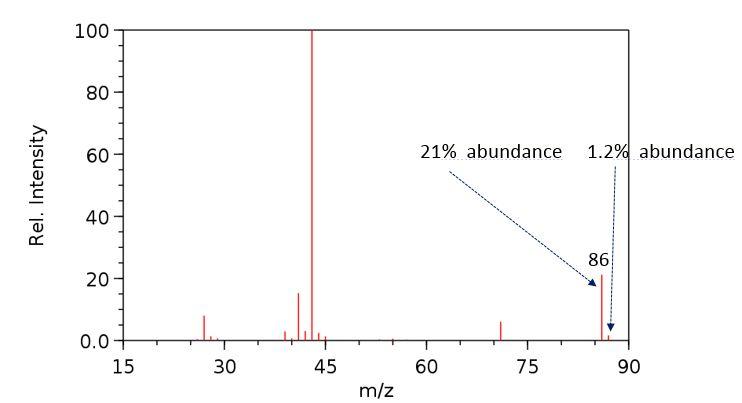
Consider the spectra below of an unknown compound. 100 80 60 21% abundance 1.2% abundance 40 86
Fantastic news! We've Found the answer you've been seeking!
Question:
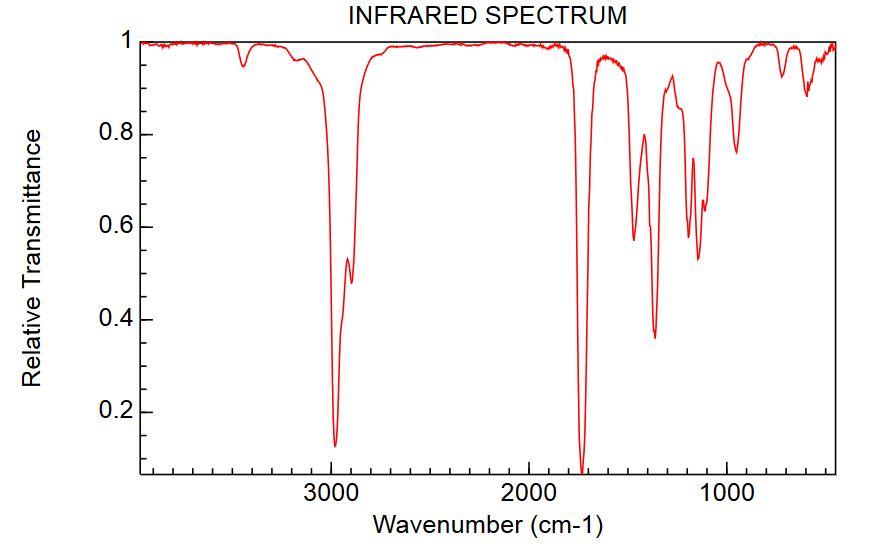
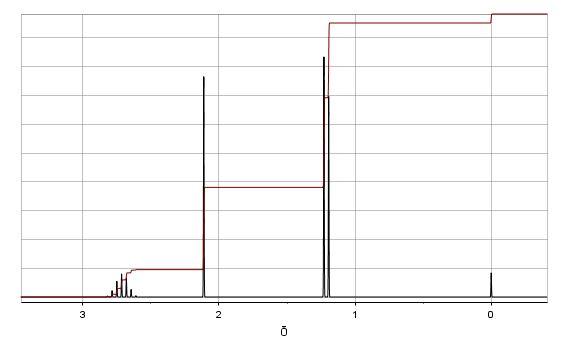
Consider the spectra below of an unknown compound.
Transcribed Image Text:
100 80 60 21% abundance 1.2% abundance 40 86 20 0.0- 15 30 45 60 75 90 m/z Rel. Intensity INFRARED SPECTRUM 0.8 0.6 0.4 0.2- 3000 2000 1000 Wavenumber (cm-1) Relative Transmittance 3 100 80 60 21% abundance 1.2% abundance 40 86 20 0.0- 15 30 45 60 75 90 m/z Rel. Intensity INFRARED SPECTRUM 0.8 0.6 0.4 0.2- 3000 2000 1000 Wavenumber (cm-1) Relative Transmittance 3
Expert Answer:
Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
The UV spectrum of an unknown compound shows values of λmax at 225 nm (ε = 10,000) and at 318 nm 1e = 402. The mass spectrum shows a molecular ion at m/z 96 and a prominent...
