How many kilograms of wheat costing Rs. 18 per kg must be mixed with 54 kg...
Fantastic news! We've Found the answer you've been seeking!
Question:
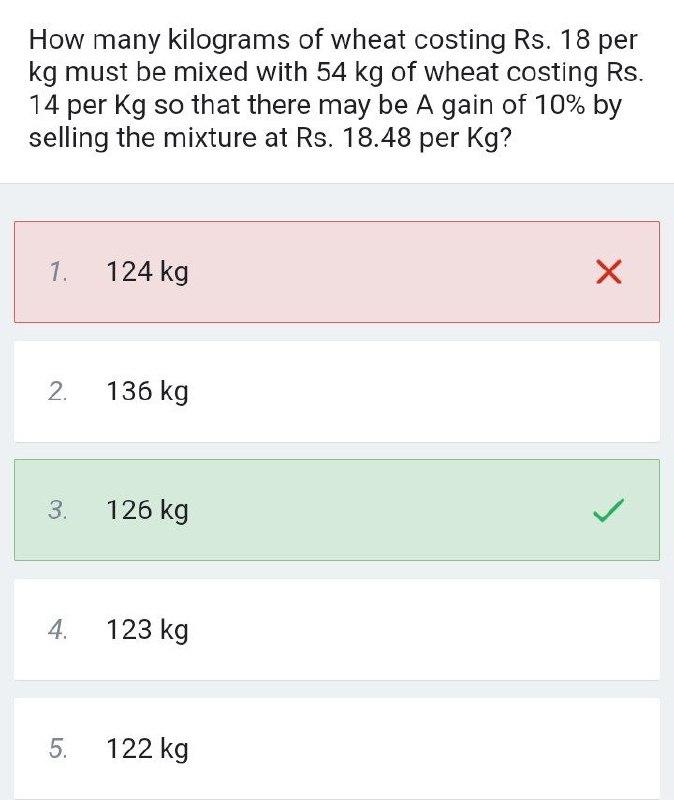
Transcribed Image Text:
How many kilograms of wheat costing Rs. 18 per kg must be mixed with 54 kg of wheat costing Rs. 14 per Kg so that there may be A gain of 10% by selling the mixture at Rs. 18.48 per Kg? 1. 124 kg 2. 136 kg 3. 126 kg 4. 123 kg 122 kg 5. How many kilograms of wheat costing Rs. 18 per kg must be mixed with 54 kg of wheat costing Rs. 14 per Kg so that there may be A gain of 10% by selling the mixture at Rs. 18.48 per Kg? 1. 124 kg 2. 136 kg 3. 126 kg 4. 123 kg 122 kg 5.
Expert Answer:
Answer rating: 100% (QA)
Solution 3 Type I wheat Rs 18kg Type II wheat Rs 14kg 3D Qu... View the full answer

Related Book For 

Posted Date:
Students also viewed these mathematics questions
-
How many kilograms of water must be processed to obtain 2.0 L of D2 at 25C and 0.90 atm pressure? Assume that deuterium abundance is 0.015 percent and that recovery is 80 percent.
-
How many kilograms of nickel must be added to 1.75 kg of copper to yield a liquids temperature of 1300(C?
-
How many kilograms of nickel must be added to 5.43 kg of copper to yield a solidus temperature of 1200(C?
-
On a cold winter day, the atmospheric temperature is - (on Celsius scale) which is below 0C. A cylindrical drum of height h made of a bad conductor is completely filled with water at 0C and is kept...
-
When should you not apologize?
-
Draw a PPF that represents the production possibilities for goods X and Y if there are constant opportunity costs. Next, represent an advance in technology that makes it possible to produce more of...
-
Repeat Example 15-10 but with a mass transfer coefficient that is 10 times larger (use \(\delta=0.001 \mathrm{~m})\). Report \(\mathrm{x}_{\mathrm{NH} 3}, \mathrm{y}_{\mathrm{NH} 3, \text { surface...
-
The Houston Corp needs to raise money for an addition to its plant. It will issue 300,000 shares of new common stock. The new shares will be priced at $60 per share with an 8.5% spread on the offer...
-
Use the calculators at Dinkytown.net to answer the following questions:a . You need $ 5 , 0 0 0 for a trip to Europe in two years. How much would you have to deposit monthly in a savings account...
-
Figure 2 shows the support structure for a suspended apparatus in a laboratory. The apparatus is hung from the 1/4" A36 plate that is welded to the 3/8" plate using E70XX electrodes. Assuming that...
-
At 0C 20g of ice are added to 50g of water at 30C in a vessel that has a water equivalent of 20g. Calculate the entropy changes in the systems and in the surroundings. The heat of fusion of ice at 0C...
-
Compute break - even units using the following formula: Break - Even Units = Total Fixed Costs / ( Unit Selling Price Unit Variable Cost ) suppose that a firm has an existing product with a combined...
-
4. Solve for n if 2(nC) = n+1C3-
-
Novak Company's standard materials cost per unit of output is $10 (2 kg x $5). During July, the company purchases and uses 3,300 kg of materials costing $16,929 in making 1,500 units of finished...
-
Suppose f(x)=2x 3x+5 and g(x) = x+3x-4. (f-9)(x)
-
Company ABC is a manufacturing company of plastic components that go into other organizations finished products. They have international operations. In the last 24 months the organization has had a...
-
Create an activation record instance for the C skeletal function shown below by selecting appropriate contents.
-
In the series connection below, what are the respective power consumptions of R, R2, and R3? R R www 4 V=6V P1-3 W; P2=3W; and P3= 3 W OP10.5 W; P2-1 W; and P3= 1.5 W P1=1.5 W; P2=1 W; and P3= 0.5 W...
-
Which of the following reagents can oxidize H2O to O2(g) under standard-state conditions? H+(aq),Cl-(aq), Cl2(g), Cu2+(aq), Pb2+(aq), MnO4- (aq) (in acid).
-
Which of the following metals can react with water? (a) Au, (b) Li, (c) Hg, (d) Ca, (e) Pt.
-
Consider the reaction Comment on the changes in the concentrations of SO2, O2, and SO3 at equilibrium if we were to (a) Increase the temperature; (b) Increase the pressure; (c) Increase SO2; (d) Add...
-
The Oasis Hotel is planning its cash payments for operations for the fourth quarter (October-December), 2003. The Accrued Expenses Payable balance on October 1 is \($136,000.\) The budgeted expenses...
-
Rough Trails Bicycle Company manufactures mountain bikes. The following data for March of the current year are available: Determine the direct labor rate and time variances.. Quantity of direct labor...
-
Curtis Container Company (CCC) manufactures plastic 2-liter bottles for the beverage industry. The cost standards per 100 2-liter bottles are as follows: At the beginning of May, CCC management...

Study smarter with the SolutionInn App


