Determine the stoichiometric coefficients for the combustion of PMMA and oxygen (for stoichiometric conditions, d = 0).
Fantastic news! We've Found the answer you've been seeking!
Question:
Determine the stoichiometric coefficients for the combustion of PMMA and oxygen (for stoichiometric conditions, d = 0).
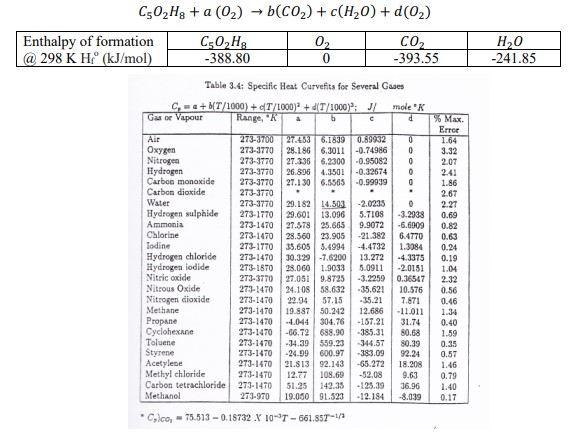
Transcribed Image Text:
C502H8a (02) b(CO2)+ c(HO) + d(0) Enthalpy of formation @ 298 K Hr (kJ/mol) C502H8 -388.80 -> 02 0 CO -393.55 HO -241.85 Table 3.4: Specific Heat Curvefits for Several Gases C, a+b(T/1000)+(T/1000)+d(T/1000); J/ mole "K Gas or Vapour Range, "K a b d % Max. Error Air 273-3700 Oxygen Nitrogen Hydrogen Carbon monoxide Carbon dioxide 27.453 6.1839 0.89932 273-3770 28.186 6.3011 -0.74986 273-3770 27.336 6.2300 -0.95082 273-3770 26.896 4.3501 -0.32674 273-3770 27.130 6.5565 273-3770 0 1.64 0 3.32 0 2.07 2.41 -0.99939 0 1.86 . . 2.67 Water Hydrogen sulphide Ammonia Chlorine 273-3770 29.182 14.503 273-1770 29.601 13.096 273-1470 27.578 25.665 273-1470 28.560 23.905 -2.0235 0 2.27 5.7108 -3.2938 0.69 9.9072 -6.6909 0.82 -21.382 6.4770 0.63 Iodine Hydrogen chloride 273-1770 273-1470 Hydrogen iodide 273-1870 Nitric oxide 273-3770 Nitrous Oxide Nitrogen dioxide Methane Propane Cyclohexane 35.605 5.4994 30.329 -7.6200 28.060 1.9033 27.051 9.8725 273-1470 24.108 58.632 273-1470 22.94 57.15 273-1470 19.887 50.242 273-1470 -4.044 304.76 273-1470 -66.72 688.90 -385.31 -4.4732 1.3084 0.24 13.272 -4.3375 0.19 5.0911 -2.0151 1.04 -3.2259 0.36547 2.32 -35.621 10.576 -35.21 7.871 0.56 0.46 12.686 -11.011 1.34 -157.21 31.74 0.40 80.68 1.59 Toluene 273-1470 -34.39 559.23 -344.57 80.39 0.35 Styrene 273-1470 -24.99 600.97 -383.09 92.241 0.57 Acetylene 273-1470 21.813 92.143 Methyl chloride 273-1470 12.77 108.69 Carbon tetrachloride 273-1470 51.25 142.35 -125.39 36.96 Methanol 273-970 19.050 91.523 -12.184 -8.039 Clco, 75.513-0.18732 X 10-7-661.857-1/3 -65.272 18.208 1.46 -52.08 9.63 0.79 1.40 0.17 C502H8a (02) b(CO2)+ c(HO) + d(0) Enthalpy of formation @ 298 K Hr (kJ/mol) C502H8 -388.80 -> 02 0 CO -393.55 HO -241.85 Table 3.4: Specific Heat Curvefits for Several Gases C, a+b(T/1000)+(T/1000)+d(T/1000); J/ mole "K Gas or Vapour Range, "K a b d % Max. Error Air 273-3700 Oxygen Nitrogen Hydrogen Carbon monoxide Carbon dioxide 27.453 6.1839 0.89932 273-3770 28.186 6.3011 -0.74986 273-3770 27.336 6.2300 -0.95082 273-3770 26.896 4.3501 -0.32674 273-3770 27.130 6.5565 273-3770 0 1.64 0 3.32 0 2.07 2.41 -0.99939 0 1.86 . . 2.67 Water Hydrogen sulphide Ammonia Chlorine 273-3770 29.182 14.503 273-1770 29.601 13.096 273-1470 27.578 25.665 273-1470 28.560 23.905 -2.0235 0 2.27 5.7108 -3.2938 0.69 9.9072 -6.6909 0.82 -21.382 6.4770 0.63 Iodine Hydrogen chloride 273-1770 273-1470 Hydrogen iodide 273-1870 Nitric oxide 273-3770 Nitrous Oxide Nitrogen dioxide Methane Propane Cyclohexane 35.605 5.4994 30.329 -7.6200 28.060 1.9033 27.051 9.8725 273-1470 24.108 58.632 273-1470 22.94 57.15 273-1470 19.887 50.242 273-1470 -4.044 304.76 273-1470 -66.72 688.90 -385.31 -4.4732 1.3084 0.24 13.272 -4.3375 0.19 5.0911 -2.0151 1.04 -3.2259 0.36547 2.32 -35.621 10.576 -35.21 7.871 0.56 0.46 12.686 -11.011 1.34 -157.21 31.74 0.40 80.68 1.59 Toluene 273-1470 -34.39 559.23 -344.57 80.39 0.35 Styrene 273-1470 -24.99 600.97 -383.09 92.241 0.57 Acetylene 273-1470 21.813 92.143 Methyl chloride 273-1470 12.77 108.69 Carbon tetrachloride 273-1470 51.25 142.35 -125.39 36.96 Methanol 273-970 19.050 91.523 -12.184 -8.039 Clco, 75.513-0.18732 X 10-7-661.857-1/3 -65.272 18.208 1.46 -52.08 9.63 0.79 1.40 0.17
Expert Answer:

