Exercise 10.35 Determine the electron geometry, molecular geometry, and idealized bond angles for each of the...
Fantastic news! We've Found the answer you've been seeking!
Question:

Transcribed Image Text:
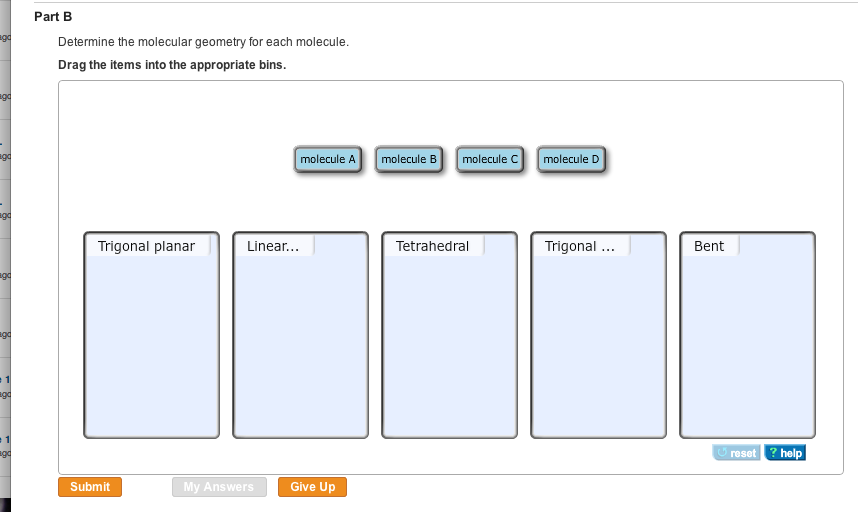
Exercise 10.35 Determine the electron geometry, molecular geometry, and idealized bond angles for each of the following molecules. In which cases do you expect deviations from the idealized bond angle? 1. PF3 2. SBT₂ 3. CH₂Br 4. BCls ago ago go ago agd go 1 ago 1 ago Part B Determine the molecular geometry for each molecule. Drag the items into the appropriate bins. Trigonal planar Submit Linear... My Answers molecule A Give Up molecule B molecule C Tetrahedral molecule D Trigonal ... Bent reset ? help Exercise 10.35 Determine the electron geometry, molecular geometry, and idealized bond angles for each of the following molecules. In which cases do you expect deviations from the idealized bond angle? 1. PF3 2. SBT₂ 3. CH₂Br 4. BCls ago ago go ago agd go 1 ago 1 ago Part B Determine the molecular geometry for each molecule. Drag the items into the appropriate bins. Trigonal planar Submit Linear... My Answers molecule A Give Up molecule B molecule C Tetrahedral molecule D Trigonal ... Bent reset ? help
Expert Answer:

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
For each of the following molecules that contain polar covalent bonds, indicate the positive and negative ends of the dipole, using the symbol v. Refer to Table 1.2 as needed. (a) HCI (c) HI (e) HOCI...
-
For each of the following molecules that contain polar covalent bonds, indicate the positive and negative ends of the dipole, using the symbol v. Refer to Table 1.2 as needed. (a) HCl (b) ICl (c) HI...
-
Give an example for each of the following control activities in the conversion cycle: transaction authorization, segregation of duties, and access.
-
Suppose that a 7% semi-annual coupon bond with a time to maturity of 8 years and a par value of $100 has a price of $106,4. This bond is first callable in 6 years at a redemption price of $104,8....
-
How much tax did Ajinkya keep from the tax it collected based on the purchases it made from supplier A? Ajinkya Electronic Systems, a company in India that manufactures many different electronic...
-
1. What are the three phases in the IT strategic planning process? Describe the steps in each phase. 2. Intel formulates a two-to-five-year strategic plan. Given this time frame, why is the plan...
-
Great Wall Chinese Cuisine had \($93,000\) of total assets and \($31,000\) of total stock holders equity at May 31,2010. At May 31, 2011, Great Wall Chinese Cuisine had assets totaling \($147,000\)...
-
Phil Emley owns a garage and is contemplating purchasing a tire retreading machine for $18,000. After estimating costs and revenues, Phil projects a net cash flow from the retreading machine of...
-
A firm will need to take out a$200,000 loan 60days from now for a180-day interval. It purchases a call with X=4.1%.The call expires in 60days and the underlying is a180-day corporate loan rate. What...
-
In 2023, Lisa and Fred, a married couple, have taxable income of $400,000. If they were to file separate tax returns, Lisa would have reported taxable income of $225,000 and Fred would have reported...
-
Actuarial finance Consider an investor, who is an expected utility maximizer, with a utility function u (x) = x. The investor has 100 currency units and the opportunity to take long positions in a...
-
Shown as follows is a segmented income statement for Drexel-Hall during the current month: Profit Centers Sales Variable costs Drexel-Hall Dollars % Store 1 Dollars Store 2 % Dollars % Dollars $...
-
Consider the following network of routers (A, B, C and D) in which the numbers above each link indicate the relative routing cost of that link. Assume a distance vector routing algorithm has been...
-
What must go inside of class B in this code? public interface A { public void doStuff(); public static int x = 2; } public class 8 implements A { // ? }
-
Three charged metal spheres, 91 = -5.54 nC, 92 2.90 nC, and 93 -2.56 nC, are arrayed in the xy plane so that they form an equilateral triangle with d = 12.0 cm, as shown in the figure. What is the...
-
Let f(z) = f(2) = f( 1) = (G) - 32 I H-3 Find the following. Write answers as fractions, not decimals.
-
One form of Ohm's law is: E = IXR where, E is the applied voltage and is measured in volts (V) I is the circuit current and is measured in amperes (A) R is the circuit resistance and is measures...
-
What is your assessment of the negotiations process, given what you have studied? What are your recommendations for Mr. Reed? You must justify your conclusions
-
Many oxidation reduction reactions can be balanced by inspection. Try to balance the following reactions by in-spection. In each reaction, identify the substance reduced and the substance oxidized....
-
The decomposition of nitrosyl chloride was studied: 2NOCl(g) 2NO(g) + Cl2(g) The following data were obtained, where a. What is the rate law? b. Calculate the rate constant. c. Calculate the rate...
-
Using data from Appendix 4, calculate Ho, So, and K (at 298 K) for the synthesis of ammonia by the Haber process: N2(g) + 3H2(g) 2NH3(g) Calculate G for this reaction under the following conditions...
-
What forms of business organization are permitted under Rule 505-Form of Practice and Firm Name?
-
There currently are thirteen Rules of Conduct. Listed below are circumstances pertaining to some of these rules: 1. A member shall exercise due professional care in the performance of an engagement....
-
a. What aspects of a company's financial statements are covered in the first three reporting standards? b. What is the objective of the fourth standard of reporting?

Study smarter with the SolutionInn App


