HSO3 is a strong acid weak acid strong base weak base neither acid nor base QUESTION...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
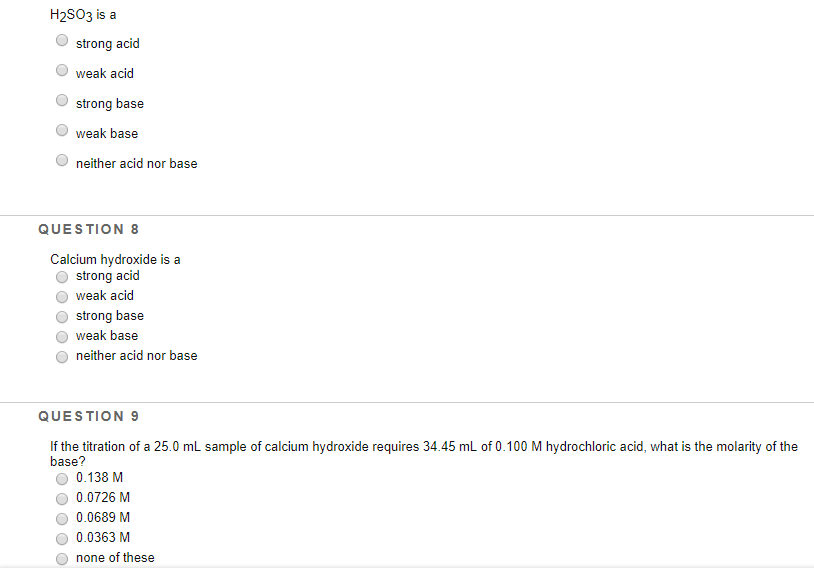
H₂SO3 is a strong acid weak acid strong base weak base neither acid nor base QUESTION 8 Calcium hydroxide is a strong acid weak acid strong base weak base neither acid nor base QUESTION 9 If the titration of a 25.0 mL sample of calcium hydroxide requires 34.45 mL of 0.100 M hydrochloric acid, what is the molarity of the base? 0.138 M 0.0726 M ● 0.0689 M O 0.0363 M none of these H₂SO3 is a strong acid weak acid strong base weak base neither acid nor base QUESTION 8 Calcium hydroxide is a strong acid weak acid strong base weak base neither acid nor base QUESTION 9 If the titration of a 25.0 mL sample of calcium hydroxide requires 34.45 mL of 0.100 M hydrochloric acid, what is the molarity of the base? 0.138 M 0.0726 M ● 0.0689 M O 0.0363 M none of these
Expert Answer:
Answer rating: 100% (QA)
Answers Question 7 HSO3 is a strong acid This is because it dissociates Fully in water Question ... View the full answer

Related Book For 

Cambridge International AS And A Level Chemistry Coursebook
ISBN: 9781316637739
2nd Edition
Authors: Lawrie Ryan, Roger Norris
Posted Date:
Students also viewed these chemistry questions
-
HCl (hydrochloric acid) is a strong acid and CH3COOH (aceticacid) is a weak acid. If you mix 1.00 L of 0.0550 M HCl (aq) with1.00 L of 12.0 M CH3COOH (aq), what will be the final concentrationof [H+...
-
The figure compares the titration of a monoprotic weak acid with a monoprotic weak base and the titration of a diprotic acid with strong base. (a) Write the reaction between the weak acid and the...
-
A quantity of 0.15 M hydrochloric acid is added to a solution containing 0.10 mol of sodium acetate. Some of the sodium acetate is converted to acetic acid, resulting in a final volume of 650 mL of...
-
As a security architect for a medium-sized firm, briefly describe how would you approach the following case problems: 1. Sending encrypted email to third parties. 2. Encrypting sensitive data inside...
-
Taveras Co. decides at the beginning of 2008 to adopt the FIFO method of inventory valuation. Taveras had used the LIFO method for financial reporting since its inception on January 1, 2006, and had...
-
Are all intangibles reported in a single line on the balance sheet? Explain.
-
Question: Dr. James Leonard wrote Dr. Edward Jacobson offering him the position of Chief of Audiology at Jefferson Medical College in Philadelphia. In the letter, Leonard stated that this appointment...
-
Presented below is an excerpt from Starbucks Note 1, Summary of Significant Accounting Policies, in its September 28, 2008 Annual Report. a. The above passages indicate that Starbucks generates...
-
1) List three ways in which reserve information can affect the financial statements. 2)List the costs that are treated the same under successful efforts and full cost accounting. List the costs that...
-
A beam of T section is supported and loaded as shown. a) (15 pts) Construct the shear and bending moment diagram, provide expressions for the shear and bending moment on each segment, provide graphs,...
-
Ques 2.Tierney Company begins operations on April 1. Information from job cost sheets shows the following. Manufacturing Costs Assigned Job Month Number April May June Completed 10 $5,200 $4,400 May...
-
Suppose a thief stole a car that he can resell for $10,000. The thief spent $100 on tools in preparation for his act. During the day the thief works as a laborer and earns $20,000/year. The thief...
-
Catskills Equipment guarantees its snowmobiles for three years. Company experience indicates that warranty costs will be approximately 3% of sales. Assume that the Catskills Equipment dealer in...
-
A brief analysis of the drivers impacting oil prices, to include, fast-growing demand due to high global economic growth, declining supply or anticipated shortages in supply, coordinated action on...
-
Name three benefits for consumers of locally grown food that are most important to your household. Briefly discuss three benefits of locally grown food to the U.S. economy ? What are three...
-
Why do individuals or entities choose to invest in the capital stock of corporations? 2.When is a misstatement considered fraud? 3.What type of assurance does an audit provide? 4.What is materiality?...
-
Heat at a rate of 70 kW is transferred to a thermal energy reservoir at 25C. What is the entropy change of the reservoir during this process? -70/25 kW/K -70/298 kW/K 70/298 kW/K 70105F 1JNIK
-
From the choice of simple moving average, exponential smoothing, and linear regression analysis, which forecasting technique would you consider the most accurate? Why? please write it in word...
-
Copy and complete the table below to compare the properties of giant ionic, giant molecular, giant metallic and simple molecular structures. Giant Giant ionic molecular Metallic Simple molecular Two...
-
a. Write half-equations for the three reactions taking place in the half-cells shown on the left in Figure 20.10. b. What are the standard electrode potentials for these half-cell reactions? c. List...
-
a. How does a non-solvent-based adhesive, such as an SMP, set to become a solid? b. What type of monomers react together to form an epoxy resin? c. i. Draw the displayed formula of the monomer used...
-
Which statement is false? a. In terms of actual numbers of events, women commit more fraud than men. b. Fraudsters act alone about 70 percent of the time. c. Employees are the largest number of...
-
Which statement is false? a. Women are more likely than men to report fraudulent activity. b. Older employees are more likely to report fraudulent activities than younger employees. c. For each \(\$...
-
What would be the worst strategy to catch ghost employees? a. Insure segregation of payroll preparation, disbursement, and distribution functions. b. Check for inflated invoices. c. Examine payroll...

Study smarter with the SolutionInn App


