For Experiment 3 in Chem 255 Lab, students need to extract the fat trimyristin from the...
Fantastic news! We've Found the answer you've been seeking!
Question:


Transcribed Image Text:
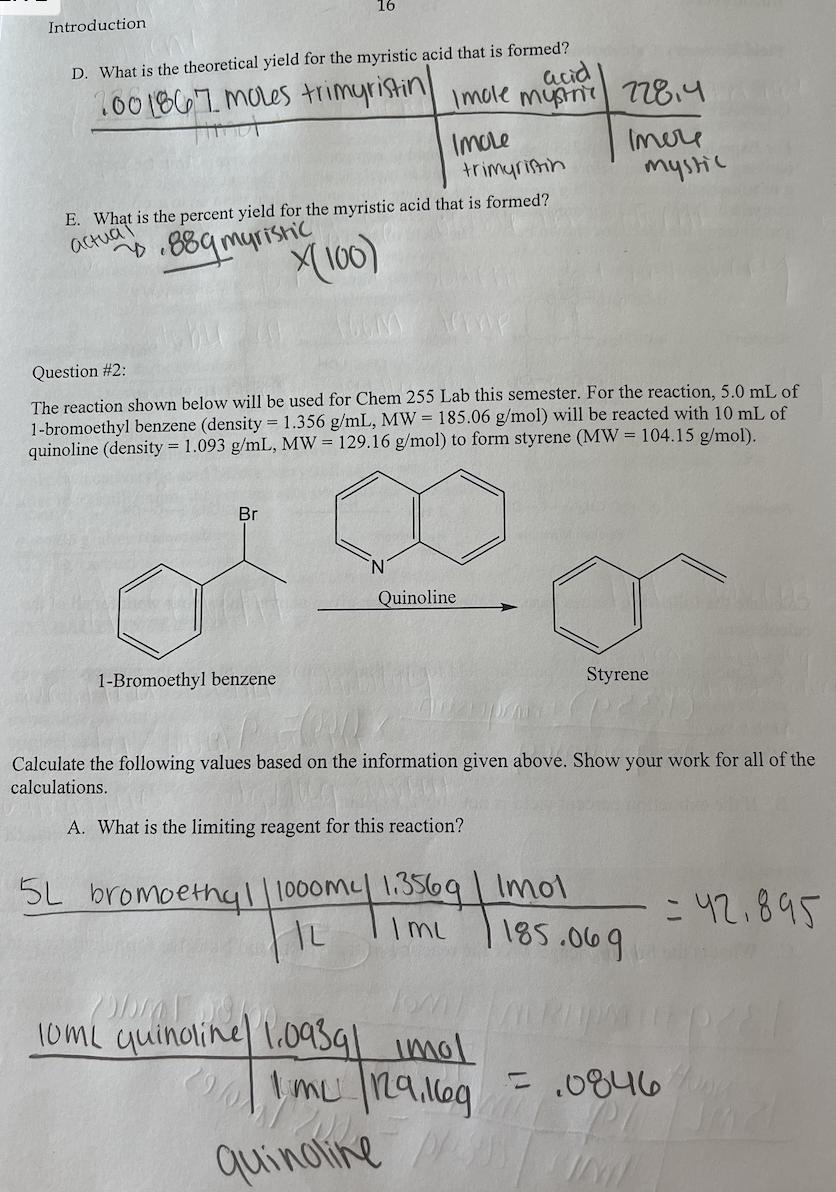
For Experiment 3 in Chem 255 Lab, students need to extract the fat trimyristin from the common cooking spice nutmeg before it can be converted to myristic acid. A student was able to extract 2.65 g of trimyristin from a 15.00 g sample of the nutmeg. This student then went on to use 1.35 g of the trimyristin (MW = 723.2 g/mol) and 15 mL of 2M NaOH to synthesize myristic acid (MW = 228.4 g/mol) using the reactions shown below. The yield of purified myristic acid was 0.88 g. Reaction 1 Reaction 2 H3C(CH2) 12-C-0-CH₂ H3C(CH₂) 12-C-0-CH +3 NaOH 요 H3C(CH₂) 12-C-0-CH₂ Trimyristin 3 H3C(CH₂) 12- -ONa Sodium Myristate + 3 HCI H₂O CH3CH₂OH Reflux Hydrochloric acid H₂O HO–CH2 HO-CH3 H3C(CH₂) 12-C-ONa Jawap HỌ–CH2 Glycerol __8_OH 3 H3C(CH2) 12- Sodium Myristate Myristic acid + 3 NaCl Iculate the following values based on the information given above. Show your work for all of the culations. A. Calculate the extraction percent yield for the trimyristin recovered from the nutmeg. (1359) trimyristinx (100) = 9% 159 nutmeg B. If the extraction percent yield is not 100%, explain why not. Sodium chloride What is the limiting reagent for the reaction of trimyristin and NaOH to form myristic acid? Introduction D. What is the theoretical yield for the myristic acid that is formed? 100 1867 moles trimyristin Hind acid Imole mystn 228.4 Imole trimyristin E. What is the percent yield for the myristic acid that is formed? actual 889 myristic X(100) 16 5L Question #2: The reaction shown below will be used for Chem 255 Lab this semester. For the reaction, 5.0 mL of 1-bromoethyl benzene (density = 1.356 g/mL, MW = 185.06 g/mol) will be reacted with 10 mL of quinoline (density= 1.093 g/mL, MW = 129.16 g/mol) to form styrene (MW = 104.15 g/mol). Br 1-Bromoethyl benzene Quinoline Imere mystic AZ Calculate the following values based on the information given above. Show your work for all of the calculations. A. What is the limiting reagent for this reaction? bromoethyl/1000my 1.3569 | Imol |L I mu 10mL quinoline 1.0939| imol 1 mu 129.169 quinoline // Styrene 185.069 = 42.895 = .0846/00g hil B. What is the theoretical yield for the reaction? 10/1/50846 mol quinaine Imole percent yield OH ole istiting quina he Strene n C. In Exp. 7, the styrene that is produced will be used to make polystyrene. In order to determine the success of the reaction, a ratio of the mass of polystyrene produced is compared to the mass of styrene used in the reaction. What type of calculation should be used for this comparison? 104,159 Imele Question #3: For Experiment 11 in Chem 255 lab, a student used 5.0 mL of 3- octanol (density = 0.818 g/mL, MW= 130.23 g/mol) and allowed it to react with excess H₂SO4 to form a variety of octenes (MW = 112.24 g/mol). The reaction is shown below. IML 71-8.819¹02 H₂SO4 cis- and trans-2-octene cis- and trans-3-octene Calculate the theoretical yield for the octenes produced by this reaction. SmL3-octanol.8189 | Imol | 112.249 130.239 Imol pub= 3,5250 Ormoctar 11₂ 504 For Experiment 3 in Chem 