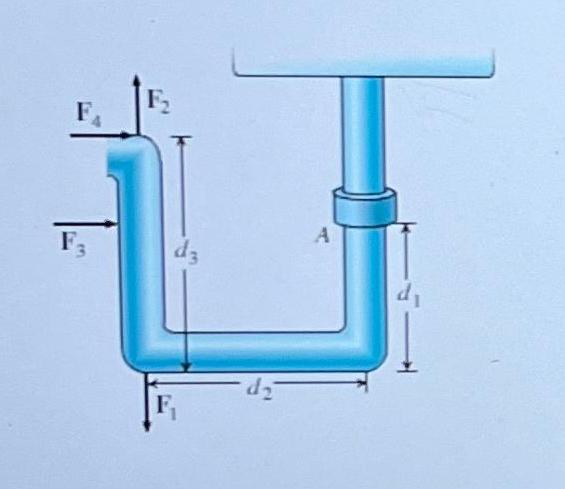
As shown, a pipe is anchored to a wall at point A. During the pipe's installation,...
Fantastic news! We've Found the answer you've been seeking!
Question:

Transcribed Image Text:
As shown, a pipe is anchored to a wall at point A. During the pipe's installation, several forces are applied to the pipe at different locations. If F₁ = 76.9 N, F2 = 76.2 N, F3 = 74.0 N, F4 = 70.5 N, d₁ = 0.150 m, d2 = 0.220 m, and d3 = 0.200 m, what is MR, the net moment about point A due to these forces? Assume that moments acting counterclockwise about point A are positive whereas moments acting clockwise are negative. Express your answer numerically in Newton-meters to three significant figures. FA F3 F. A d3 d2 As shown, a pipe is anchored to a wall at point A. During the pipe's installation, several forces are applied to the pipe at different locations. If F₁ = 76.9 N, F2 = 76.2 N, F3 = 74.0 N, F4 = 70.5 N, d₁ = 0.150 m, d2 = 0.220 m, and d3 = 0.200 m, what is MR, the net moment about point A due to these forces? Assume that moments acting counterclockwise about point A are positive whereas moments acting clockwise are negative. Express your answer numerically in Newton-meters to three significant figures. FA F3 F. A d3 d2
Expert Answer:
Answer rating: 100% (QA)
Determining the Net Moment MRA at Point A Based on the information provided and the image we can cal... View the full answer

Related Book For 

Posted Date:
Students also viewed these finance questions
-
Does Money market instrument meets short term requirements of the borrowers and provides liquidity to the lenders. Why or why not ?Explain
-
The following additional information is available for the Dr. Ivan and Irene Incisor family from Chapters 1-5. Ivan's grandfather died and left a portfolio of municipal bonds. In 2012, they pay Ivan...
-
Planning is one of the most important management functions in any business. A front office managers first step in planning should involve determine the departments goals. Planning also includes...
-
On the income statement for the year ending December 31, Y1, the accountant for ABC calculated operating income before taxes of $300,000. This $300,000 did not include the effect of any of the...
-
Under Armour has the following current assets: cash, $102 million; receivables, $94 million; inventory, $182 million; and other current assets, $18 million. Under Armour has the following...
-
Explain how exchange rate fluctuations pose a risk to manufacturing companies who rely upon an export strategy to compete in foreign markets.
-
In February 2014, defendant Ibrahim M. Shihadeh, d/b/a Creative Designs Kitchen and Baths, agreed to purchase 25% of his anticipated natural gas needs at a fixed price for the 201415 and 201516...
-
The United Mutual and Accident Insurance Company has a large pool of clerical employees who process insurance application forms on networked computers. When the company hires a new clerical employee,...
-
Part A The device shown in (Figure 1) is designed to exert a large force on the stationary object at the left. The jack exerts a 225-lb upward force at B. As a result, the link BC exerts a force on...
-
Brian and Corrine Lee are married taxpayers filing jointly. They live in the home they own, located at 3301 Pacific Coast Hwy., Laguna Beach, CA 92651. Brian is an optometrist who owns his business;...
-
Calculate the under-allocated or over-allocated manufacturing overhead for each of the following two departments and for the Square at Mindanao plant as a whole on the basis of information below...
-
(2) - Agt! Predict products and write balanced FUE and NIE equations for the following precipitation reactions. i) silver(I) nitrate with sodium nitrate Ag No3 + Na No3 - Ag No3 + NaNO3 No3 Not, Nos...
-
Agriculture accounted for 3/8 of greenhouse gas emissions in 1980 and 2/11 in 2000. What's the relative change in greenhouse emissions from agriculture between 1980 and 2000? 20.1% 19.3% 51.5% 48.3%...
-
The type of reaction accompanied by release of energy is called O exothermic. O extrothermic. O introthermic. O endothermic
-
Calcium carbonate decomposes at high temperatures to form carbon dioxide and calcium oxide: 2 CaCO3(s) CO2(g) + CaO(s) How many grams of calcium carbonate will I need to form 3.45 liters of carbon...
-
Currently, the primary source of sulfur dioxide emissions into the atmosphere is a . diesel trucks. b . plastic manufacturing. c . coal burning power plants. d . gasoline - powered lawnmowers.
-
Describe the steps and reaction mechanisms to obtain product C, from reagents A and B, adding the reagents that you consider pertinent. NH
-
Uniform electric field in Figure a uniform electric field is directed out of the page within a circular region of radius R = 3.00 cm. The magnitude of the electric field is given by E = (4.50 x 10-3...
-
Pick any pair of rivals (such as Boeing and Airbus, Apple and Samsung, Toyota and Volkswagen) and explain why one outperforms another.
-
Barny Kpf had just returned from Spoga, the international trade fair for outdoor equipment and furniture held in Germany. Despite the global economic crisis, the results achieved at the fair had been...
-
If you were a CEO or a business unit head, under what conditions would you consider moving your headquarters overseas?
-
A company issues $30,000,000 face value of fi ve-year bonds dated January 1, 2015 when the market interest rate on bonds of comparable risk and terms is 5%. Th e bonds pay 4% interest annually on...
-
For a bond issued at a premium, using the eff ective interest rate method, the: A . carrying amount increases each year. B . amortization of the premium increases each year. C . premium is evenly...
-
Which of the following is an example of an affi rmative debt covenant? Th e borrower is: A . prohibited from entering into mergers. B . prevented from issuing excessive additional debt. C . required...

Study smarter with the SolutionInn App


