An ideal gas (the weight is m kg) in an idealized piston-cylinder assembly undergoes a series...
Fantastic news! We've Found the answer you've been seeking!
Question:
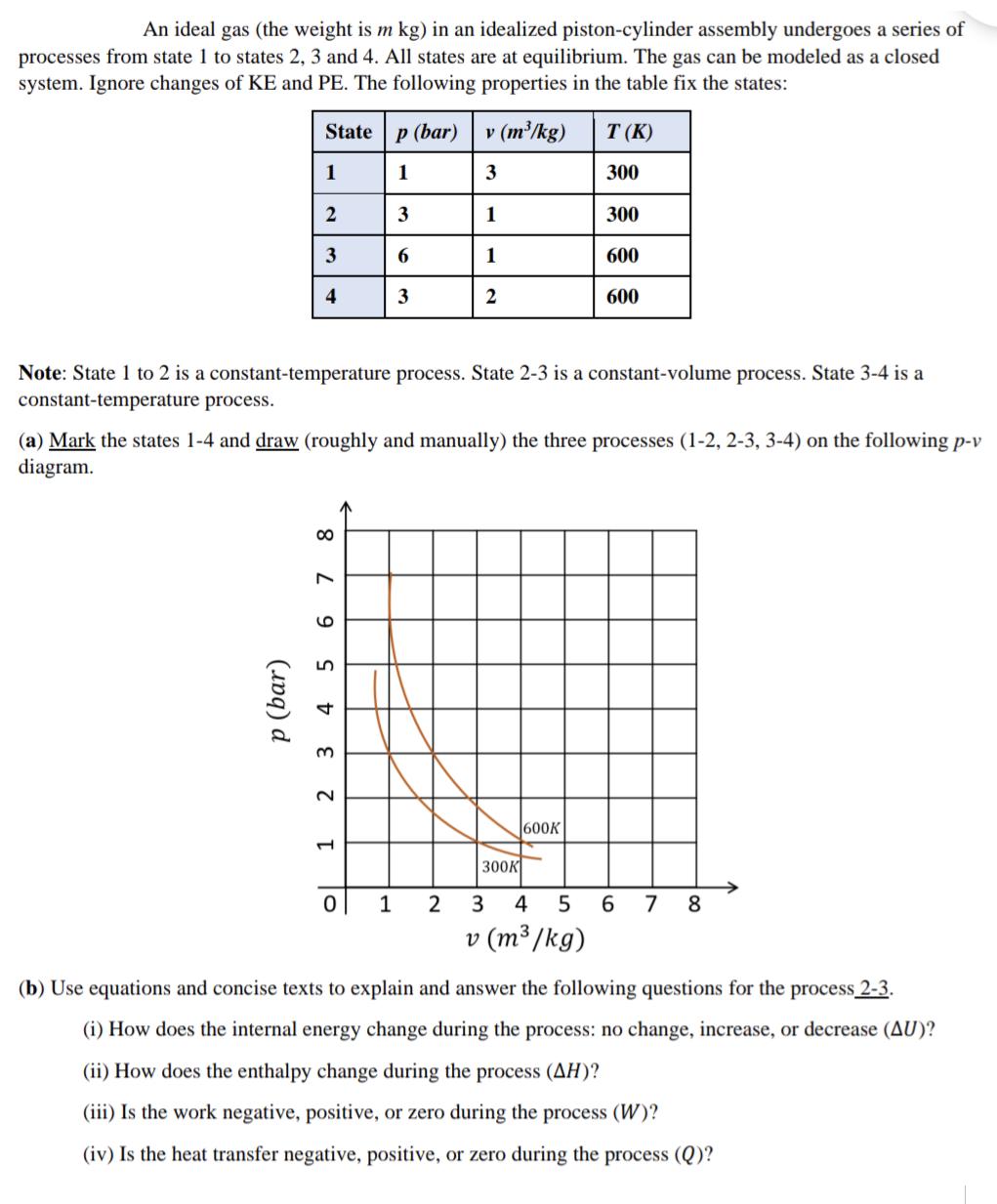
Transcribed Image Text:
An ideal gas (the weight is m kg) in an idealized piston-cylinder assembly undergoes a series of processes from state 1 to states 2, 3 and 4. All states are at equilibrium. The gas can be modeled as a closed system. Ignore changes of KE and PE. The following properties in the table fix the states: State 1 p (bar) 2 3 4 8 6 7 5 tz 3 2 1 Note: State 1 to 2 is a constant-temperature process. State 2-3 is a constant-volume process. State 3-4 is a constant-temperature process. Ol p (bar) 1 (a) Mark the states 1-4 and draw (roughly and manually) the three processes (1-2, 2-3, 3-4) on the following p-v diagram. 0 3 6 1 3 v (m³/kg) 3 1 1 2 2 300K T (K) 300 600K 300 3 4 5 v (m³/kg) 600 600 6 7 8 (b) Use equations and concise texts to explain and answer the following questions for the process 2-3. (i) How does the internal energy change during the process: no change, increase, or decrease (AU)? (ii) How does the enthalpy change during the process (AH)? (iii) Is the work negative, positive, or zero during the process (W)? (iv) Is the heat transfer negative, positive, or zero during the process (Q)? An ideal gas (the weight is m kg) in an idealized piston-cylinder assembly undergoes a series of processes from state 1 to states 2, 3 and 4. All states are at equilibrium. The gas can be modeled as a closed system. Ignore changes of KE and PE. The following properties in the table fix the states: State 1 p (bar) 2 3 4 8 6 7 5 tz 3 2 1 Note: State 1 to 2 is a constant-temperature process. State 2-3 is a constant-volume process. State 3-4 is a constant-temperature process. Ol p (bar) 1 (a) Mark the states 1-4 and draw (roughly and manually) the three processes (1-2, 2-3, 3-4) on the following p-v diagram. 0 3 6 1 3 v (m³/kg) 3 1 1 2 2 300K T (K) 300 600K 300 3 4 5 v (m³/kg) 600 600 6 7 8 (b) Use equations and concise texts to explain and answer the following questions for the process 2-3. (i) How does the internal energy change during the process: no change, increase, or decrease (AU)? (ii) How does the enthalpy change during the process (AH)? (iii) Is the work negative, positive, or zero during the process (W)? (iv) Is the heat transfer negative, positive, or zero during the process (Q)?
