(a) The absorption spectrum of the HCL molecule is shown in Figure 1. Explain the reason...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
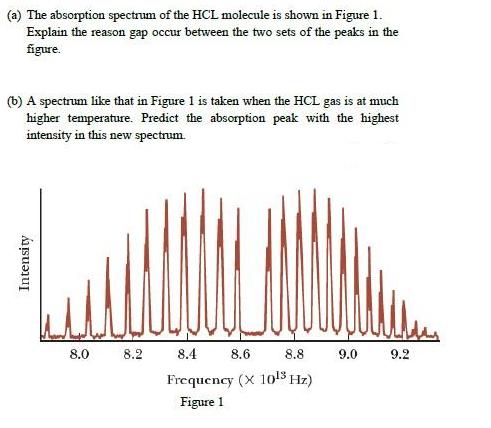
(a) The absorption spectrum of the HCL molecule is shown in Figure 1. Explain the reason gap occur between the two sets of the peaks in the figure. (b) A spectrum like that in Figure 1 is taken when the HCL gas is at much higher temperature. Predict the absorption peak with the highest intensity in this new spectrum. 8.0 8.2 8.4 8.6 8.8 9.0 9.2 Frequency (X 1o!5 Hz) Figure 1 Intensity (a) The absorption spectrum of the HCL molecule is shown in Figure 1. Explain the reason gap occur between the two sets of the peaks in the figure. (b) A spectrum like that in Figure 1 is taken when the HCL gas is at much higher temperature. Predict the absorption peak with the highest intensity in this new spectrum. 8.0 8.2 8.4 8.6 8.8 9.0 9.2 Frequency (X 1o!5 Hz) Figure 1 Intensity
Expert Answer:
Answer rating: 100% (QA)
1 A classic among molecular spectra the infrared absorption spectrum of HCl can be analyzed to gain information about both rotation and vibration of the molecule The absorption lines shown involve tra... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
A system with unity feedback is shown in Figure E5.11. Determine the steady-state error for a step and a ramp input when Figure E5.11 Unity feedback system. 5(s + 8) s(s 1)(s 4)(s 10) G(s) Gs) Yts)
-
A spacecraft with a camera is shown in Figure AP6.6(a).The camera slews about 16° in a canted plane relative to the base. Reaction jets stabilize the base against the reaction torques from the...
-
A cooling curve is shown in Figure 9 - 21. Determine (a) The pouring temperature; (b) The solidification temperature; (c) The superheat; (d) The cooling rate, just before solidification begins; (e)...
-
HL Co. uses the high-low method to derive a total cost formula. Using a range of units produced from 1,500 to 7,500, and a range of total costs from $21,000 to $45,000, producing 2,000 units will...
-
Robinsons Plastic Pipe Corporation uses a data processing system for inventory. The input to this system is shown in Table. You are using an input controls matrix to help audit the source data...
-
An electric dipole consisting of charges of magnitude 1.50nC separated by 6.20 pm is in an electric field of strength 1100 N/C. What are? (a) The magnitude of the electric dipole moment and (b) The...
-
Payables are: a. Customers who pay cash b. Customers who owe money to the business c. Suppliers who are owed money by the business d. Suppliers who have been paid by the business
-
Taylor Orlando began Taylor Roofing, his roofing business, on March 1, 2022. On March 31, the balances in the accounts were as follows. Cash $9,000, Accounts Receivable $2,000, Equipment $8,000,...
-
K Evaluate the following integral. Sx***** dx 1 6 +9 dx= +0 3 log 6
-
A dog training business began on December 1. The following transactions occurred during its first month. December 1 Receives $23,000 cash as an owner investment in exchange for common stock. December...
-
Required: Prepare a statement of cash flows, In proper format, for the year ended December 31, 2023. Use the termplate provided in your response area. A- BI Tesla Co. Statement of Cash Flows For the...
-
If the first payment is $600, find the present worth of a series of 10 payments, which are expected to inflate at a rate of 6% per year, and the market discount rate is 9%.
-
PELLE, SA is a company dedicated to the manufacturing and sales of leather shoes and bags. The data corresponding to the costs of the last financial year are as follows. There were no stocks of work...
-
B. Analyze the ratio of fixed assets to long-term liabilities measure of solvency for Sparkit using the attached Figure A1.2 Excel file by doing the following: 1. Discuss why it is important for...
-
Question 3 (30 marks) (a)Define interest rate risk on bond prices. (2 marks) (b)Anson is studying the interest rate risk of two bonds that he is holding. Bond A is with longer term to maturity than...
-
1. Abond with face value of $1,000 and a 7.25% coupon matures in 5 years and is currently selling at 109. Your discount rate is 8%. Determine the value of the bond to you.Show answer to 2-decimal...
-
After preparing its draft final accounts for the year ended 31 August 21Y0 and its draft balance sheet as at 31 August 21Y0, VWX Ltd discovered an error in the stock lists used to compute stock value...
-
At the beginning of its fiscal year, Lakeside Inc. leased office space to LTT Corporation under a seven-year operating lease agreement. The contract calls for quarterly rent payments of $25,000 each....
-
An adaptive suspension vehicle uses a legged locomotion principle. The control of the leg can be represented by a unity feedback system with [12] We desire to achieve a steady-state error for a ramp...
-
A system has the transfer function (a) Construct a state variable representation of the system. (b) Determine the element Ï11(t) of the state transition matrix for this system. Y(s) R(s)
-
One of the most potentially beneficial applications of an automotive control system is the active control of the suspension system. One feedback control system uses a shock absorber consisting of a...
-
Which of the following is NOT a common type of white box test of controls? a. completeness tests b. redundancy tests c. inference tests d. authenticity tests
-
In an automated payroll processing environment, a department manager substituted the time card for a terminated employee with a time card for a fictitious employee. The fictitious employee had the...
-
Which of the following is NOT a test for identifying application errors? a. reconciling the source code b. reviewing test results c. retesting the program d. testing the authority table

Study smarter with the SolutionInn App