-
The mass spectrum and infrared spectrum of an unknown compound are shown in Figures 13.27 and 13.28, respectively. Identify the compound. Figure 13.27 The mass spectrum for Problem 28. 100 E 80 3 60...
-
The mass spectrum of an unknown compound has a molecular ion peak with a relative intensity of 43.27% and an M + 1 peak with a relative intensity of 3.81%. How many carbon atoms are in the compound?
-
Describe how organizations should apply the general principles of the Uniform Guidelines on Employee Selection Procedures to practical selection decisions.
-
When Chet died on March 12, 2013, he owned $900,000 in stock of ABC Corporation and $100,000 in City of Omaha bonds. The ABC Corporation declared a cash dividend on March 1 that was payable to...
-
HW Score: 58. Homework: HW 6 Annuity and Perpetuity < Question 2, P6-15 (simila... > O Points: 0 of (Present value of an annuity) What is the present value of a(n) 9-year annuity that pays $4,500...
-
Plot equilibrium data from Table 13-A1 for tri-ethylamine (solvent), carbon tetrachloride (solute), acetic acid (diluent) on a right triangle diagram with ordinate \(=\) mole fraction...
-
Hillyard Company, an office supplies specialty store, prepares its master budget on a quarterly basis. The following data have been assembled to assist in preparing the master budget for the first...
-
Hunter's property, with an adjusted basis of $250,000, is condemned by the state. Hunter receives property with a fair market value of $275,000 as compensation for the property taken. What is...
-
Jack Tasker opened his Auto Repair Shop in November 2023. The balance sheet at November 30, 2023, prepared by an inexperienced part-time bookkeeper, is shown below. Required Prepare a correct balance...
-
Suppose a company hopes to obtain P2 500 000 in an initial public offering. Based on a variety of factors, including the firm's expected earnings over the next few years, it's investment bankers...
-
What is the magnitude of vector labeled with " M " ? Provide your answer in cm , with 2 significant digits ( not in scientific notation ) . What is the magnitude of vector labeled with " N " ?...
-
What is the output of the following code: Hinclude int main () \{ int Tab [5] = $\{1,2,3,4,1\} ;$ int *ptr = Tab; printf("\\%d,%d,%d ", Tab[0],*(ptr+2), ptr[2]); return 0; \}
-
Compute the realized and recognized gain or loss for each of the following transactions and classify as short-term or long-term: a. Marge, a single taxpayer, receives $8,000 for the sale of stock she...
-
You have just been hired by FAB Corporation, the manufacturer of a revolutionary new garage door opening device. The presid asked you to review the company's costing system and "do what you can to...
-
Stefan is single and is living in Toronto. He is 66 years old and has worked with the same employer for over 35 years. Stefan is the store manager at a very successful bookstore, Magenta Inc....
-
Motorcade Company has three service departments (S1, S2, and S3) and two production departments (P1 and P2). The following data relate to Motorcade's allocation of service department costs: Round to...
-
Calculate Total Contribution Margin for the same items. Total Revenue Total Variable Costs Total Contribution Margin $50.00 a. $116.00 $329.70 b. $275.00 $14,796.00 $7,440.00 c. $40,931.25 d....
-
Which compound would form a lactone more rapidly? a. b. H. COOH H3C COOH OH OH or COOH COOH CH3 or
-
The rate law for the reaction of HO- with tert-butyl bromide to form an elimination product in 75% ethanol 25% water at 30 C is the sum of the rate laws for the E2 and E1 reactions: rate = 7.1 ...
-
The organoborane used in a Suzuki reaction is prepared by the reaction of catecholborane with an alkene or an alkyne. What hydrocarbon would you use to prepare the organoborane of Problem 35? Problem...
-
Stanford Corporation began the year with 150,000 shares of common stock outstanding. On March 1, an additional 10,000 shares of common stock were issued. On August 1, another 16,000 shares of common...
-
The following summarized data relate to Robert Corporation's current operations: Required Prepare a multiple-step income statement for Robert Corporation for the year. Include earnings per share...
-
Net sales, net income, and total asset figures for Janice Controls, Inc., for five consecutive years are given below (Janice manufactures pollution controls): Required a. Calculate trend percentages,...

Study smarter with the SolutionInn App



