For a chemical reaction at 60 C, a plot of the inverse of the reactant concentration...
Fantastic news! We've Found the answer you've been seeking!
Question:
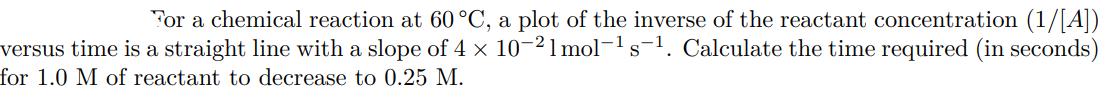
Transcribed Image Text:
For a chemical reaction at 60 C, a plot of the inverse of the reactant concentration (1/[A]) versus time is a straight line with a slope of 4 x 10-21 mol- s. Calculate the time required (in seconds) for 1.0 M of reactant to decrease to 0.25 M. S For a chemical reaction at 60 C, a plot of the inverse of the reactant concentration (1/[A]) versus time is a straight line with a slope of 4 x 10-21 mol- s. Calculate the time required (in seconds) for 1.0 M of reactant to decrease to 0.25 M. S
Expert Answer:
Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
14 Thermodynamics and Thermochemistry . The reaction, MgO(s) + C(s) Mg(s) + CO(g ) 18 The entropy change associated with the conversion of 1 kg of ice at 273 K to water vapours at 383 K is for which...
-
The following additional information is available for the Dr. Ivan and Irene Incisor family from Chapters 1-5. Ivan's grandfather died and left a portfolio of municipal bonds. In 2012, they pay Ivan...
-
Chemical reactions in foods that follow zero-order kinetics exhibit a constant rate of change in the concentration of a reactant or a product. When the concentration is plotted against time, a...
-
The MLC Corporation produces electronic microwave ovens and uses a perpetual inventory system. Its job costing records yield the following information. Job No. 1 m 2 3 4 *5 6 7 Date Finished Sold...
-
An imaginary space rocket launched from the Earth moves with an acceleration w' = 10g which is the same in every instantaneous co-moving inertial reference frame. The boost stage lasted = 1.0 year of...
-
The following income statement was prepared for Rice Company for Year 1: During the year-end audit, the following errors were discovered: 1. An $1,800 payment for repairs was erroneously charged to...
-
Below are listed various costs that are found in organizations. 1. Hamburger buns in a Wendy's outlet. 2. Advertising by a dental office. 3. Apples processed and canned by Del Monte. 4. Shipping...
-
What examples of primary research that Embraces founders completed appear in the case?
-
Nexis Corp. issues 1,380 shares of $8 par value common stock at $18 per share. When the transaction is recorded, what credit entry or entries are made Oa. Common Stock $11,040 and Paid-in Capital in...
-
Hauck Financial Services has a number of passive, buy-and-hold clients. For these clients, Hauck offers an investment account whereby clients agree to put their money into a portfolio of mutual funds...
-
Mahan purchases 1,000 shares of Bluebird Corporation stock onOctober 3,2021 for $300,000. On December 12,2021, Mahan purchasesan additional 750 shares of Bluebird stock for $210,000. Accordingto ma 2...
-
Discuss a minimum of three (3) core components of Grievance Procedures within aviation labor relations and analyze how this can benefit the relationship between union labor and management.
-
Research and explain to the Client the following: What is the Weighted Average Cost of Capital (WACC) Why companies compute weighted average cost of capital Whether WACC can be used to value an...
-
If the market rate is 7 % , calculate the issue price. ( FV of $ 1 , PV of $ 1 , FVA of $ 1 , and PVA of $ 1 ) Will the bonds issue at face amount, a discount, or a premium?
-
Discuss the THREE (3) broad processing categories of services and their marketing implications for Starbucks (American multinational coffeehouse). Support your answers with examples. People...
-
A company has $20 million face value of bonds outstanding that pay a coupon of 10 percent annually and have 8 years to maturity. The bonds have a 12 percent yield to maturity. What value should be...
-
Describe the key elements of the Medicare cost report. Why is this report so important for hospital net revenues? The medicare cost report is the primary means for the government to monitor...
-
The Higher the time period of the financial security the higher the. ............... risk. O a. Maturity O b. Default and Maturity Oc. Default O d. Liquidity
-
Many oxidation reduction reactions can be balanced by inspection. Try to balance the following reactions by in-spection. In each reaction, identify the substance reduced and the substance oxidized....
-
In 1994 chemists at Texas A&M University reported the synthesis of a non-naturally occurring amino acid: a. To which naturally occurring amino acid is this compound most similar? b. A tetrapeptide,...
-
At 25oC, Kp = 2.9 10-3 for the reaction NH4OCONH2(s) 2NH3(g) + CO2(g) In an experiment carried out at 25oC, a certain amount of NH4OCONH2 is placed in an evacuated rigid container and allowed to...
-
The systematic risk of a security is also called its a. b. c. d. perceived risk unique or asset-specific risk market risk fundamental risk
-
A company announces that its earnings have decreased 50 percent from the previous year, but analysts only expected a 25 percent decrease. What is the likely effect on the stock price? a. the stock...
-
The unsystematic risk of a security is also called its a. b. c. d. perceived risk unique or asset-specific risk market risk fundamental risk

Study smarter with the SolutionInn App



