Four bonding machines (A, B, C, D) were used in a certain plant to bond circuit...
Fantastic news! We've Found the answer you've been seeking!
Question:

Transcribed Image Text:
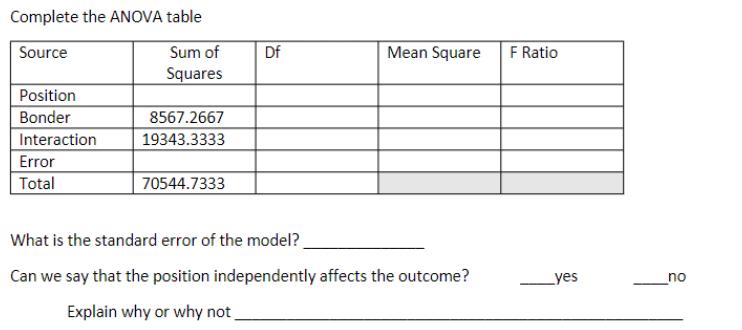
Four bonding machines (A, B, C, D) were used in a certain plant to bond circuit wires into a board. The bonding took place in three different positions (1,2,3) on the piece being bonded. Data including this position follows. Position 2 3 A 204 181 201 203 214 262 246 230 256 288 220 232 235 231 220 Bonder B 197 223 206 232 213 207 259 223 195 197 214 248 191 197 222 C 264 226 228 249 246 255 186 237 236 240 215 176 171 208 180 D 248 138 273 220 186 304 330 268 295 276 208 248 247 220 241 Complete the ANOVA table Source Position Bonder Interaction Error Total Sum of Squares 8567.2667 19343.3333 70544.7333 Df Mean Square What is the standard error of the model? Can we say that the position independently affects the outcome? Explain why or why not F Ratio yes no Four bonding machines (A, B, C, D) were used in a certain plant to bond circuit wires into a board. The bonding took place in three different positions (1,2,3) on the piece being bonded. Data including this position follows. Position 2 3 A 204 181 201 203 214 262 246 230 256 288 220 232 235 231 220 Bonder B 197 223 206 232 213 207 259 223 195 197 214 248 191 197 222 C 264 226 228 249 246 255 186 237 236 240 215 176 171 208 180 D 248 138 273 220 186 304 330 268 295 276 208 248 247 220 241 Complete the ANOVA table Source Position Bonder Interaction Error Total Sum of Squares 8567.2667 19343.3333 70544.7333 Df Mean Square What is the standard error of the model? Can we say that the position independently affects the outcome? Explain why or why not F Ratio yes no
Expert Answer:
Related Book For 

Posted Date:
Students also viewed these accounting questions
-
Find the best alternative using incremental IRR analysis. A B D Initial cost $2000 4000 5000 3000 Annual benefit 800 1300 500 400 Salvage value 1400 2000 1500 3000 Life, in years 6 4 MARR required 6%...
-
Under what conditions can we say that "a thermometer measures its own temperature"?
-
1. [10] Let A = 2 4 a b c d e f g h i 3 5, B = 2 4 d + 5g e+ 5h f+ 5i a 2g b 2h c 2i g h i 3 5, C = 2 4 2g 2h 2i 3d 3e 3f 1a 1b 1c 3 5. Suppose that det A = 5. Find det B = , det C = , and det (AC) =
-
You are appraising a three-year-old, single-tamily residence. The total square footage of the livable area is 2,500. The garage is 500 sq. ft. According to figures obtained from a cost-estimating...
-
Is it possible to predict the annual number of business bankruptcies by the number of firm births (business starts) in the United States? The following data published by the U.S. Small Business...
-
Prepare a presentation on Computer/techno crime, enterprise. The focus of your presentionation must relate to white collar crime - how can white collar crime be committed in your topic area?
-
The bookkeeper of Beautiful World Landscaping, Inc., prepared the companys balance sheet while the accountant was ill. The balance sheet contains numerous errors. In particular, the bookkeeper knew...
-
1. What recommendations can you offer Tanya to be more effective in dealing with Marks tardiness problem? 2. To what extent do you think Tanya should be more tolerant with respect to Marks problem of...
-
2. Full-service financial firms provide a wide variety comprehensive financial services, including retail and commercial banking, insurance, and investment banking, etc. In the past 90 years, the...
-
Refer back to the beginning of this chapter to the excerpt from a Los Angeles Times article about Reed Slatkin's fraud. The article insinuates that the FBI and IRS's raiding of Slatkin's office...
-
soda chips pizza 27 18 40 21 15 35 12 12 25 1.As a cool co-worker you are invited to every party. You choose 5 items which works out to be __ soda __ chips and __ pizza before becoming full according...
-
Two leases in far West Texas are being combined to form the West End Unit. REQUIRED: a. Determine the participation factors for each party, assuming the participation factors are based on the acreage...
-
Heagy Oil Company has production on a lease in Louisiana with the following ownership interest: During April, 5,000 (gross) barrels of oil (after correction for temperature, gravity, and BS&W) were...
-
Differentiate between tax evasion and tax avoidance.
-
What factors should you consider when deciding whether to refinance your mortgage to reduce the monthly payment? How can the refinancing decision be made?
-
Kathleen and Sean Madden have been notified that they are being audited. What should they do to prepare for the audit?
-
Systems Considerations in the Design of an HRIS Vignette Revisited 1. Develop the first few steps of the project plan. 2. Discuss the potential political necessities outlined in this section as they...
-
A police officer pulls you over and asks to search your vehicle because he suspects you have illegal drugs inside your car. Since he doesn't have reasonable suspicion to search your car, legally he...
-
Find the eigen functions and eigen values of dx.
-
For a particle in a box of length l, use the variation function ( = xk (l - x)k for 0 ( x ( l. You will need the integral Where the gamma function obeys the relation The definition of the gamma...
-
(a) For the hydrogen-atom ground state, find the probability of finding the electron farther than 2a from the nucleus. (b) For the H-atom ground state, find the probability of finding the electron in...
-
Calculate the change in entropy for the conversion of \(1 \mathrm{~mol}\) of ice to liquid at \(273 \mathrm{~K}\) and \(1 \mathrm{~atm}\). The latent heat of fusion is \(6500 \mathrm{~J} /...
-
Calculate the change in entropy when 5 moles of an ideal gas expands from a volume of \(5 \mathrm{~L}\) to \(50 \mathrm{~L}\) at \(27^{\circ} \mathrm{C}\).
-
Calculate the entropy change when \(96 \mathrm{~g}\) of methane is heated from \(35^{\circ} \mathrm{C}\) to \(200^{\circ} \mathrm{C}\) at constant volume. Assume \(C_{V}=1.735 \mathrm{~kJ} /...

Study smarter with the SolutionInn App



