How many electrons in an atom can have the quantum numbers n= 4,1 2? O None...
Fantastic news! We've Found the answer you've been seeking!
Question:
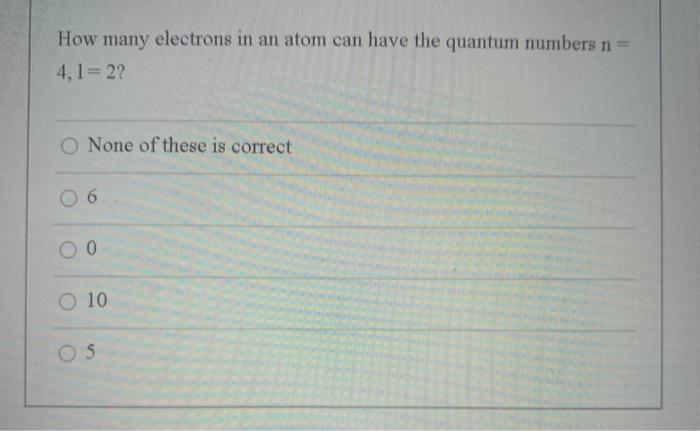
Transcribed Image Text:
How many electrons in an atom can have the quantum numbers n= 4,1 2? O None of these is correct 0 6 O 10 O 5 How many electrons in an atom can have the quantum numbers n= 4,1 2? O None of these is correct 0 6 O 10 O 5
Expert Answer:
Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
How many electrons in an atom can have the designation 1p, 6dx2-y2, 4f, 7py, 2s, and n = 3?
-
In general, an atom can have both orbital angular momentum and spin angular momentum. The total angular momentum is defined to be J(vector) = L(vector) + S(vector). The total angular momentum is...
-
Six observed hadrons have the quantum numbers (Q, B, S, C, B) = (2, 1, 0, 1, 0), (0, 1,2, 1, 0), (0, 0, 1, 0,1), (0,1, 1, 0, 0), (0, 1,1, 1, 0) and (1, 1,3, 0, 0), respectively. Identify their quark...
-
11. Identify the location of oxidation in an electrochemical cell. A) the salt bridge B) the socket C) the cathode D) the electrode E) the anode 12. Determine the cell notation for the redox reaction...
-
Titanium is stiffer than aluminum, has a lower thermal expansion coefficient than aluminum, and has a higher melting temperature than aluminum. On the same graph, carefully and schematically draw the...
-
Let gGDPt denote the annual percentage change in gross domestic product and let intt denote a short term interest rate. Suppose that gGDPt is related to interest rates by gGDPt = a0 + (0intt + (1...
-
How does multiplicity relate to business rules? Give an example.
-
1. As stated in the case, The New York Times chose to deploy their innovation support group as a shared service across business units. What do you think this means? What are the advantages of...
-
Sandhill Inc.'s adjusted trial balance at May 31, 2025 is shown here. Compute the retained earnings balance as of May 31. Sandhill Inc. reported net income of $10,600 during May. Account Titles Debit...
-
1. What might Lewis have done to avoid the result in this case? Discuss. 2. Suppose that Lewis had already been reimbursed for the two vehicles that had been sold, and Whitney sought to obtain the...
-
Shawn Mendez had earned income for the first time in 2019. At the end of the year, his unused RRSP deduction limit was $12,000. For the 2020 tax year, his new contribution room was $11,250. Shawn is...
-
An asset was purchased 7 years ago for $10000. It was estimated to have a 10 year service life and a salvage value of $2000 at the end of its service life. If the value of the asset is believed to...
-
identify which priorities aren't delivering value for CANNTRUST operation and what performance gaps exist in then please use a polar diagram and demonstrate with polar diagram. demonstrate the answer...
-
How challenging is the IRB approval process?How many times can you submit to the IRB for approval?Since my research does involve human subjects, what is meant by an equitable selection process?The...
-
Question 1 Consider the "Analytics Exercise: Forecasting Supply Chain Demand-Starbucks Corporation" at the end of Chapter18 in your course textbook. Open a blank Excel workbook, and rename the second...
-
Adam and Liz are married and have two children who are financially independent: Robert, age 32, and Clare, age 28. Robert is married and has one child and Clare is going to get married. Both Robert...
-
8. Determine the magnetic force on a 0.35 m segment of a wire carrying a current of 12 A in a 0.92 T magnetic field, as illustrated in the diagram. In which direction is the magnetic force on the...
-
The swap spread is the difference between the swap rate and the equivalent-maturity Treasury bond yield. Explain why a widening swap spread may be a signal of deteriorating economic conditions. Plot...
-
Draw all the structural and geometric (cis-trans) isomers of C4H7F.
-
Consider 1.0 mole of a monatomic ideal gas in a container fitted with a piston. The initial conditions are 5.0 L and P = 5.0 atm at some constant T. a. If the external pressure is suddenly changed to...
-
Which of the following statements is(are) true? For the false statements, correct them. a. At constant temperature, the lighter the gas molecules, the faster the average velocity of the gas...
-
For coordinates \(\left(x^{1}, x^{2} ight)\) and metric \(g=\operatorname{diag}\left(g_{11}, g_{22} ight)\), the Gaussian curvature is For a sphere with coordinates defined in the following figure,...
-
Consider the holonomic basis defined in Box 26.1 . Using that the tangent vector for a curve can be written \(t=t^{\mu} e_{\mu}=\left(d x^{\mu} / d \lambda ight) e_{\mu}\), show that Thus, \(g_{\mu...
-
The Lie bracket of vector fields \(A\) and \(B\) is defined as their commutator, \([A, B]=\) \(A B-B A\). The Lie bracket of two basis vectors vanishes for a coordinate basis but not for a...

Study smarter with the SolutionInn App



