coins. a. Show that the greedy algorithm given below returns the smallest number of coins for the coin set...
-
10. Investigators at Hunter College, perform a study to investigate the effect of Stress on Coronary Heart Disease (CHD) in Black vs. White staff members over a 10 year period. Stress High Low Total...
-
Based upon your in-depth knowledge of Lululemon, you are to identify and describe your firm's most persuasive growth opportunity: Your growth opportunity may fall within 1 of the 3 common situations...
-
Parra Building and Construction Ltd ( Parra Ltd ) was born as a small public company by an enterprising individual. Parra Ltd thrived during a period of economic expansion. It has millions in issued...
-
XYZ's last year's annual dividend was 3.50 a share, and you estimate that dividends will grow at 5.5% for the long term. The current stock price is $75 a share. What is your expected return for the...
-
EBITDA as reported on the company's most recent financial statements: $2,100,000 Sustaining capital reinvestment: $450,000 PV of CCA tax shield on sustaining capital investment: $75,000 Marketable...
-
Create a weather-tracking application. Define a list to store daily temperature data for a week. Each element should be a dictionary representing a day with keys such as date, high temperature, low...
-
SUBMISSION PLATFORM: SMART3UMS Manis Bhd has made an offer of one of its shares for every two of Masin Bhd. Synergistic benefits from the merger would result in increased after-tax earnings of RM4m...
-
Annual dividends of ATTA Corp grew from $0.96 in 2005 to $1.76 in 2017. What was the annual growth rate?
-
When an electron occupies a 2s orbital on an N atom it has a hyperfine interaction of 55.2 mT with the nucleus. The spectrum ofN02 shows an isotropic hyperfine interaction of 5.7 mT. For what...
-
Planets lose their atmospheres over time unless they are replenished. A complete analysis of the overall process is very complicated and depends upon the radius of the planet, temperature,...
-
What are the most likely locations of a particle in a box of length L in the state 11= 5?
-
Identify organizations in your area that fit each business market category: producer, reseller, government, and institutional. Explain your classifications.
-
Barajas Printing Corp. uses a job order cost system. The following data summa- Prepare entries for manufacrize the operations related to the first quarter's production. turing costs. (SO 2, 3, 4. 5)...
-
Purchases by businesses may be described as new-task, modified rebuy, or straight rebuy. Categorize the following purchase decisions and explain your choices. a. Bob has purchased toothpicks from...

Study smarter with the SolutionInn App



