(i) Calculate the double bond equivalents (DBE) for each molecule. (ii) Assign the 'H NMR spectra...
Fantastic news! We've Found the answer you've been seeking!
Question:

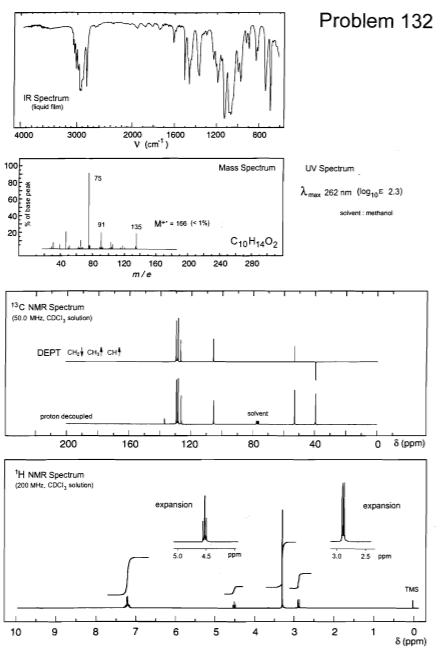
Transcribed Image Text:
(i) Calculate the double bond equivalents (DBE) for each molecule. (ii) Assign the 'H NMR spectra completely, rationalise the number of peaks, relative integral, their multiplicity or splitting pattern where appropriate. (iii) Rationalise the 13C NMR spectra. (iv) Rationalise all numbered peaks in the mass spectra and IR spectra. (v) Provide a brief logical explanation for how you arrived at unambiguous structures for compounds A and B. Problem 132 IR Spectrum (iquid fim) 2000 1600 V (cm") 4000 3000 1200 800 100 Mass Spectrum UV Spectrum 75 80 Ame 262 nm (log,E 23) scivent methanal 91 M- 100 < 1% 135 40 80 120 160 200 240 280 m/e 13C NMR Spectrum (50.0 MHz, CDCI, souson) DEPT CH Cnt cHt solvent proton decouplied 200 160 120 80 40 8 (ppm) TH NMR Spectrum (200 Mitz. CDCI, solution expansion expansion 5.0 45 ppm 10 25 ppm TMS 10 6 3 2 8 (ppm) (i) Calculate the double bond equivalents (DBE) for each molecule. (ii) Assign the 'H NMR spectra completely, rationalise the number of peaks, relative integral, their multiplicity or splitting pattern where appropriate. (iii) Rationalise the 13C NMR spectra. (iv) Rationalise all numbered peaks in the mass spectra and IR spectra. (v) Provide a brief logical explanation for how you arrived at unambiguous structures for compounds A and B. Problem 132 IR Spectrum (iquid fim) 2000 1600 V (cm") 4000 3000 1200 800 100 Mass Spectrum UV Spectrum 75 80 Ame 262 nm (log,E 23) scivent methanal 91 M- 100 < 1% 135 40 80 120 160 200 240 280 m/e 13C NMR Spectrum (50.0 MHz, CDCI, souson) DEPT CH Cnt cHt solvent proton decouplied 200 160 120 80 40 8 (ppm) TH NMR Spectrum (200 Mitz. CDCI, solution expansion expansion 5.0 45 ppm 10 25 ppm TMS 10 6 3 2 8 (ppm)
Expert Answer:

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
(i) Calculate the double bond equivalents (DBE) for each molecule. (ii) Assign the 1H NMR spectra completely, rationalise the number of peaks, relative integral, their multiplicity or splitting...
-
(i) Calculate the double bond equivalents (DBE) for each molecule. (ii) Assign the 'H NMR spectra completely, rationalise the number of peaks, relative integral, their multiplicity or splitting...
-
The IR spectrum, 13C NMR spectrum, and 1HNMR spectrum of an unknown compound (C6H8O3) appear next. Determine the structure, and show how it is consistent with the spectra. wavelength (um) 5 5.5 6 710...
-
On February 1, 2020, Sheridan Company sells merchandise on account to Carla Vista Company for $6490. The entry to record this transaction by Sheridan Company is Sales Revenue Accounts Payable Notes...
-
One big box store sells a Toshiba notebook computer for $549.99. Each computer has a 57% markup based on selling price. What is the markup and cost of each computer?
-
Give four examples of reactions of C60 that are consistent with the presence of C = C bond character.
-
What is the purpose of the materials requisition?
-
Representatives of the Franklin Insurance Company take medical information over the phone from prospective policy applicants prior to a visit to the applicant's place of residence by a registered...
-
Complete this question by entering your answers in the tabs below. Req A and B Req C Req D Req E to H Req I Req J a. Calculate return on investment, based on net income and average total assets, for...
-
You are the brand manager for your favorite brand of clothing, food, vehicle, or other consumer product. Write a one-page branding statement summarizing your brand for your company's VP of Marketing....
-
Logan is a new hire as a transportation analyst. One of his carriers that provides service for the shipping lane from Philadelphia to Indianapolis asks Logan would it be possible to receive a...
-
At a local university, the academic dean has informed the chairperson of the economics department that she must schedule 8 different graduate classes and 18 different undergraduate classes....
-
tions Places System Qets Pet Your Motor 10-11 dose per incident fluence/Gy em 3e-11e 2.5e-11 2e-11- 1.5e-11 le-11 5e-12 Wed Sep 20 19:58:05 2023 5 Compare your 10 MeV dose profiles with and without...
-
Windows is a small company that installs windows. Its cost structure is asfollows: Selling price from each window installation $900 Variable cost of each window installation $850 Annual fixed costs...
-
After introducing and briefly describing the drivers of excess inventory, discuss the implications of inventory volatility (i.e. periods of very low or zero stock followed by periods of excess stock)...
-
Compare Blessing with Island Man showing how the poets use description to convey the thoughts and feelings of the characters in the poems. Blessing and Island Man are two very different poems. The...
-
Fanning Entertainment sponsors rock concerts. The company is considering a contract to hire a band at a cost of $90,000 per concert. Required What are the total band cost and the cost per person if...
-
Three forces with magnitudes of 70pounds, 40 pounds, and 60 pounds act on an object at angles of 30, 45, and 135, respectively, with the positive x-axis. Find the direction and magnitude of the...
-
Which carbocation in each of the following pairs is more stable? a. b. CH3CH2 or H2C CH H2C= CH or HC- C
-
Give the products of the following reactions: (a) (b) (c) (d) CCH,CH,CHC.ACl 2. H20 CCHCH2C .AICI 2. H20 CH2CH2 CC .AICl3 CH CH2CH,C .AICI3 2. H20
-
Mark off the isoprene units in lycopene and -carotene. Can you detect a similarity in the way in which squalene, lycopene, and -carotene are biosynthesized?
-
The Mann Corporation began operations in 2015. Information relating to the companys purchases of inventory and sales of products for 2015 and 2016 is presented below. Calculate the LIFO cost of goods...
-
The Mann Corporation began operations in 2015.Information relating to the companys purchases of inventory and sales of products for 2015 and 2016 is presented below. Calculate the weighted-average...
-
Presented below is financial data for two companies that are identical yma in every respect except that Company X uses the FIFO method to value its inventory and Company Z uses the LIFO method to...

Study smarter with the SolutionInn App