255 Lab, students need to extract the fat trimyristin from the common cooking spice nutmeg before it can be converted to myristic acid. A student was able to extract 2.65 g of trimyristin from a 15.00 g sample of the nutmeg. This student then went on to use 1.35 g of the trimyristin (MW = 723.2 g/mol) and 15 mL of 2M NaOH to synthesize myristic acid (MW = 228.4 g/mol) using the reactions shown below. The yield of purified myristic acid was 0.88 g. Reaction 1 Reaction 2 H3C(CH2) 12-C-0-CH₂ H3C(CH₂) 12-C-0-CH +3 NaOH 요 H3C(CH₂) 12-C-0-CH₂ Trimyristin 3 H3C(CH₂) 12- -ONa Sodium Myristate + 3 HCI H₂O CH3CH₂OH Reflux Hydrochloric acid H₂O HO–CH2 HO-CH3 H3C(CH₂) 12-C-ONa Jawap HỌ–CH2 Glycerol __8_OH 3 H3C(CH2) 12- Sodium Myristate Myristic acid + 3 NaCl Iculate the following values based on the information given above. Show your work for all of the culations. A. Calculate the extraction percent yield for the trimyristin recovered from the nutmeg. (1359) trimyristinx (100) = 9% 159 nutmeg B. If the extraction percent yield is not 100%, explain why not. Sodium chloride What is the limiting reagent for the reaction of trimyristin and NaOH to form myristic acid? Introduction D. What is the theoretical yield for the myristic acid that is formed? 100 1867 moles trimyristin Hind acid Imole mystn 228.4 Imole trimyristin E. What is the percent yield for the myristic acid that is formed? actual 889 myristic X(100) 16 5L Question #2: The reaction shown below will be used for Chem 255 Lab this semester. For the reaction, 5.0 mL of 1-bromoethyl benzene (density = 1.356 g/mL, MW = 185.06 g/mol) will be reacted with 10 mL of quinoline (density= 1.093 g/mL, MW = 129.16 g/mol) to form styrene (MW = 104.15 g/mol). Br 1-Bromoethyl benzene Quinoline Imere mystic AZ Calculate the following values based on the information given above. Show your work for all of the calculations. A. What is the limiting reagent for this reaction? bromoethyl/1000my 1.3569 | Imol |L I mu 10mL quinoline 1.0939| imol 1 mu 129.169 quinoline // Styrene 185.069 = 42.895 = .0846/00g hil B. What is the theoretical yield for the reaction? 10/1/50846 mol quinaine Imole percent yield OH ole istiting quina he Strene n C. In Exp. 7, the styrene that is produced will be used to make polystyrene. In order to determine the success of the reaction, a ratio of the mass of polystyrene produced is compared to the mass of styrene used in the reaction. What type of calculation should be used for this comparison? 104,159 Imele Question #3: For Experiment 11 in Chem 255 lab, a student used 5.0 mL of 3- octanol (density = 0.818 g/mL, MW= 130.23 g/mol) and allowed it to react with excess H₂SO4 to form a variety of octenes (MW = 112.24 g/mol). The reaction is shown below. IML 71-8.819¹02 H₂SO4 cis- and trans-2-octene cis- and trans-3-octene Calculate the theoretical yield for the octenes produced by this reaction. SmL3-octanol.8189 | Imol | 112.249 130.239 Imol pub= 3,5250 Ormoctar 11₂ 504
Expert Answer:

Posted Date:
Students also viewed these chemistry questions
-
The enthalpy of combustion of benzoic acid (C6H5COOH) is commonly used as the standard for calibrating constant-volume bomb calorimeters; its value has been accurately determined to be -3226.7...
