Constants I Periodic Table When a photon passes through matter, there are three types of light-matter...
Fantastic news! We've Found the answer you've been seeking!
Question:
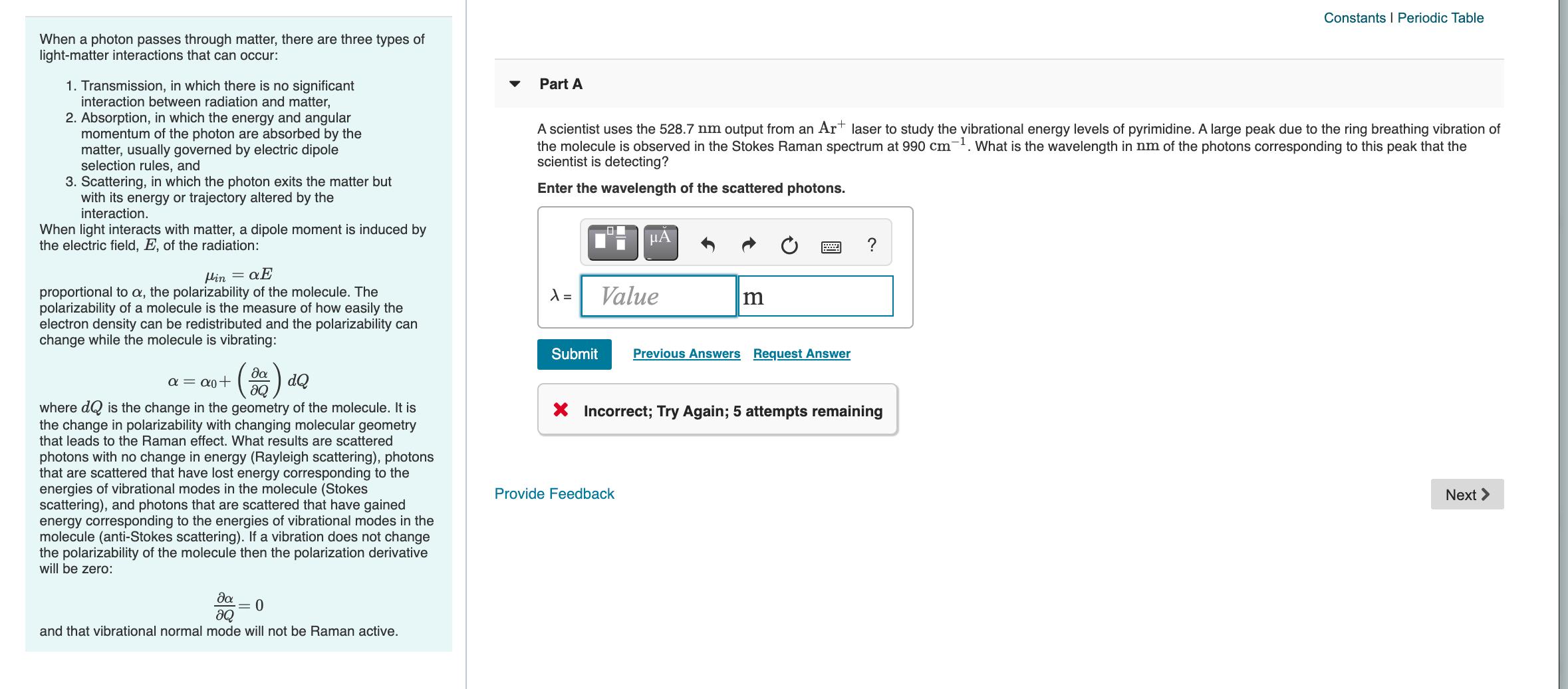
Transcribed Image Text:
Constants I Periodic Table When a photon passes through matter, there are three types of light-matter interactions that can occur: Part A 1. Transmission, in which there is no significant interaction between radiation and matter, 2. Absorption, in which the energy and angular momentum of the photon are absorbed by the matter, usually governed by electric dipole selection rules, and 3. Scattering, in which the photon exits the matter but with its energy or trajectory altered by the interaction. A scientist uses the 528.7 nm output from an Art laser to study the vibrational energy levels of pyrimidine. A large peak due to the ring breathing vibration of the molecule is observed in the Stokes Raman spectrum at 990 cm-. What is the wavelength in nm of the photons corresponding to this peak that the scientist is detecting? Enter the wavelength of the scattered photons. When light interacts with matter, a dipole moment is induced by the electric field, E, of the radiation: HA ? Hin = Value proportional to a, the polarizability of the molecule. The polarizability of a molecule is the measure of how easily the electron density can be redistributed and the polarizability can change while the molecule is vibrating: Submit Previous Answers Request Answer da a = a0+ aQ dQ where dQ is the change in the geometry of the molecule. It is the change in polarizability with changing molecular geometry that leads to the Raman effect. What results are scattered X Incorrect; Try Again; 5 attempts remaining photons with no change in energy (Rayleigh scattering), photons that are scattered that have lost energy corresponding to the energies of vibrational modes in the molecule (Stokes scattering), and photons that are scattered that have gained energy corresponding to the energies of vibrational modes in the molecule (anti-Stokes scattering). If a vibration does not change the polarizability of the molecule then the polarization derivative will be zero: Provide Feedback Next > da = 0 and that vibrational normal mode will not be Raman active. Constants I Periodic Table When a photon passes through matter, there are three types of light-matter interactions that can occur: Part A 1. Transmission, in which there is no significant interaction between radiation and matter, 2. Absorption, in which the energy and angular momentum of the photon are absorbed by the matter, usually governed by electric dipole selection rules, and 3. Scattering, in which the photon exits the matter but with its energy or trajectory altered by the interaction. A scientist uses the 528.7 nm output from an Art laser to study the vibrational energy levels of pyrimidine. A large peak due to the ring breathing vibration of the molecule is observed in the Stokes Raman spectrum at 990 cm-. What is the wavelength in nm of the photons corresponding to this peak that the scientist is detecting? Enter the wavelength of the scattered photons. When light interacts with matter, a dipole moment is induced by the electric field, E, of the radiation: HA ? Hin = Value proportional to a, the polarizability of the molecule. The polarizability of a molecule is the measure of how easily the electron density can be redistributed and the polarizability can change while the molecule is vibrating: Submit Previous Answers Request Answer da a = a0+ aQ dQ where dQ is the change in the geometry of the molecule. It is the change in polarizability with changing molecular geometry that leads to the Raman effect. What results are scattered X Incorrect; Try Again; 5 attempts remaining photons with no change in energy (Rayleigh scattering), photons that are scattered that have lost energy corresponding to the energies of vibrational modes in the molecule (Stokes scattering), and photons that are scattered that have gained energy corresponding to the energies of vibrational modes in the molecule (anti-Stokes scattering). If a vibration does not change the polarizability of the molecule then the polarization derivative will be zero: Provide Feedback Next > da = 0 and that vibrational normal mode will not be Raman active.
Expert Answer:

