Consider a cylindrical crew module with a diameter of 5 meters, a length of 15 meters,...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
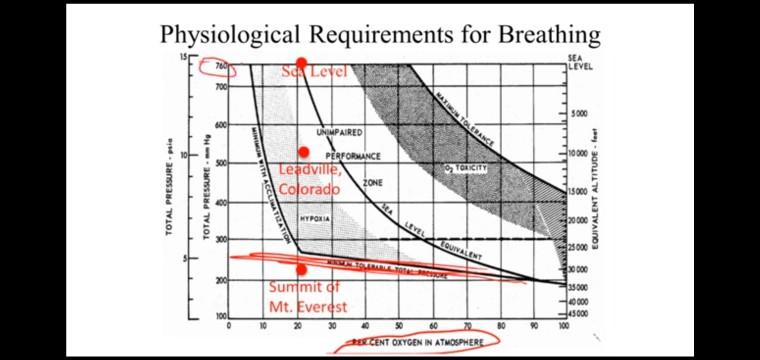
Consider a cylindrical crew module with a diameter of 5 meters, a length of 15 meters, and a crew of 6 people. The module contains air that is at standard sea-level atmospheric pressure and temperature. The sea level density of air at 20 degrees Celsius is 1,204 kg/m. Additional information necessary to solve this problem (eg., daily oxygen metabolic rate, cabin atmosphere composition) can be found in the videos in Subsection 3.2.2 and Subsection 3.3.2. What is the volume of the crew module? Provide your answer in m' with at least 2 decimal places. Assuming typical atmospheric composition in the crew module, what is the mass of oxygen in the cabin under normal operating conditions? Provide your answer in kg with at least 2 decimal places. Suppose the oxygen production system fails and the amount of oxygen in the cabin decreases due to crew respiration. The cabin pressurization system will continue to supply nitrogen to maintain an overall sea level pressure, and the CO2 scrubbing system will continue to keep CO2 at. acceptable levels. Hypoxia will begin to set in when the VOLUME percentage of oxygen in the atmosphere drops to about 15%. Assuming normal metabolic rates for each crew member, how long does the crew have before the oxygen level reaches the hypoxic threshold? Enter your answer in days with at least 2 decimal places. Submt You have used 0 of 3 attempts Save Physiological Requirements for Breathing SEA LEVEL S Leyel 700- S000 UNIMPAIRED 10000 PERFORMANCE Teadville ZONE Colorado TOICITY 15000 20000 HYPOKIA LEVEL LOUIVALENT 25 000 0 000 Summit of Mt. Everest 35000 He000 daso00 100L 60 80 100 20 30 CENT OXYGEN IN ATMOSPHERE TOTAL PRESSURE pie anssand TVIOL EQUIVALENT ALTITUDE - feet Consider a cylindrical crew module with a diameter of 5 meters, a length of 15 meters, and a crew of 6 people. The module contains air that is at standard sea-level atmospheric pressure and temperature. The sea level density of air at 20 degrees Celsius is 1,204 kg/m. Additional information necessary to solve this problem (eg., daily oxygen metabolic rate, cabin atmosphere composition) can be found in the videos in Subsection 3.2.2 and Subsection 3.3.2. What is the volume of the crew module? Provide your answer in m' with at least 2 decimal places. Assuming typical atmospheric composition in the crew module, what is the mass of oxygen in the cabin under normal operating conditions? Provide your answer in kg with at least 2 decimal places. Suppose the oxygen production system fails and the amount of oxygen in the cabin decreases due to crew respiration. The cabin pressurization system will continue to supply nitrogen to maintain an overall sea level pressure, and the CO2 scrubbing system will continue to keep CO2 at. acceptable levels. Hypoxia will begin to set in when the VOLUME percentage of oxygen in the atmosphere drops to about 15%. Assuming normal metabolic rates for each crew member, how long does the crew have before the oxygen level reaches the hypoxic threshold? Enter your answer in days with at least 2 decimal places. Submt You have used 0 of 3 attempts Save Physiological Requirements for Breathing SEA LEVEL S Leyel 700- S000 UNIMPAIRED 10000 PERFORMANCE Teadville ZONE Colorado TOICITY 15000 20000 HYPOKIA LEVEL LOUIVALENT 25 000 0 000 Summit of Mt. Everest 35000 He000 daso00 100L 60 80 100 20 30 CENT OXYGEN IN ATMOSPHERE TOTAL PRESSURE pie anssand TVIOL EQUIVALENT ALTITUDE - feet
Expert Answer:

