In nitration of acetophenone, an Electrophilic Aromatic Substitution reaction, shown below, m-nitroacetophenone appears to be the...
Fantastic news! We've Found the answer you've been seeking!
Question:
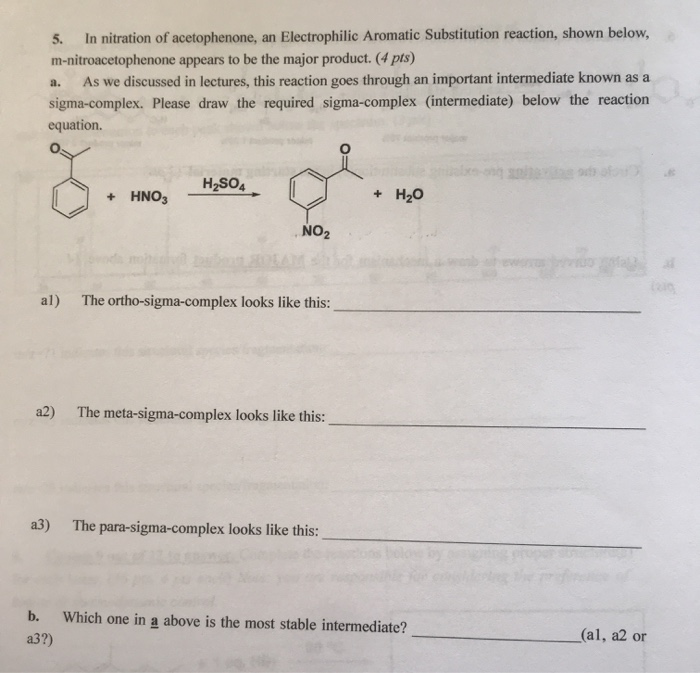
Transcribed Image Text:
In nitration of acetophenone, an Electrophilic Aromatic Substitution reaction, shown below, m-nitroacetophenone appears to be the major product. (4 pts) a. As we discussed in lectures, this reaction goes through an important intermediate known as a sigma-complex. Please draw the required sigma-complex (intermediate) below the reaction equation. 5. + HNO3 H₂SO4 NO₂ al) The ortho-sigma-complex looks like this: a2) The meta-sigma-complex looks like this: a3) The para-sigma-complex looks like this: + H₂O b. Which one in a above is the most stable intermediate? a3?) (al, a2 or (210 In nitration of acetophenone, an Electrophilic Aromatic Substitution reaction, shown below, m-nitroacetophenone appears to be the major product. (4 pts) a. As we discussed in lectures, this reaction goes through an important intermediate known as a sigma-complex. Please draw the required sigma-complex (intermediate) below the reaction equation. 5. + HNO3 H₂SO4 NO₂ al) The ortho-sigma-complex looks like this: a2) The meta-sigma-complex looks like this: a3) The para-sigma-complex looks like this: + H₂O b. Which one in a above is the most stable intermediate? a3?) (al, a2 or (210
Expert Answer:
Answer rating: 100% (QA)
Ans5 a HNO3 HSO4 HNO3 HSO Ortho sigma complex a 10 NO NO... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
Which compound will undergo an electrophilic aromatic substitution reaction more rapidly, benzene or hexadeuteriobenzene? or D- H.
-
Which of these media executions appears to be the most innovative?
-
Which of these ideas appears to be the most creative use of new technology?
-
Common structural steel can be represented on drawings using a letter, immediately followed by a number, then another number (for example, S14x30). The two numbers for a steel member W16x60 designate...
-
Determine how the accounts would be affected (increase or decrease and debit or credit) for the following transactions occurring in April 2011 for Computer Solutions, Inc.: 1. The company paid $4,500...
-
Refer to TMCs project selection problem presented in this chapter. In the solution shown in Figure, The probability of success for project 4 is only 0.3488. Thus, project 4 is almost twice as likely...
-
Bert C. Roberts Jr. was chairman of WorldComs board of directors. Immediately before that, he had been chairman of MCI, which WorldCom acquired on September 14, 1998, in a transaction valued at...
-
Watchdog Company maintains and repairs warning lights, such as those found on radio towers and lighthouses. Watchdog Company prepared the end-of-period spreadsheet shown below at July 31, 2014, the...
-
Research Web-based database technologies and identify a database management system (other than SQL Server, MySQL, or Oracle) that is used to deploy applications to the Web and the cloud. Discuss the...
-
1. Create and upload a histogram of the salary data for the city of Bell, where each bar width is about 50,000 US dollars. (Data for the histogram is at the bottom). a.) Is the distribution of the...
-
Analysis shows that a metal oxide has the empirical formula of M 0.96 O 1.00 . Calculate the percentage of M 2+ and M 3+ ions in this crystal.
-
How does your leadership, coaching, and conflict management styles (core/adaptive leadership, collaborating conflict management style) work together as you lead and coach others?
-
How does acquisition planning help the COR get the best value for acquisition? It allows the COR to give new work to Contractors with whom they have worked before. It allows the COR to identify the...
-
7. A new composite material has been developed -- iron reinforced PVC (polyvinyl chloride). The following test results were obtained: Tissue Culture Cell growth normal in culture of muscle cells ...
-
Precision Bearings Inc. makes two bearings, B100 and B200, for use in its own production. Data regarding these two bearings follow: B100B200Machine hours required per unit2.53.0Cost per unit: Direct...
-
Human tissue. Who owns bone, sinew, corneas, and other saleable body parts? Who can consent to its post-mortem removal? What do state statutes say about the nonconsensual removal of human tissue? Why...
-
Partial derivatives (a) For the functions below, evaluate all first and second derivatives, and simplify as much as possible. m (7/2+1), (x, y, 2) = V/2 ii. f(z,g)=sinh(ry), f(2,3)=tan'(ry),...
-
(a) As Section 17.3 discusses, high-frequency sound waves exhibit less diffraction than low-frequency sound waves do. However, even high-frequency sound waves exhibit much more diffraction under...
-
For each of the following reactions, (1) indicate whether elimination will occur via an E2 or an E1 reaction, and (2) give the major elimination product of each reaction, ignoring stereoisomers: a....
-
The rate of the reaction of 1-bromo-2-butene with ethanol is increased if silver nitrate is added to the reaction mixture. Explain.
-
The 1H NMR spectra of three isomers with molecular formula C7H14O are shown here. Which isomer produces which spectrum? a. b. c. QUESTION CONTINUE TO NEXT PAGE 0 (ppm) frequency 6 (ppm) frequency 8...
-
Company data for dividend per share (DPS), earnings per share (EPS), share price, and price-to-earnings ratio (P/E) for the most recent five years are presented in Exhibit 10-9. In addition,...
-
The best model to use when valuing a young dividend-paying company that is just entering the growth phase is most likely the: A. Gordon growth model. B. Two-stage dividend discount model. C....
-
For the next three years, the annual dividends of a stock are expected to be 2.00, 2.10, and 2.20. The stock price is expected to be h20.00 at the end of three years. If the required rate of return...

Study smarter with the SolutionInn App


