Write the balanced chemical equation and solve each of the following. 1. Aluminum metal (Al) and...
Fantastic news! We've Found the answer you've been seeking!
Question:

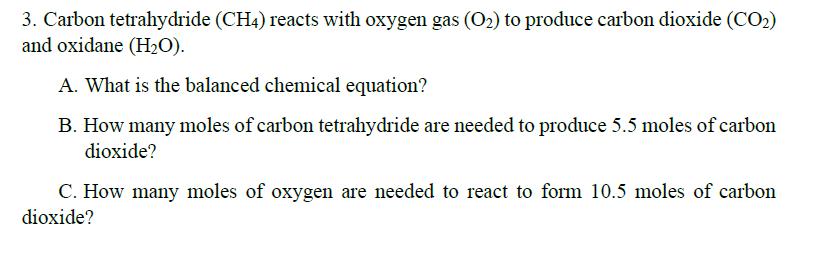
Transcribed Image Text:
Write the balanced chemical equation and solve each of the following. 1. Aluminum metal (Al) and hydrogen chloride (HCI) react to form aluminum chloride (AICl3) and hydrogen gas (H2). A. What is the balanced chemical equation? B. How many moles of aluminum metal are needed to produce 4 moles of aluminum chloride? C. How many moles of hydrogen chloride are needed to react with this number of moles of aluminum metal? 2. Aluminum bromide (AIB13) and sodium hydroxide (NaOH) react to form aluminum hydroxide (Al(OH)3) and sodium bromide (NaBr). A. What is the balanced chemical equation? B. How many moles of sodium bromide can be formed from 2.5 moles of aluminum bromide? C. How many moles of aluminum hydroxide may be formed from 9 moles of sodium hydroxide? 3. Carbon tetrahydride (CH4) reacts with oxygen gas (O2) to produce carbon dioxide (CO2) and oxidane (H2O). A. What is the balanced chemical equation? B. How many moles of carbon tetrahydride are needed to produce 5.5 moles of carbon dioxide? C. How many moles of oxygen are needed to react to form 10.5 moles of carbon dioxide? Write the balanced chemical equation and solve each of the following. 1. Aluminum metal (Al) and hydrogen chloride (HCI) react to form aluminum chloride (AICl3) and hydrogen gas (H2). A. What is the balanced chemical equation? B. How many moles of aluminum metal are needed to produce 4 moles of aluminum chloride? C. How many moles of hydrogen chloride are needed to react with this number of moles of aluminum metal? 2. Aluminum bromide (AIB13) and sodium hydroxide (NaOH) react to form aluminum hydroxide (Al(OH)3) and sodium bromide (NaBr). A. What is the balanced chemical equation? B. How many moles of sodium bromide can be formed from 2.5 moles of aluminum bromide? C. How many moles of aluminum hydroxide may be formed from 9 moles of sodium hydroxide? 3. Carbon tetrahydride (CH4) reacts with oxygen gas (O2) to produce carbon dioxide (CO2) and oxidane (H2O). A. What is the balanced chemical equation? B. How many moles of carbon tetrahydride are needed to produce 5.5 moles of carbon dioxide? C. How many moles of oxygen are needed to react to form 10.5 moles of carbon dioxide?
Expert Answer:

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
Write the balanced chemical equation and the Ksp expression for the slight solubility of Mg(OH)2(s).
-
Write the balanced chemical equation and the Ksp expression for the slight solubility of Fe2(SO4)3(s).
-
From the following written description, write the balanced chemical equation for the reaction including state symbols. A diatomic gaseous molecule that contains 17 protons per atom is reacted with a...
-
In the United States, a principal responsibility for preserving endangered species (e.g., a pair of endangered birds that chooses to nest on private land) and the costs of exercising that...
-
= 1 + 2x a. Decide, at the 10% significance level, whether the data provide sufficient evidence to conclude that x is useful for predicting y. b. Find a 90% confidence interval for the slope of the...
-
Prove in each of the following ways. a. By integrating the differential cross section computed using the first-order born approximation. b. By applying the optical theorem to the forward-scattering...
-
Explain the difference between an asset and an expense in conventional financial accounting. Are pollution permits an asset? Comment on the view that letting companies hold pollution allowances as...
-
Discuss the importance of B-to-B marketing and a strong B-to-B brand to GE.
-
For the last 2 years sales and profits are as follows: Rs. Rs. 2009 sales 5,00,000 profit - 40,000 2010 sales 6,00,000 profit - 60,000 Calculate P/V ratio, BEP sales, sales to get a profit of Rs....
-
Suppose Allied Company's bonds had a deferred call provision that permitted the company, if it desired, to call them 10 years after their issue date at a price of $1,100. Suppose further that market...
-
a) The energy stored in a CC bond is 348 10.mola and its length is 0.154 nm. Assume that the bond stretches twice its equilibrium length before rupture. Calculate the lifting capacity of a single CC...
-
2. Convert the following for loop to a while loop for loop sum= 0; for k=1: 100; sum +k; end while loop
-
How much is the total output VAT? How much is the total claimable input VAT? How much is the VAT payable for the 3rd quarter of 2021? The following information were lifted from the subsidiary...
-
Gleeson Ltd. has completed an initial public offering via a Dutch auction. Before the IPO, the firm had an existing capital of 1,200 million shares. The terms of the IPO were as follows: Initial...
-
The formula R = 1 R 1 + 1 gives the total electrical resistance R (in ohms, 2) when two resistors of resistance R, and R, are connected in parallel R (a) Simplify the complex fraction on the right....
-
After 10 years of marriage, Kino Lam died as a resident citizen of the Philippines. His wife presented the following properties and deductions estate tax purposes: Properties 1. Residential house and...
-
1. In the following statement, printf("Result = %f", i + (j * 5) % 2); a. How many tokens are there? b. What are tokens? 2. Write one C statement by using printf function to print the following...
-
Rewrite the code of Figure 7.3 in Ada, Java, or C#. Figure 7.3: template class queue { item items [max_items]; int next_free, next_full, num_items; public: queue () : next_free (0), next_full(0),...
-
Which of the following molecules contains only double bonds? If none do, so state. a. NCCN b. CO 2 c. C 2 H 4 d. O 3 e. N 2
-
A 6.01-g aqueous solution of isopropyl alcohol contains 3.67 g of isopropyl alcohol. What is the mass percentage of isopropyl alcohol in the solution?
-
What is the difference between a sigma bond and a pi bond?
-
An important statistic to consider when using a classical statistical sampling audit plan is the population variability. The population variability is measured by the a. Sample mean. b. Standard...
-
During the course of an audit engagement, Mr. Command, the senior, decided to use non-statistical sampling on a certain substantive test. The sampling plan included the following: Required: a. If the...
-
An auditor selects a preliminary sample of 100 items out of a population of 1,000 items. The sample statistics generate an arithmetic mean of \($60\), a standard deviation of \($6\), and a standard...

Study smarter with the SolutionInn App


