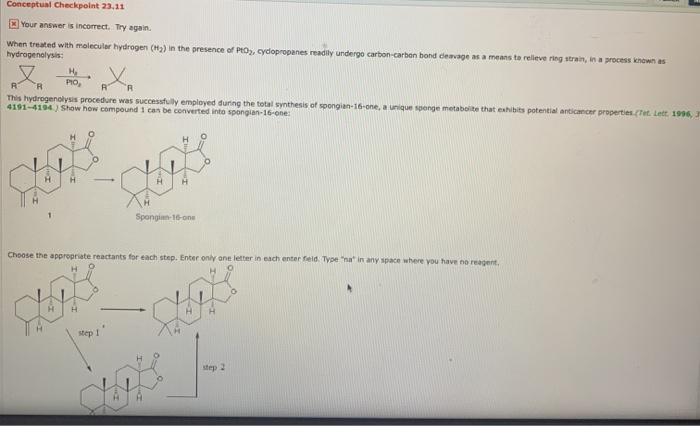
Conceptual Checkpoint 23.11 O Your answer is incorect. Try again. When treated with molecular hydrogen (H3)...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
Conceptual Checkpoint 23.11 O Your answer is incorect. Try again. When treated with molecular hydrogen (H3) in the presence of PtO, cydopropanes readily undergo carbon-carbon bond deavage as a means ta releve ring strain, in a process known as hydrogenolysis: H PIO, This hydrogenolysis procedure was successfully employed during the total synthesis of spongian-16-one, a unique spenge metaboite that eshibits potential anticancer properties (Tet. Lett. 1996, 4191-4194) Show how compound 1 can be converted into spongian-16-one: H Sponginn-16-one Choose the appropriate reactants for each step. Enter only ane letter in each enter feld. Type "na" in any space where you have no reagent. step 1 step 2 Conceptual Checkpoint 23.11 O Your answer is incorect. Try again. When treated with molecular hydrogen (H3) in the presence of PtO, cydopropanes readily undergo carbon-carbon bond deavage as a means ta releve ring strain, in a process known as hydrogenolysis: H PIO, This hydrogenolysis procedure was successfully employed during the total synthesis of spongian-16-one, a unique spenge metaboite that eshibits potential anticancer properties (Tet. Lett. 1996, 4191-4194) Show how compound 1 can be converted into spongian-16-one: H Sponginn-16-one Choose the appropriate reactants for each step. Enter only ane letter in each enter feld. Type "na" in any space where you have no reagent. step 1 step 2
Expert Answer:

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
Show how benzoyl chloride can be converted into each of the following compounds. Benzaldehyde
-
When the following compound is hydrated in the presence of acid, the unreacted alkene is found to have retained the deuterium atoms: What does the preceding statement tell you about the mechanism of...
-
Show how 1-butanol can be converted into the following compounds: a. CH3CH2CH2CH2Br b. c. CH3CH2CH2CH2OCH3 d. CH3CH2CH2CH2NHCH2CH3 e. CH3CH2CH2CH2SH f. CH3CH2CH2CH2C==N CH CH2CHCH2OCCH2CH,
-
What have researchers found about the use of job costing to record the cost of handproducing a bound book?
-
Name the specific form the following aldose shown here (b) HOCH OH OH OH
-
Multiple-Choice Questions 1. Under the UCC Article 9, which of the following actions will best perfect a security interest in a negotiable instrument against any other party? (a) Filing a security...
-
Gavin wants to dig deep into pricing theory, so he decides to work out an application of Eq. (16.11). He suggests to himself that a simple model of interest rates in the risk-neutral world might be...
-
On January 1, 2012, Ginsberg, Corp., issued $400,000 of 7.375%, five-year bonds payable when the market interest rate was 8%. Ginsberg pays interest annually at year-end. The issue price of the bonds...
-
Sri Lanka debt crisis was due to the economic mismanagement. Sri Lanka's current debt to GDP had skyrocketed from 42% in 2019 to 104% in 2021.According to Bloomberg, Sri Lanka has about $8.6 billion...
-
Cortez Manufacturing intends to increase capacity by overcoming a bottleneck operation by adding new equipment. Two vendors have presented proposals. The fixed costs are $50,000 for proposal A and...
-
The northern and southern blot procedures are nearly the same. Which step can you discard for RNA? detection of probe annealing of probe denaturing
-
On 1/1/2016, Wilson buys 25% of Taylormade for $500,000 and accounts for the investment using the equity method. The carrying value of net assets at date of purchase totaled $1,800,000. Fair value...
-
Imagine you are the owner of a portfolio of electricity generation assets in California with a total average annual production of 485,000 MWh. The State legislature has been considering a bill...
-
Answers provided. Please explain how it is solved. The following information applies to Questions 21-25. On January 1, 2020, Adamson, Inc. acquired the outstanding voting common stock of Skyview...
-
Renegade Corporation has 362,000 shares of $20 par common stock outstanding. On June 19, Renegade Corporation declared a 9% stock dividend to be issued on July 31 to stockholders of record on July...
-
The adjusted trial balance of Michael Co. for the year ending December 31, 2025, contains the following Michael Co. Adjusted Trial Balance December 31, 2025 Cash Accounts Receivable Prepaid Rent...
-
A cultural event or advocacy even for people with disabilities, a 2-page response on people with disabilities and why this event is essential, and what was learned from the event and how it...
-
Complete problem P10-21 using ASPE. Data from P10-21 Original cost ................................................................. $7,000,000 Accumulated depreciation...
-
List each set of compounds in order of increasing boiling point. (a) Hexane, octane, and decane (b) Octane, (CH3)3 C-C(CH3)3 and CH3CH2C(CH3)2CH2CH2CH3
-
Explain why the addition of a small amount of glycerol to the polymerization mixture gives a stiffer urethane foam.
-
Make a model and draw a three-dimensional structure for each compound. Then draw the mirror image of your original structure and determine whether the mirror image is the same compound. Label each...
-
In the ground state of a Fermi system, the chemical potential is identical to the Fermi energy: \((\mu)_{T=0}=\varepsilon\left(p_{F} ight)\). Making use of the energy spectrum \(\varepsilon(p)\) of...
-
Solve the Gross-Pitaevskii equation (11.2.23) in a harmonic trap for the case when the scattering length \(a\) is zero. Show that this reproduces the properties of the ground state of the...
-
Solve the Gross-Pitaevskii equation and evaluate the mean field energy, see equations (11.2.21) and (11.2.23), for an isotropic harmonic oscillator trap with frequency \(\omega_{0}\) for the case \(N...

Study smarter with the SolutionInn App


