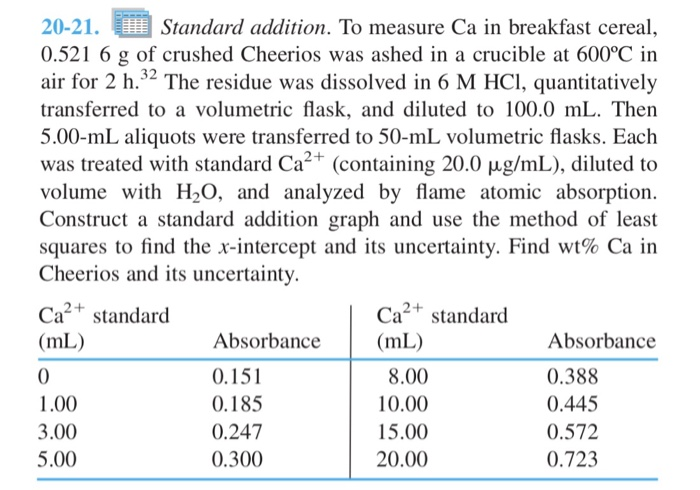
20-21. Standard addition. To measure Ca in breakfast cereal, 0.521 6 g of crushed Cheerios was...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
20-21. Standard addition. To measure Ca in breakfast cereal, 0.521 6 g of crushed Cheerios was ashed in a crucible at 600°C in air for 2 h.32 The residue was dissolved in 6 M HCl, quantitatively transferred to a volumetric flask, and diluted to 100.0 mL. Then 5.00-mL aliquots were transferred to 50-mL volumetric flasks. Each was treated with standard Ca²+ (containing 20.0 µg/mL), diluted to volume with H₂O, and analyzed by flame atomic absorption. Construct a standard addition graph and use the method of least squares to find the x-intercept and its uncertainty. Find wt% Ca in Cheerios and its uncertainty. Ca²+ standard (mL) 0 1.00 3.00 5.00 Absorbance 0.151 0.185 0.247 0.300 Ca²+ standard (mL) 8.00 10.00 15.00 20.00 Absorbance 0.388 0.445 0.572 0.723 20-21. Standard addition. To measure Ca in breakfast cereal, 0.521 6 g of crushed Cheerios was ashed in a crucible at 600°C in air for 2 h.32 The residue was dissolved in 6 M HCl, quantitatively transferred to a volumetric flask, and diluted to 100.0 mL. Then 5.00-mL aliquots were transferred to 50-mL volumetric flasks. Each was treated with standard Ca²+ (containing 20.0 µg/mL), diluted to volume with H₂O, and analyzed by flame atomic absorption. Construct a standard addition graph and use the method of least squares to find the x-intercept and its uncertainty. Find wt% Ca in Cheerios and its uncertainty. Ca²+ standard (mL) 0 1.00 3.00 5.00 Absorbance 0.151 0.185 0.247 0.300 Ca²+ standard (mL) 8.00 10.00 15.00 20.00 Absorbance 0.388 0.445 0.572 0.723
Expert Answer:
Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
To measure Ca in breakfast cereal, 0.521 6 g of crushed Cheerios was ashed in a crucible at 600ºC in air for 2 h.32 The residue was dissolved in 6 M HCl, quantitatively transferred to a...
-
What is the shear capacity of the RC beam described below considering the steel reinforcement and using the formula: VRsyAw 2fyd cot 8/s The shear reinforcement in the beam is provided by sets of...
-
A 50 m3 insulated chamber containing air at 40oC, 100 kPa and R.H. 20% is connected to another 50 m3 insulated chamber containing air at 20oC, 100 kPa and R.H. 100%. The valve is opened and the...
-
Data on the length, in seconds, of a sample of 50 songs by The Beatles are presented in the accompanying data table. Complete parts (a) through (d) below. Click the icon to view the table of song...
-
Write an equation for each of the following. a. Burning of lithium metal in oxygen b. Burning of methylamine, CH3NH2, in excess oxygen (N ends up as N2) c. Burning of diethyl sulfide, (C2H5)2S, in...
-
Wesley Corp. stock is trading for $27/share. Wesley has 20 million shares outstanding and a market debt-equity ratio of 0.49. Wesleys debt is zero-coupon debt with a 5-year maturity and a yield to...
-
If the spare part of a cement mixer has the Weibull failure-time distribution with the parameters \(\alpha=\) 0.001 per hour and \(\beta=0.65\), find the probability that it will not operate...
-
Silver Corporation has 2,000 shares of common stock outstanding. Howard owns 600 shares, Howards grandfather owns 300 shares, Howards mother owns 300 shares, and Howards son owns 100 shares. In...
-
Explain how high-pressure distillation systems influence the volatility of compounds and discuss the engineering challenges associated with operating at elevated pressures .
-
Sullivan Ranch Corporation has purchased a new tractor. The following information is given: $150,000 $10,000 Cost: Estimated Residual: Estimated Life in years: Estimated Life in hours: Actual Hours:...
-
Calculate the solubility (in g dm) of lead bromide in: (a) distilled water; (b) 0.08 mol dm KBr. Comment on the difference in results. Note: KSP (PbBr2)= 2.1 x 106 mol dm9 RAM's Pb: 207.212 Br: 79.904
-
Until a year ago, your business regularly used a local stationery and printing service. They were not that reliable, but you wanted to support a local business. One day, they suddenly informed you...
-
Are promotional slogans such as Best sushi in town or Atlantas most popular restaurant ethical? Why or why not?
-
An ethical __________ exists when a person is faced with conflicting but ethical choices or alternatives that are neither entirely right nor entirely wrong; an ethical __________ occurs when a person...
-
Shari Willison worked as a geologist in your civil engineering firm for 20 years before succumbing to leukemia last week. With only a few dozen employees, the company has always been a tight-knit...
-
Working as an associate lecturer for a well-known and successful online university, you use your own home as your office. Periodically, you teach residential courses and short, locally based...
-
As the globalization of business continues, it becomes more and more important for financial accounting standards to be uniform across countries. The FASB and the International Accounting Standards...
-
(a) What do data breach notification laws require? (b) Why has this caused companies to think more about security?
-
Solutions with a wide range of Hg 2+ concentrations were prepared to calibrate an experimental Hg 2+ ion-selective electrode. For the range 10 -5 < [Hg 2+ ] [ < 10 -1 M, Hg(NO 3 ) 2 was used...
-
The pH of microscopic vesicles (compartments) in living cells can be estimated by infusing an indicator (HIn) into the compartment and measuring the quotient [In-]/[HIn] from the spectrum of the...
-
A 50.00-mL solution containing NaBr was treated with excess AgNO3 to precipitate 0.214 6 g of AgBr (FM 187.772). What was the molarity of NaBr in the solution?
-
Describe which characteristics of HR metrics and workforce analytics are likely to result in greater return on investment and organizational impact.
-
Why are information security and privacy important considerations in the design, development, and maintenance of an HRIS?
-
List and discuss the major information security and privacy threats to organizations.

Study smarter with the SolutionInn App