-
Under constant-volume conditions, the heat of combustion of benzoic acid (C6H5COOH) is 26.38 kJ/g. A 2.760-g sample of benzoic acid is burned in a bomb calorimeter. The temperature of the calorimeter...
-
Experiment name : Heat of Combustion of benzoic acid and Naphthalene in Bomb Calorimetry Q1:Purpose of experiment Q2:Theory Q3:Discusion Please explain the solution in detail for each question
-
If you deposit $2,000 today into an account earning an annual rate of return of 9 percent, what would your account be worth In 30 years? a. If you deposit $2, 000 today into an account earning an...
-
Mill Creek Golf Club purchased equipment on January 1, 2016, for $31,500. Suppose Mill Creek Golf Club sold the equipment for $22,000 on December 31, 2018. Accumulated Depreciation as of December 31,...
-
Show how you would synthesize the following compounds, using the indicated starting materials. (a) 3-phenylbutan-1-ol from styrene (b) (c) CH CH OCH, from anisole OCH CH,CN from toluene
-
Create recommendations for an organization that is facing resistance to change from its own HR department. What are some of the likely causes of this resistance? How can they be overcome?
-
At the beginning of October, Cornerstone Printers Company budgeted 16,000 books to be printed in October at standard direct materials and direct labor costs as follows: Direct materials $24,000...
-
2 Is it possible to factor 1 - sin x two different ways? If so, show and explain. If not, why not?
-
Consider an abatement cost function C(e,)=(abe) 2 /2b, where is the amount of private investment to reduce abatement costs. There are J identical firms. The social damage function is D(E)=dE 2 /2....
-
Simon Company's year-end balance sheets follow. At December 31 Assets Cash Accounts receivable, net Merchandise inventory Prepaid expenses Plant assets, net Total assets Liabilities and Equity...
-
Why does Alphabet own widely different subsidiaries under the same corporate structure? What are the benefits of this choice? What are the costs? What is the rationale behind the...
-
Identify the problem faced by UPS. Was it a technology problem, an organizational problem, or a management problem? Explain your answer.
-
When conducting a thorough content analysis on your chosen brand, think about the following points: Analyzing the types of platforms used by the brand, what is their digital footprint (i.e. is there...
-
As the personality trait defender or protector was identified at 16personalities typology: Summarize the results of your personality test. What qualities do you possess that are useful to you as a...
-
Identify the provider type you selected earlier in the course and discuss the following, providing support where necessary. Make sure to share your provider type. The three best quality performance...
-
An ophthalmology center negotiates a contract with Aetna's HMO to pay 50% of charges on cataract surgeries. The typical cataract procedure generates $5,000 in charges. Patient A's surgery runs as...
-
a. Determine the domain and range of the following functions.b. Graph each function using a graphing utility. Be sure to experiment with the window and orientation to give the best perspective of the...
-
The group \(\mathrm{D}_{3}\) in Schoenflies notation (32 in international notation, which is read "three-two"; see Table 5.1 ) consists of the proper (those not reflections or inversions) covering...
-
Derive the two-dimensional matrix representation Tic)=(2) Tin)=(3) Tex)=(37) (69) T(c2b)= 1 TO)-(71) 10-(11) TO=(9) = for the group D3, using the basis (e1, e2) defined in the following figure.
-
Prove that the matrix representation of \(\mathrm{D}_{3}\) worked out in Problem 5.6 is irreducible. Data from Problem 5.6 Derive the two-dimensional matrix representation Tic)=(2) Tin)=(3)...

Study smarter with the SolutionInn App

