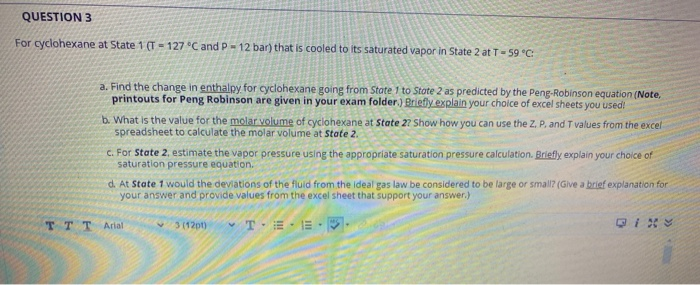
QUESTION 3 For cyclohexane at State 1 (T-127 C and P-12 bar) that is cooled to...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
QUESTION 3 For cyclohexane at State 1 (T-127 °C and P-12 bar) that is cooled to its saturated vapor in State 2 at T-59 °C: a. Find the change in enthalpy for cyclohexane going from State 1 to State 2 as predicted by the Peng-Robinson equation (Note. printouts for Peng Robinson are given in your exam folder.) Briefly explain your choice of excel sheets you used! b. What is the value for the molar volume of cyclohexane at State 27 Show how you can use the Z. P. and T values from the excel spreadsheet to calculate the molar volume at State 2. c. For State 2, estimate the vapor pressure using the appropriate saturation pressure calculation. Briefly explain your choice of saturation pressure equation. d. At State 1 would the deviations of the fluid from the ideal gas law be considered to be large or small? (Give a brief explanation for your answer and provide values from the excel sheet that support your answer.) TE E P TTT Arial 3 (12pt) QUESTION 3 For cyclohexane at State 1 (T-127 °C and P-12 bar) that is cooled to its saturated vapor in State 2 at T-59 °C: a. Find the change in enthalpy for cyclohexane going from State 1 to State 2 as predicted by the Peng-Robinson equation (Note. printouts for Peng Robinson are given in your exam folder.) Briefly explain your choice of excel sheets you used! b. What is the value for the molar volume of cyclohexane at State 27 Show how you can use the Z. P. and T values from the excel spreadsheet to calculate the molar volume at State 2. c. For State 2, estimate the vapor pressure using the appropriate saturation pressure calculation. Briefly explain your choice of saturation pressure equation. d. At State 1 would the deviations of the fluid from the ideal gas law be considered to be large or small? (Give a brief explanation for your answer and provide values from the excel sheet that support your answer.) TE E P TTT Arial 3 (12pt)
Expert Answer:
Answer rating: 100% (QA)
Do 08m di 0816 m niculate for other size M33X2 It area ... View the full answer

Related Book For 

Fundamentals of Thermodynamics
ISBN: 978-0471152323
6th edition
Authors: Richard E. Sonntag, Claus Borgnakke, Gordon J. Van Wylen
Posted Date:
Students also viewed these chemistry questions
-
You have exams in economics and chemistry coming up, and you have 5 hours available for studying. The following table shows the trade-offs you face in allocating the time you will spend studying each...
-
What stereo chemistry do you expect for the tri-substituted alkene obtained by E2 elimination of the following alkyl halide on treatment with KOH? (Reddish brown =Br.)
-
Name the following amine, including R, S stereo chemistry and draw the product of its reaction with excess iodomethane followed by heating with Ag2O (Hofmann elimination). Is the stereochemistry of...
-
Which is the most effective means of motivating trainees for a training program? a. Pay extra for training time. b. Require the training only for newly hired workers. c. Explain why the training is...
-
The use of control theory to provide insight into neurophysiology has a long history. As early as the beginning of the last century, many investigators described a muscle control phenomenon caused by...
-
Evaluate the line integral using the Fundamental Theorem of Line Integrals. Use a computer algebra system to verify your results. Repeat Exercise 31 using the integral Data from in Exercise 31...
-
The structure shown in Fig. P9.71 consists of three cylindrical support posts to which an elliptical flat plate sign is attached. Estimate the drag on the structure when a \(50-\mathrm{mph}\) wind...
-
Derby, Inc. manufactures golf clubs in three models. For the year, the Eagle line has a net loss of $20,000 from sales $200,000, variable expenses $180,000, and fixed expenses $40,000. If the Eagle...
-
Explain the principles and applications of root cause analysis (RCA) in quality control, examining methods such as the "5 Whys," fishbone diagrams, and fault tree analysis for identifying underlying...
-
1. Since Mr. Snodgrass doesnt have any historical data, (only an estimate of the number of customers hell serve each month), which specific type of qualitative method is he using? What would be the...
-
The Green County school system has 2,997 high school students, 3,831 middle school students, and 5,084 elementary school students. How many total students are in the Green County school system?
-
Discuss the main features and importance of the key financial statements.Include the information each statement contains.
-
If you were designing and implementing a reverse logistics process for your firm, what are the key factors you would consider for implementation, and why?
-
Social media and mobile marketing are big components of most companies promotional campaigns. Discuss how these methods of marketing affect you and your buying behavior. Do these marketing techniques...
-
How can new technologies improve trade facilitation?
-
Congratulations! You have received a large bonus and you want to invest the bonus in the stock market. Which type of investment would you most likely invest in if you are looking for capital...
-
Enter transactions to the appropriate journals ( Sales , ?Purchases, Cash Receipts, Cash Disbursements or General Journal ) . ?An example is shown for each special journal - ?transactions b , ?c , ?...
-
A Alkynes can be made by dehydrohalogenation of vinylic halides in a reaction that is essentially an E2 process. In studying the stereochemistry of this elimination, it was found that...
-
A cannon-ball of 5 kg acts as a piston in a cylinder of 0.15 m diameter. As the gun-powder is burned a pressure of 7 MPa is created in the gas behind the ball. What is the acceleration of the ball if...
-
I cool moist air, do I reach the dew first in a constant P or constant V process?
-
At the beach, atmospheric pressure is 1025 mbar. You dive 15 m down in the ocean and you later climb a hill up to 250 m elevation. Assume the density of water is about 1000 kg/m3 and the density of...
-
How are the identifiable assets and liabilities acquired in a business combination measured?
-
What is meant by deferred consideration and how is it accounted for?
-
Distinguish between a business combination and a non-business acquisition.

Study smarter with the SolutionInn App


