What stereo chemistry do you expect for the tri-substituted alkene obtained by E2 elimination of the following
Question:
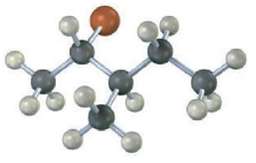
What stereo chemistry do you expect for the tri-substituted alkene obtained by E2 elimination of the following alkyl halide on treatment with KOH? (Reddish brown =Br.)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 81% (11 reviews)
Strategy As in the previous problem draw the structure convert it to a Newma...View the full answer

Answered By

Turvin Amwoma
I have a lot of educational and tutoring experience in various fields including but not limited to biological Sciences, Chemistry, Computer Science and Mathematics. I enjoy helping students and I have been tutoring for more than two years now. I always give students my best because nothing less is worth it.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
What stereo chemistry do you expect for the alkene obtained by E2 elimination of (1R, 2R)-1, 2-dibromo-1, 2-diphenylethane? Draw a Newman projection of the reacting conformation?
-
What hybridization do you expect for the atom indicated in red in each of the following species? (a) CH3CO2- (b) PH4+ (c) AlF3 (d) H2C==CH-CH2+
-
Treatment of the alkyl halide in Problem 6.9 with KOH in methanol gives mainly a mixture of the alkenes whose structures you drew. But treatment with only methanol gives a different product. What is...
-
What are the roles and responsibilities of civil society in relation to the Sustainable Development Goals? In your country, how has civil society been engaged in the dialogue or discussion on...
-
How does it rate in terms of being acceptable and delivery specific feedback?
-
Identify the functional groups in each of the following compounds: (a) H3 C--- CH2--- OH (b) (c) (d) (e) CH3CH2CH2CH2CHO H3C-N CH2CH CH2 CH3C CCH2COOH
-
Judging the Adequacy of Control Procedures Dunbar Camera, Inc., manufactures high-priced motion picture cameras for the movie industry. The component parts specifications are vital to the filmmaking...
-
Deluxe Ezra Company purchases equipment on January 1, Year 1, at a cost of $469,000. The asset is expected to have a service life of 12 years and a salvage value of $40,000. Instructions (a) Compute...
-
A pendulum inside a moving car is seen to make an angle of 30 o with the vertical for a duration of 20s. Calculate the change in velocity of the car in this duration?
-
Shirley had a three-sevenths interest in a partnership. She sold three-fifths of her interest for $27,000. a. What is the implied value of Shirleys remaining partnership interest? b. What is the...
-
What alkyl halides might the following alkenes have been madefrom? (b) C CH (a) (a) CH CHH2H2%3DCH2 "CH
-
Which isomer would you expect to undergo E2 elimination faster, trans1-bromo-4-tert-butylcyclohexane or cis-1-bromo-4-tert-butylcyclohexane? Draw each molecule in its more stable chair conformation,...
-
As strip steel leaves the last set of rotters in a hot rolling mill. it is quenched by planar water jets before being coiled. Due to the large plate temperatures, film boiling is achieved shortly...
-
Imagine that you were a member of the jury that heard the Ancheff case. Would you have concluded that John Ancheff was in a research study? What pieces of information would have been most important...
-
Draft a memo to your boss Sydney setting forth the legal responsibilities (if any) towards this person. Discuss in detail, in relation to both federal and a specific state law, if the company has to...
-
Question 1 a) What is meant by the term 'the rule of law'? b) What does it mean to say that the law (or a law) is weak because of a 'weak social contract'? c) What is communicative action and how can...
-
There are two definitive paths to terrorism; the "top-down process" and the "bottom-up process" (CSIS, 2007, in your week three content folder). Unfortunately, due to certain laws, civil rights, and...
-
Whats the case brief with Carpenter v. United States and the Supreme Court's extension of Fourth Amendment protection to cell phones and locations? What is the dissenting opinions of Justices...
-
A tree and a pole are 3000 m apart. Each is suddenly hit by a bolt of lightning. Mark, who is standing at rest midway between the two, sees the two lightning bolts at the same instant of time. Nancy...
-
Several months have passed and the Managing Partner approved and properly filed the Complaint and properly submitted the Request for Production of Documents that you drafted. In fact, it has been 75...
-
Consider this reaction at equilibrium: Predict whether the reaction will shift left, shift right, or remain unchanged after each disturbance. a. COCl 2 is added to the reaction mixture. b. Cl 2 is...
-
Draw the structures of the following compounds. (a) Ethanoic acid (b) Terephthalic acid (c) Magnesium formate (d) Malonic acid (e) Dichloroacetic acid (f) Salicylic acid (g) Zinc undecanoate...
-
Show how you would use extractions with a separatory funnel to separate a mixture of the following compounds: benzoic acid, phenol, benzyl alcohol, aniline.
-
Arrange each group of compounds in order of increasing basicity. (a) CH3COO-, ClCH2COO-, PhO- (b) Sodium acetylide, sodium amide, and sodium acetate (c) Sodium benzoate, sodium ethoxide, and sodium...
-
Marea's Cakes sells its cakes for $30 each. Ingredients cost Marea on average $10 per cake and she pays her baker $10 per cake. For the upcoming year, Marea's fixed costs will be $50,000 and she...
-
The statement of cash flow consists of three main activities, as follows: Operating Investing Financing Each section provides the reader with details on where the cash came from and where the cash...
-
How would i calculate the desired ROI per unit based on this information: $65Per Unit Direct Materials $48.00Per Unit Direct Labour $16.00Per Unit Variable Manufacturing Overhead $12.00Per Unit...

Study smarter with the SolutionInn App


