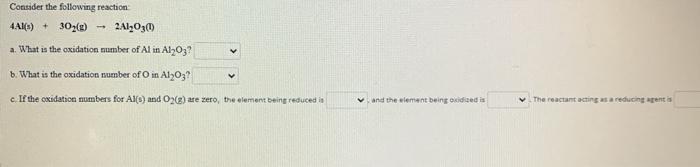
Consider the following reaction: 4.Al(s) + 302(g) 1 2AlO3(1) a. What is the oxidation number of...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
Consider the following reaction: 4.Al(s) + 302(g) 1 2Al₂O3(1) a. What is the oxidation number of Al in Al2O3? b. What is the oxidation number of O in Al₂O37 c. If the oxidation numbers for Al(s) and O2(g) are zero, the element being reduced is and the element being oxidized is The reactant acting as a reducing agent is Consider the following reaction: 4.Al(s) + 302(g) 1 2Al₂O3(1) a. What is the oxidation number of Al in Al2O3? b. What is the oxidation number of O in Al₂O37 c. If the oxidation numbers for Al(s) and O2(g) are zero, the element being reduced is and the element being oxidized is The reactant acting as a reducing agent is
Expert Answer:
Answer rating: 100% (QA)
Sol 4 Als 3 O g 2 AlO3 1 a Oxidation number of oxygen is generally t... View the full answer

Related Book For 

Chemistry The Central Science
ISBN: 978-0321696724
12th edition
Authors: Theodore Brown, Eugene LeMay, Bruce Bursten, Catherine Murphy, Patrick Woodward
Posted Date:
Students also viewed these chemical engineering questions
-
Consider the following reaction at some temperature: H2O(g) + CO(g) H2(g) + CO2(g) K = 2.0 Some molecules of H2O and CO are placed in a 1.0- L container as shown below. When equilibrium is reached,...
-
Consider the following reaction at 800. K: N2(g) + 3F2(g) 2NF3(g) An equilibrium mixture contains the following partial pressures: PN2 = 0.021 atm, PF2 = 0.063 atm, and PNF3 = 0.48 atm. Calculate Go...
-
Consider the following reaction at 298 K: 2SO2(g) + O2(g) 2SO3(g) An equilibrium mixture contains O2(g) and SO3(g) at partial pressures of 0.50 atm and 2.0 atm, respectively. Using data from...
-
The accountant for a subunit of Mountain Sports Company went on vacation before completing the subunits monthly performance report. This is as far as she got: Requirements 1. Complete the performance...
-
A company's customer service 800 telephone system is set up so that the caller has six options. Each of these six options leads to a menu with four options. For each of these four options, three more...
-
An ejector pump operates by injecting a high-speed fluid stream into a slower stream to increase its pressure. Consider water flowing at a rate of $50 \mathrm{gpm}$ through a $90^{\circ}$ elbow in a...
-
What is a first article system? Is there only one instance of a first article system?
-
Table illustrates the demand and supply schedules for television sets in Venezuela, a small nation that is unable to affect world prices. On graph paper, sketch Venezuelas demand and supply schedules...
-
Sustainability efforts can positively impact other business areas, such as customer satisfaction or increased marketing/brand awareness. How sustainability efforts have impacted other areas of the...
-
(Each part of this problem is quite long and best worked by groups of students.) Peak intensities of the molecular ion region are listed in parts (a) - (g) and shown in the figure. Identify which...
-
Assume the five-year lease in which ELC entered requires the same lease payments as the preceding operating lease. That is, Este Lauder agrees to pay $150,000 in year 1, $155,000 in year 2, $159,000...
-
What symmetry elements does this compound have: Tetrabromopalladinate (PdBr) (planar) As this compound is planar, what is the molecular geometry around the palladium atom? Therefore, what is the...
-
Consider an asset with value Sm at time-step mt and expected value Sm+1 under a continuous random walk model, 00 E[Sm+1|Sm] = ["S'p(Sm&t; S', (m + 1)&t)dS', 0 where p(S,t; S't') is the probability...
-
Year 3 calculate the additional amount of synergies (beyond the $100 million already included in the projections) needed to get to zero dilution or accretion if the number of share outstanding after...
-
A saturated clayey layer has been sandwiched between two sandy layers. The underground water level is 2 m below the surface. By the consideration of several factors, the foundation of the Aerospace...
-
We want to evaluate the performance of a managed portfolio (M) against a benchmark portfolio (B). Below is the required information. Asset Class Equity Bonds Cash Equity Sectors Capital Goods Energy...
-
3 At June 30, 2023, its year-end, Oberlo. held the following short-term investments: Short Term Investment - CAE Corp bonds $ 180,000 The fair value of the bonds were $ 173,000 Short Term Investment...
-
l ask this second time correnct answer is 38,01 can we look pls Consider a non-conducting rod of length 8.8 m having a uniform charge density 4.5 nC/m. Find the electric potential at P, a...
-
The following electron configurations represent excited states. Identify the element, and write its ground-state condensed electron configuration. (a) 1s2 2s2 3p2 4p1, (b) [Ar] 3d10 4s1 4p4 5s1, (c)...
-
Many gases are shipped in high-pressure containers. Consider a steel tank whose volume is 55.0 gallons that contains O2 gas at a pressure of 16,500 kPa at 23oC. (a) What mass of O2 does the tank...
-
KCl has the same structure as NaCl. The length of the unit cell is 6.28 . The density of KCl is 1.984 g/cm3, and its formula mass is 74.55 amu. Using this information, calculate Avogadro's number?
-
Determine whether each experiment is a binomial experiment. If it is, specify the values of n, p, and q, and list the possible values of the random variable x. If it is not, explain why. 1. A certain...
-
In a survey, U.S. adults were asked to identify which social media platforms they use. The results are shown in the figure. Six adults who participated in the survey are randomly selected and asked...
-
Rotator cuff surgery has a 90% chance of success. The surgery is performed on three patients. Find the probability of the surgery being successful on exactly two patients.

Study smarter with the SolutionInn App


