Imagine an electron in the 5f orbital of a neutral atom. Decide how well each of...
Fantastic news! We've Found the answer you've been seeking!
Question:
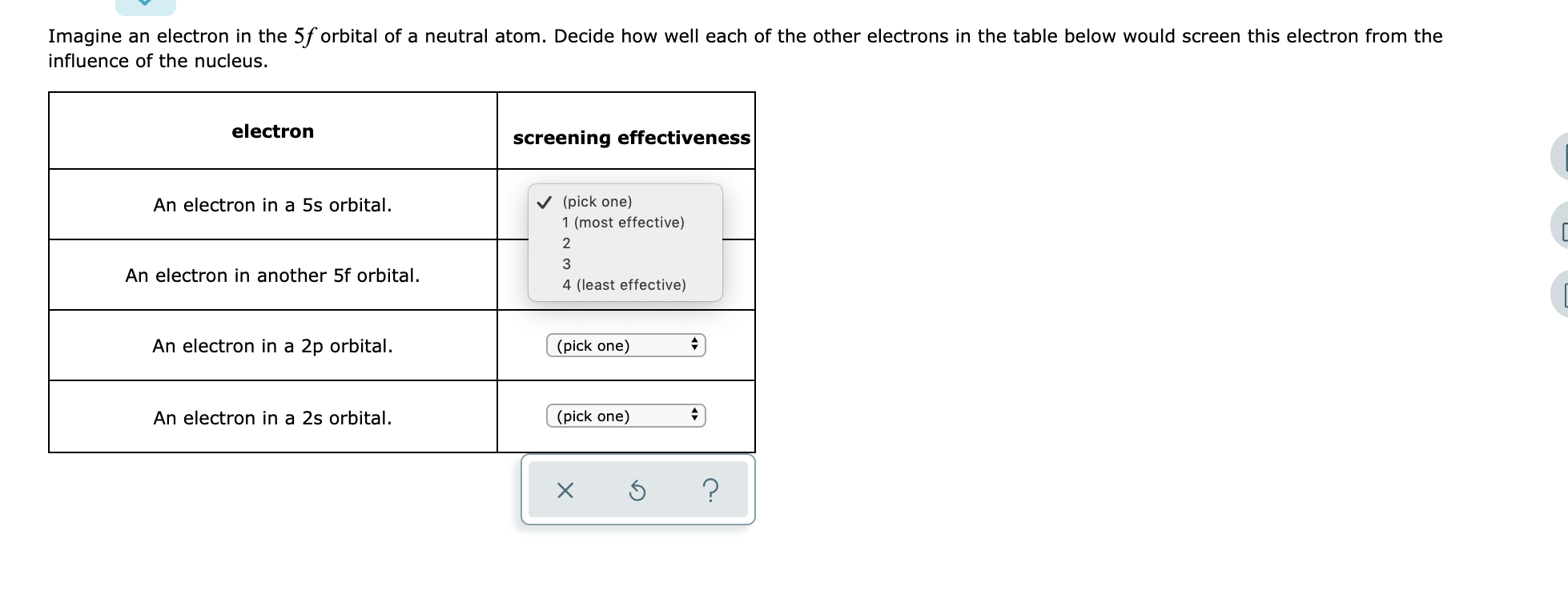
Transcribed Image Text:
Imagine an electron in the 5f orbital of a neutral atom. Decide how well each of the other electrons in the table below would screen this electron from the influence of the nucleus. electron An electron in a 5s orbital. An electron in another 5f orbital. An electron in a 2p orbital. An electron in a 2s orbital. screening effectiveness ✓ (pick one) 1 (most effective) 2 3 4 (least effective) (pick one) (pick one) X + ♦ ? Imagine an electron in the 5f orbital of a neutral atom. Decide how well each of the other electrons in the table below would screen this electron from the influence of the nucleus. electron An electron in a 5s orbital. An electron in another 5f orbital. An electron in a 2p orbital. An electron in a 2s orbital. screening effectiveness ✓ (pick one) 1 (most effective) 2 3 4 (least effective) (pick one) (pick one) X + ♦ ?
