A solution contains 5.0 g of a non-electrolyte solute with a molar mass of 121 g/mol...
Fantastic news! We've Found the answer you've been seeking!
Question:

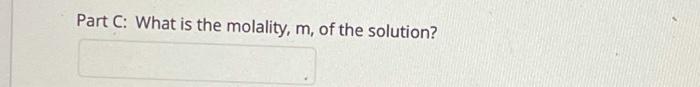


Transcribed Image Text:
A solution contains 5.0 g of a non-electrolyte solute with a molar mass of 121 g/mol dissolved in 95.4 g of chloroform. The solution has a freezing point of -65.8°C. The freezing point of pure chloroform is -63.5°C. What is the freezing point constant, kf, for this solvent? Part A: How many moles of solute are present in the solution? Part B: How many kg of chloroform are present in the solution? Part C: What is the molality, m, of the solution? Part D: What is the change in freezing point, ATf, for the solvent? Part E: What is the freezing point constant, kf, for this solvent? A solution contains 5.0 g of a non-electrolyte solute with a molar mass of 121 g/mol dissolved in 95.4 g of chloroform. The solution has a freezing point of -65.8°C. The freezing point of pure chloroform is -63.5°C. What is the freezing point constant, kf, for this solvent? Part A: How many moles of solute are present in the solution? Part B: How many kg of chloroform are present in the solution? Part C: What is the molality, m, of the solution? Part D: What is the change in freezing point, ATf, for the solvent? Part E: What is the freezing point constant, kf, for this solvent?
Expert Answer:
Answer rating: 100% (QA)
PART A Moles of solute Mass of soluteMolar mass of solute So moles of solute 5 ... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
How many moles of solute are present in 225 mL of a 1.44 M CaCl2 solution?
-
How many moles of MgCl2 are present in 0.0331 L of a 2.55 M solution?
-
How many moles of NH4Br are present in 88.9 mL of a 0.228 M solution?
-
Education is a very important job because it can change and shape people's lives. It gives people the knowledge, skills, and attitudes they need to be successful in their personal and work lives. As...
-
What are the benefits and drawbacks of using a qualitative risk impact matrix for classifying the types of project risk?
-
When companies offer new debt security issues, they publicize the offerings in the financial press and on Internet sites. Assume the following were among the debt offerings reported in December 2018:...
-
The peak current through a capacitor is \(2.0 \mathrm{~A}\). What is the peak current if a. The peak emf \(\mathcal{E}_{0}\) is doubled? b. The capacitance \(C\) is doubled? c. The frequency \(f\) is...
-
A process tank has two input streamsStream 1 at mass flow rate w1 and Stream 2 at mass flow rate w2. The tanks effluent stream, at flow rate w, discharges through a fixed valve to atmospheric...
-
Create two sets such that A x B = B x A . What are the elements of the A x B in this case?
-
Find the transfer function, G(s) = V o (s)/V i (s), for each operational amplifier circuit shown in Figure P2.7. 100 kQ 2 F 500 k2 2 F 100 k2 100 k2 2 F (1)'a 100 k2 2 uF (b)
-
Design a sequence generator that generates the following serial sequence: If your Entry no. is 2021EE1X1X2X3X4, then the serial sequence to be generated is: {3-bit binary of (X3%8), 3-bit binary of...
-
In what direction is the international law system heading? How does international law compare with domestic law?
-
What are the reasons why States obey International Law ?, and why some States do not obey such International laws?
-
On September 1, 2002, Biff receives a coupon payment of $750. The market interest rate on bonds like his remains 6 percent. Biff sells his bond to Cass at that time, for a price equal to the present...
-
what is origin of International law? discuss the role, importance and limitations of international law of Indo-pak territorial disputes
-
How does the Texas Restaurant Association method differ from the overhead contribution method?
-
1. A real object, 10cm tall, is located 50cm to the left of a-4.00D lens. Determine if the image isreal/virtual, upright/inverted, height of the image and the imagedistance, relative to the lens.c 1...
-
Bobbie Singh provides writing services for small businesses. He blogs for companies that need professionally written content. His business records at November 15, 2023, are shown below: During the...
-
Consider the following potential-energy curves for two different reactions: a. Which reaction has a higher activation energy for the forward reaction? b. If both reactions were run at the same...
-
What variables determine the height of the liquid in a manometer?
-
Define the units curie, rad, and rem.
-
Climate change (a) is a subject most scientists disagree about. (b) refers to the effect of greenhouse gases on atmospheric temperature. (c) refers to temperature increases in all of Earths climate...
-
Why do earthquakes produce seismic waves?
-
In an ice age, glaciers (a) advance. (b) retreat. (c) cause sea level to rise. (d) reflect incoming solar radiation.

Study smarter with the SolutionInn App