Related Book For 

Intermediate Accounting
ISBN: 978-0324300987
10th Edition
Authors: Loren A Nikolai, D. Bazley and Jefferson P. Jones
Posted Date:
Students also viewed these chemistry questions
-
There are three types of oxygen160, 170, and 180. What are these varieties called?
-
There are three types of risk-related financing options: business risk, financial risk, and instrument risk. List each of the following under the appropriate risk concept: Bonds .................
-
There are three types of money: commodity money, commodity-backed money, and fiat money. Which type of money is used in each of the following situations? a. Bottles of rum were used to pay for goods...
-
In Exercises find the second derivative of the function. f(x) = x + 3x-3
-
Here is a quote from an investment website about an investment strategy using options: One strategy investors are applying to the XYZ options is using "synthetic stock."Asynthetic stock is created...
-
Best Drugs is a distributor of pharmaceutical products. Its ABC system has five activities: Activity Area Cost Driver Rate in 2013 1. Order processing .........$ 42 per order 2. Line-item ordering...
-
Identify and describe the key attributes that might mitigate a red flag, suggesting that no bad act or compliance issues exist.
-
Homepride Carpet Company manufactures carpets. Fiber is placed in process in the Spinning Department, where it is spun into yarn. The output of the Spinning Department is transferred to the Tufting...
-
the tuition fee for maths tuition for john is expected to be 6500 per year if he attend tuition for 4 year starting from 4 years from now how much does he have to have in her account which yields 6...
-
You have just been hired as a brand manager at Kelsey-White, an American multinational consumer goods company. Recently the firm invested in the development of K-W Vision, a series of systems and...
-
Presented below is pension information related to Sofa Inc. for the calendar year 2020. Current service costs............. Interest on accrued benefit obligation................. Expected and actual...
-
How might different settings for a focus group affect the overall quality of the research?
-
Explain what a riskless hedge is and how the riskless hedge concept is used in the Black-Scholes OPM.
-
What aspects of computer-assisted surveys make them attractive to marketing researchers?
-
Provide examples of decision alternatives. How might these alternatives affect the outcome of the research?
-
Explain why thematic analysis is a useful technique for analyzing qualitative data.
-
A rental company spent $2500 on a new air compressor 7 years ago. The annual rental income from the compressor has been $750. The $100 spent on maintenance the first year has in- creased each year by...
-
What key concerns must functional tactics address in marketing? Finance? POM? Personnel?
-
What are two purposes of a companys balance sheet?
-
On December 8, 2007, Lynch Incorporated sold $9,000 of merchandise with terms 2/10, n/EOM. On December 18, 2007, collections were made on sales originally billed for $5,000, and on December 31, 2007,...
-
Why is a preferred stock similar to a long-term bond? Why is it similar to common stock?
-
When you exhale, all of the air in your lungs must exit through the trachea. If you exhale through your nose, this air subsequently leaves through your nostrils. The area of your nostrils is less...
-
A balloon is at a height of \(5.0 \mathrm{~km}\) and is descending at a constant rate. The buoyancy force is directed ; the drag force is directed A. Up, up B. Up, down C. Down, up D. Down, down...
-
A balloon is launched at sea level, where the air pressure is \(100 \mathrm{kPa}\). The density in the hot-air chamber is \(1.0 \mathrm{~kg} / \mathrm{m}^{3}\). What is the density of the air when...

Study smarter with the SolutionInn App