Related Book For 

Exploring Engineering An Introduction to Engineering and Design
ISBN: 978-0123747235
2nd edition
Authors: Philip Kosky, George Wise, Robert Balmer, William Keat
Posted Date:
Students also viewed these mechanical engineering questions
-
This assignment requires you to complete the 2022 tax reporting for a fictional woman named Anna Smith. Question 1 T1 - step 4 - line 66 This is Anna's taxable income Answer: Question 2 T1 - step...
-
The following additional information is available for the Dr. Ivan and Irene Incisor family from Chapters 1-5. Ivan's grandfather died and left a portfolio of municipal bonds. In 2012, they pay Ivan...
-
Calculate the CGT payable in relation to each of the following disposals, assuming in each case that the annual exemption is fully utilised against other gains, that there are no allowable losses and...
-
A mole of liquid octane at 1 atm and 25 8C is burned in an insulated rigid container with 120% theoretical air. Find the final temperature, pressure, and volume if the reaction is supplied with a)...
-
Amazon.com, lnc.'s financial statements are presented in Appendix D. Financial statements of Walmart Inc. are presented in Appendix E. The complete annual reports of Amazon and Walmart, including the...
-
In the spring of 1999, Source Associates, Inc. (Source), and Conrad A. Mamajek, Inc. (CAM), entered into a joint venture to act as a middleman for the sale of polymers manufactured by Mitsui...
-
On January 1, 2008, Carlin Corporation issued $2,400,000 of 5-year, 8% bonds at 95; the bonds pay interest semiannually on July 1 and January 1. By January 1, 2010, the market rate of interest for...
-
A combined solar and auxiliary energy system is used to meet the same load as in Example 12.5. The total cost of the system to cover 65% of the load (solar fraction) is $20,000. The owner will pay a...
-
What are the ingredients of Apples success as the most innovative company in the world?
-
2. (15 points) In the episode, The One with the Tea Leaves, Chandler moves in with Monica, Monica finds out Chandler has two identical copies of the 1982 hit Broadway musical, Annie. Chandler loves...
-
What is reading and writing as symbiotic processes and explain the implications this has for language teaching. Q2 Is critical writing the same as criticism? Describe TWO ways in which the two are...
-
Washington County has the following five banks in its market area: Bank Total Assets First National Bank $1,200 million Springfield Bank $730 million Third State Bank $280 million Georgetown Bank...
-
The following table supplies data of the inflation rate and the corresponding prime lending rate during the same time period. Inflation rate Prime lending rate (%) (z) (%) (y) 3,3 6,2 11,0 9,1 5,8...
-
A producer of wine in Italy forecasted that next January 22 the sales will be 50,000 and that these sales will increase in a 10% per month during the next six months. We assume that all sales are...
-
A bank offers a 24-month installment loan with an APR of 10.5%. Martha wishes to use the loan to finance a sofa for $1,000. After using Table 13-1 from your text to find the finance charge, calculate...
-
14. In testing the existence assertion, an auditor ordinarily works from the a. Financial statements to the accounting records. b. General journal to the general ledger. c. Supporting evidence to the...
-
Suppose you were going to accelerate a 2000. kg car by the Rube Goldberg contraption shown in the following figure. The fan (A) blows apples (C) off the tree (B) into the funnel and thus into the bag...
-
Based on your measurements and calculations (exercise 13), what is the compression ratio, CR, of the lawnmower engine? Refer to a 3.5 horsepower lawn mower engine at a nominal speed of 2.50 10 3...
-
The same reformer as in the previous problem has a problemthe 3.00 liters/s of H 2 is contaminated with 0.600 liters/minute of CO. Assume the shift reactor removes this contaminant down to 10.0 ppm...
-
Which of the following steps in the acquisition of goods and services occurs first? a. Appropriation b. Encumbrance c. Budget d. Expenditure
-
Fixed assets purchased from general fund revenue were received. What account, if any, should have been debited in the general fund? a. None b. Fixed Assets c. Expenditures d. Fund BalanceUnassigned
-
When a governmental units Estimated Revenues Control account is closed out at the end of the fiscal year, the excess of estimated revenues over estimated appropriations is a. Debited to Fund...

Study smarter with the SolutionInn App


