For the following cell Pb | PbCl(s) | PbCl(soln.) | AgCl(s) | Ag the potential at...
Fantastic news! We've Found the answer you've been seeking!
Question:
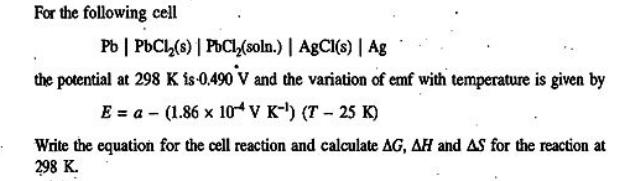
Transcribed Image Text:
For the following cell Pb | PbCl₂(s) | PbCl₂(soln.) | AgCl(s) | Ag the potential at 298 K is 0.490 V and the variation of emf with temperature is given by E = a (1.86 x 104 V K-¹) (T-25 K) Write the equation for the cell reaction and calculate AG, AH and AS for the reaction at 298 K. For the following cell Pb | PbCl₂(s) | PbCl₂(soln.) | AgCl(s) | Ag the potential at 298 K is 0.490 V and the variation of emf with temperature is given by E = a (1.86 x 104 V K-¹) (T-25 K) Write the equation for the cell reaction and calculate AG, AH and AS for the reaction at 298 K.
Expert Answer:
Related Book For 

Chemistry The Central Science
ISBN: 978-0321696724
12th edition
Authors: Theodore Brown, Eugene LeMay, Bruce Bursten, Catherine Murphy, Patrick Woodward
Posted Date:
Students also viewed these chemistry questions
-
The voltage for the following cell is 0.490 V. Find Kb for the organic base RNH2. Pt(s) H2(1.00 bar) RNH2(aq, 0.10 M), RNH+3 Cl(aq, 0.050 M) ||' S.H.E.
-
Calculate the potential for the following cell using : a) Concentrations b) Activities Cr|Cr2(SO4)3 (2.25X10-2 M), PbSO4 (sat'd) | Pb
-
For the following cell pattern, (a) Find the reuse distance if radius of each cell is 2 km. (b) If each channel is multiplexed among 8 users, how many calls can be simultaneously processed by each...
-
j) Assume that one of these portfolio's is the Market Portfolio and all portfolios, except Portfolio G, are fairly priced according to the CAPM. Derive the Treynor Measure for these fairly priced...
-
What are some ways to research specific employers?
-
We saw a general rule that the two protons of a CH 2 group will be chemically equivalent if there are no chirality centers in the compound. An example of an exception is 3-bromopentane. This compound...
-
Two springs have spring constants \(k_{1}\) and \(k_{2}>k_{1}\). Connected as shown in Figure P8.53, they act like one spring. Compute the spring constant of the combination. Is the combination...
-
The following errors in the accounting records of the Chipp & Simon Partnership were discovered on January 10, 2011. The partners share net income and losses as follows: 65%, Chipp; 35%, Simon. 1....
-
The Future Value of a Single Amount Evans invested $187,500 today in a mutual fund earning 4% interest, compounded annually. Round answers to the nearest whole dollar. Do not use negative signs with...
-
Presented below is information related to the sole proprietorship of Kevin Johnson, attorney. Legal service revenue2010.......$350,000 Total expenses2010......... 211,000 Assets, January 1,...
-
According to data from the state blood program, 40 percent of all individuals have group A blood. If six individuals give blood, find the probability that at least 3 of the individuals have group A...
-
Answer the following openended questions: 1.) Scarcity and effciency are not included as part of the foundations of economics, do you think that they should be included or not - support the answer....
-
Suppose there are 2,613 people in the labor force. 265 workers are employed full time but were unemployed last month. Additionally, 91 workers had jobs last month but were laid off and have not found...
-
Using your own words not from the Internet, explain the main ideas of the currents of postclassical economic thought (marginalism, its main representatives) and describe how these ideas help you...
-
Wellness as the "new sustainability" The notes in this unit introduced the idea that green buildings can have positive effects on workplace productivity and employee health. The article provided to...
-
Discuss the magnitude of COVID-19 government relief money and whether or not the money is going to the correct purpose. What are some of the challenges that public administrators face with handling...
-
Starr acquires 80% shareholding. How does that translate to consideration transferred? At the date of acquisition of 80% of its issued shares by Starr Ltd, the equity of Lennon Ltd was: Share...
-
Per Bag Direct materials: 25 pounds of CWhiz-2000 @ $0.08/lb. = $ 2.00 Direct labor: 0.05 hour @ $32.00/hr. = $ 1.60 The company manufactured 100,000 bags of Cheese-Be-Good in December and used...
-
Explain why a mixture formed by mixing 100 mL of 0.100 M CH3COOH and 50 mL of 0.100 M NaOH will act as a buffer.
-
Calculate the enthalpy change for the reaction P4O6 + 2 O2(g) P4O10(s) given the following enthalpies of reaction: Pals) + 302(g) _ P406(s) P4(s) + 5 Odg)- P4010(s) --1 640.1 kJ --2940.1 kJ
-
Consider the reaction Using data in Appendix C, calculate the equilibrium pressure of CO2 in the system at (a) 400 C (b) 180 C. PbCO3(s)PbO(s) CO2(g)
-
What is the activity in \(\mathrm{Bq}\) and in \(\mathrm{Ci}\) of a \(2.0 \mathrm{mg}\) sample of \({ }^{3} \mathrm{H}\) ?
-
The activity of a sample of the cesium isotope \({ }^{137} \mathrm{Cs}\) is \(2.0 \times 10^{8} \mathrm{~Bq}\). Many years later, after the sample has fully decayed, how many beta particles will have...
-
About \(12 \%\) of your body mass is carbon; some of this is radioactive \({ }^{14} \mathrm{C}\), a beta-emitter. If you absorb \(100 \%\) of the\(49 \mathrm{keV}\) energy of each \({ }^{14}...

Study smarter with the SolutionInn App