Expert Answer:
Related Book For 

Business Communication Essentials a skill based approach
ISBN: 978-0132971324
6th edition
Authors: Courtland L. Bovee, John V. Thill
Posted Date:
Students also viewed these general management questions
-
The PV diagram in Fig. 15-23 shows two possible states of a system containing 1.35 moles of a monatomic ideal gas, (P1 = P2 = 455 N/m2, V1 = 2.00 m3, V2 = 8.00m3.) (a) Draw the process which depicts...
-
(Multiple choice) (1) The temperature change of two blocks of masses MA and MB is the same when they absorb equal amounts of heat. It follows that the specific heats are related by (a) cA =...
-
Different theories about early childhood inform approaches to children's learning and development. Early childhood educators draw upon a range of perspectives in their work ..." (EYLF p.12)....
-
Classify the following soils according to the USCS: (a) A sample of well-graded gravel with sand has 73% fine to coarse sub-angular gravel, 25% fine to coarse sub-angular sand, and 2% fines. The...
-
Blossom Company produces flash drives for computers which have variable costs of $5 per flash drive to produce. Each flash drive sells for $10 each. During the current month, 1240 flash drives were...
-
In multistage compression with intercooler, the compression obtained is: (a) Isothermal (b) Adiabatic (c) Polytropic (d) None of the above
-
Breyer Company purchased packaging equipment on January 3, 2010, for $101,250. The equipment was expected to have a useful life of three years, or 25,000 operating hours, and a residual value of...
-
Analyst Scenario: Public Company & Industry Classification Quiz for Tesla (TSLA) Question 1: Tesla Please provide the name of your company and the ticker or stock symbol, which can be found on any...
-
Of all the price analysis methodologies that exist, which are the foundational methods. Longing & Shorting. Trading & Investing. Volume & sentiment. On-chain & Macroeconomic. Mean reversion &...
-
1.Explain the meaning of the following terms Knowledge Skill- Talent- Competency- Profession - 2. Describe four managerial skills that managers need to be effective in running a sugar milling...
-
Cameron Electronics makes and sells power banks for phones. Each power bank has a selling price of $90. The following cost data per power bank which is based on a capacity of 4,000 units per period....
-
Ben has accumulated a lot of credit card debt over the years, but somehow he always manages to qualify for more credit. He has opened several credit cards that offer an introductory zero percent...
-
Complete the box below to match your ideal values and culture to trends that you and your team can personally engage with to manifest the culture for a new startup. Worth How value becomes an ideal...
-
Lululemon is considering a new project that improves inventory management for the next 5 years. The project has similar risks as the overall firm. The new technology will require an investment in...
-
what are some of the requirements of hospice social worker Explain with Example
-
MML Inc. a CCPC provides you this year's income tax related information : Active Business Income Investment Income Dividends Income "non-connected" Dividends Income"connected" CDN ABI Income $500,000...
-
Why is disclosure of depreciation or amortization methods and rates so important?
-
A not-so-secret secret is getting more attention than you'd really like after an article in Business week gave the world an inside look at how much money you and other electronics retailers make from...
-
What are the six traits of professionalism?
-
Read the following document and (a) Analyze the strengths and weaknesses of each sentence and (b) Revise the message so that it follows this chapter's guidelines. I am responding to your letter of...
-
Which of the following is true with regard to Cresseys theory of crime causation? 1. For a fraud to occur, only one of the fraud triangle attributes (perceived pressure, perceived opportunity, or...
-
Select the statement that best describes organizational versus organized crime: 1. Organizational and organized crime are essentially the sameboth center on crimes committed by organizations. 2. Some...
-
Identify from Cresseys research the six situational categories that cause nonshareable problems.

Study smarter with the SolutionInn App