Expert Answer:
Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
The electron configuration of a neutral atom is 1s22s22p63s2. Write a complete set of quantum numbers for each of the electrons. Name the element.
-
An excited hydrogen atom with an electron in the n = 5 state emits light having a frequency of 6.90 1014 s-1. Determine the principal quantum level for the final state in this electronic transition.
-
A hydrogen atom has an electron in the n = 5 level. (a) If the electron returns to the ground state by emitting radiation, what is the minimum number of photons that can be emitted? (b) What is the...
-
Problem 5: Closing entries. (10 points) Given the following accounts, prepare the closing entries in the order and method taught using Income Summary. Number each entry 1, 2, .... Accounts payable $...
-
What are the dangers in evaluating a project based on the reason(s) it was selected (such as its being a competitive necessity), rather than the goals and objectives in the project proposal or...
-
Shadee Corp. expects to sell 600 sun visors in May and 420 in June. Each visor sells for $16. Shadees beginning and ending finished goods inventories for May are 65 and 55 units, respectively. Ending...
-
What huge bankruptcy in 2001-2002 caused forensic accountants to become rising stars within the accounting profession?
-
Fantastic Props, Inc., designs and fabricates movie props such as mock-ups of star-fighters and cybernetic robots. The companys balance sheet as of January 1, the beginning of the current year,...
-
Calculation of individual costs and WACC Dilon Labs has asked its financial manager to measure the cost of each specific type of capital as well as the weighted average cost of capital. The weighted...
-
case study:- Prestige Dance academy please answer the question below. CASE 11 Prestige Dance Academy P icture a group of children pretending to be butterflies twirling and leaping around a calming...
-
KFC, Pizza Hut, and Taco Bell locations are all franchised and operated by Yum China Holdings Inc., which is a corporation that specializes in the operation of fast-food restaurants in China. In...
-
There're a total of 4 questions. Table 9.1 :https://ezt.prod.mheducation.com/Media/Connect_Production/bne/slater_14e/table_9_1.htm 6 10 points eBook det Hint Word Problem 9-21 (Static) [LU 9-2 (1)]...
-
Sales Cost of goods sold Gross margin Selling and administrative expenses Net operating loss $ 1,641,100 1,235,068 406,032 630,000 $ (223,968) Hi-Tek produced and sold 60,100 units of B300 at a price...
-
Suppose I have some function, kwyjibo: private int kwyjibo(int v) { int w = v % 2; if (w = 3) { return 8; } else { return w / 2; } } If I print out the result of calling kwyjibo, passing it some...
-
In a country where beer gardens are a popular part of everyday life, rising beer prices and bottle shortages could threaten normal buyer behavior. Consider the spillover effects of higher beer prices...
-
Reflect on the competition between Germany's breweries. Will smaller breweries be able to survive the current situation, or do you think they will be forced to sell out to larger breweries? If they...
-
1.Working from case Exhibit 9, relative to the stand-alone value, perform an APV analysis to estimate the dollar increase in DPC's value if a PE fund can obtain: a.5% revenue growth per annum (versus...
-
How will relating product contribution margin s to the amount of the constrained resource they consume help a company maximize its profits?
-
Although the VSEPR model is correct in predicting that CH4 is tetrahedral, NH3 is pyramidal, and H2O is bent, the model in its simplest form does not account for the fact that these molecules do not...
-
The following are representations of acid base reactions: a. Label each of the species in both equations as an acid or a base and explain. b. For those species that are acids, which labels apply:...
-
A sample of chloroform is found to contain 12.0 g of carbon, 106.4 g of chlorine, and 1.01 g of hydrogen. If a second sample of chloroform is found to contain 30.0 g of carbon, what is the total mass...
-
What is not a proactive approach to forensic accounting a. Investigating complaints b. Effective internal controls c. Logging of exceptions d. Reviewing variances e. Financial audits
-
\( \qquad \) refers to the basis for undertaking a fraud investigation. a. Alter ego b. Predication c. Wildcatting d. Invigilation e. Shenanigan
-
____________ _________ shows associations between people and data. a. Link analysis b. Game theory c. Timeline analysis d. Tracing theory e. Invigilation analysis

Study smarter with the SolutionInn App