Related Book For 

Posted Date:
Students also viewed these accounting questions
-
Air that is at its dew point of 5oC is drawn into a building where it is heated to 25oC. What will be the relative humidity at this temperature? Assume constant pressure of 1.0 atm. Take into account...
-
Air that is at its dew point of 5C is drawn into a building where it is heated to 22C.What will be the relative humidity at this temperature? Assume constant pressure of 1.0 atm. Take into account...
-
The atmospheric pressure at sea level is 14.7 pounds per square inch. Suppose that, at any elevation, an increase of 1 mile results in a decrease of 20% of the atmospheric pressure at that elevation....
-
Troy Engines, Ltd., manufactures a variety of engines for use in heavy equipment. The company has always produced all of the necessary parts for its engines, including all of the carburetors. An...
-
In Year 1, Utica Machinery Company uses the asset from RE11- 4 for 7,500 hours. Prepare the journal entry to record the depreciation.
-
The A-36 steel post is subjected to the forces shown. If the strain gages \(a\) and \(b\) at point \(A\) give readings of \(\epsilon_{a}=300\left(10^{-6} ight)\) and \(\epsilon_{b}=175\left(10^{-6}...
-
Timothy Muse opened a law office on October 2, 2010. During the first month of operations, the business completed the following transactions: Requirements 1. Open the following T-accounts: Cash,...
-
Marin County Enterprises has a monopoly on the production of lunar-powered homes and has the normal U-shaped average cost curve. At its present profit-maximizing output and price, it is able to earn...
-
a company cash balace on decembre 3 1 2 0 2 0 is 2 million $ and on decembre 3 1 , 2 0 1 9 it is 1 . 9 million $ . in addition a dividend of 3 0 0 0 0 0 $ was paid in 2 0 2 0 . then the total 2 0 2 0...
-
In about 300 words, explain how incubators and accelerators work. In your answer, compare these two approaches and provide at least one example of each.
-
Complete Marketing Plan Merge all the parts from Module 3, 4, 5, & 6 Assignments including the feedback from your instructor, plus any additional details to compile a Complete Marketing Plan.To...
-
Net Income $0 A Transactions that affect net income do not necessarily affect cash. Identify the effect, if any, that each of the following transactions would have upon cash and net income. The first...
-
Consider the following mass distribution where the x - and y -coordinates are given in meters: 5.2vkg at (0.0, 0.0) m, 3.4 kg at (0.0, 3.5) m, and 4.0 kg at (3.2, 0.0) m. Where should a fourth object...
-
In October 2025, MSI installed a new cloud-based inventory costing system to replace a system that had been developed in-house. The old system could no longer keep up with the complex and detailed...
-
For each of the following, say whether the series coverges or diverges. Show your work. i. +3n-4 2n-4n+6 iii. 5" (3/2) iv. n+ n-n
-
Chaz Corporation has taxable income in 2023 of $1,313,250 for purposes of computing the $179 expense and acquired the following assets during the year: Asset office furniture Computer equipment...
-
Explanation of how the EYLF guides early childhood education and care pedagogy. (word count is 200 in text citation and APA 7th reference compulsory)
-
The following selected accounts and normal balances existed at year-end. Notice that expenses exceed revenue in this period. Make the four journal entries required to close the books: Accounts...
-
In a given CRT, electrons are accelerated horizontally by 9.0 kV. They then pass through a uniform electric field E for a distance of 2.8 cm, which deflects them upward so they travel 22 cm to the...
-
Protons with momentum 4.8 10-21 kg m/s are magnetically steered clockwise in a circular path 2.2 m in diameter. Determine the magnitude and direction of the field in the magnets surrounding the...
-
A 28-g rifle bullet traveling 190 m/s embeds itself in a 3.1-kg pendulum hanging on a 2.8-m-long string, which makes the pendulum swing upward in an arc. Determine the vertical and horizontal...
-
A stirrer-container assembly contains a certain amount of fluid. The stirrer performs \(3 \mathrm{hp}\) work on the system. The heat developed by stirring is \(4000 \mathrm{~kJ} / \mathrm{h}\) and is...
-
Derive the mathematical expression of the first law of thermodynamics.
-
A system consisting of a gas confined in a cylinder undergoes a series of processes shown in Fig. 2.11. During the process A-1-B, \(70 \mathrm{~kJ}\) of heat is added while it does 45 \(\mathrm{kJ}\)...

Study smarter with the SolutionInn App


