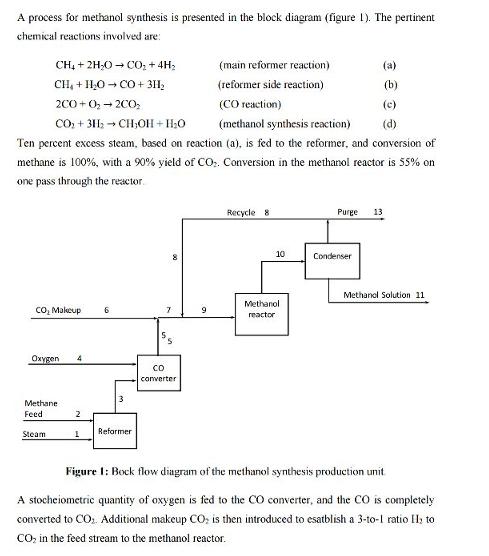
A process for methanol synthesis is presented in the block diagram (figure 1). The pertinent chemical...
Fantastic news! We've Found the answer you've been seeking!
Question:

Transcribed Image Text:
A process for methanol synthesis is presented in the block diagram (figure 1). The pertinent chemical reactions involved are: CH₂ + 2H₂O → CO₂ + 4H₂ CH₂ + H₂O →CO + 3H₂ 2C0+0₂ - 200₂ CO₂ + 3H₂CH₂OH+H₂O (methanol synthesis reaction) (d) Ten percent excess steam, based on reaction (a), is fed to the reformer, and conversion of methane is 100%, with a 90% yield of CO₂. Conversion in the methanol reactor is 55% on one pass through the reactor. CO₂ Makeup 6 Oxygen 4 Methane Feed Steam 2 1 3 Reformen CO converter (main reformer reaction) (reformer side reaction) (CO reaction) 9 Recycle 81 10 Methanol reactor (a) (b) (c) Purge 13 Condenser Methanol Solution 11 Figure 1: Bock flow diagram of the methanol synthesis production unit A stocheiometric quantity of oxygen is fed to the CO converter, and the CO is completely converted to CO₂. Additional makeup CO₂ is then introduced to esatblish a 3-to-1 ratio 1₂ to CO₂ in the feed stream to the methanol reactor. 2| Page The methanol reactor effluent is cooled to condense all the methanol and water, with the noncondensible gases recycled to the methanol reactor feed. The H/CO₂ ratio in the recylee stream is also 3-to-1. Because the methane feed contains 1% nitrogen as an impurity, a portion of the receycle stream must be purged as shown in the figure I to prevent the accumulation of nitrogen in the system. The purge stream analyzes 5% nitrogen. 1) Sketch the PFD f the process and label all the streams, blocks etc... according to the guidelines presented in "Lecture_02_bonus_GCH350", 2) On the basis of 100 mol/h of methane feed (inculding the N₂), calculate: a. How many moles of H₂ are lost in the purge. b. How many moles of makeup CO₂ are required. c. The recycle to purge ratio in mol/mol. d. How much methanol solution (in kg/h) of what strength (weight percent) is produced. 3 | Page A process for methanol synthesis is presented in the block diagram (figure 1). The pertinent chemical reactions involved are: CH₂ + 2H₂O → CO₂ + 4H₂ CH₂ + H₂O →CO + 3H₂ 2C0+0₂ - 200₂ CO₂ + 3H₂CH₂OH+H₂O (methanol synthesis reaction) (d) Ten percent excess steam, based on reaction (a), is fed to the reformer, and conversion of methane is 100%, with a 90% yield of CO₂. Conversion in the methanol reactor is 55% on one pass through the reactor. CO₂ Makeup 6 Oxygen 4 Methane Feed Steam 2 1 3 Reformen CO converter (main reformer reaction) (reformer side reaction) (CO reaction) 9 Recycle 81 10 Methanol reactor (a) (b) (c) Purge 13 Condenser Methanol Solution 11 Figure 1: Bock flow diagram of the methanol synthesis production unit A stocheiometric quantity of oxygen is fed to the CO converter, and the CO is completely converted to CO₂. Additional makeup CO₂ is then introduced to esatblish a 3-to-1 ratio 1₂ to CO₂ in the feed stream to the methanol reactor. 2| Page The methanol reactor effluent is cooled to condense all the methanol and water, with the noncondensible gases recycled to the methanol reactor feed. The H/CO₂ ratio in the recylee stream is also 3-to-1. Because the methane feed contains 1% nitrogen as an impurity, a portion of the receycle stream must be purged as shown in the figure I to prevent the accumulation of nitrogen in the system. The purge stream analyzes 5% nitrogen. 1) Sketch the PFD f the process and label all the streams, blocks etc... according to the guidelines presented in "Lecture_02_bonus_GCH350", 2) On the basis of 100 mol/h of methane feed (inculding the N₂), calculate: a. How many moles of H₂ are lost in the purge. b. How many moles of makeup CO₂ are required. c. The recycle to purge ratio in mol/mol. d. How much methanol solution (in kg/h) of what strength (weight percent) is produced. 3 | Page
Expert Answer:
Answer rating: 100% (QA)
100mol feed into the reformer 99 mol Methane N2 1 mol Reactions 2H2O CO2 4H2 CH4 main CH4 CO2 3H2 si... View the full answer

Related Book For 

Thermodynamics An Engineering Approach
ISBN: 978-0073398174
8th edition
Authors: Yunus A. Cengel, Michael A. Boles
Posted Date:
Students also viewed these chemical engineering questions
-
Determine the amount of heat removed from the reactor per mole of CH4 fed to the reactor. 2. Methane and oxygen at 25C are fed to a continuous reactor in stoichiometric amounts according to the...
-
A steady stream of equimolar N2 and CO2 mixture at 100 kPa and 18C is to be separated into N2 and CO2 gases at 100 kPa and 18C. Determine the minimum work required per unit mass of mixture to...
-
A light-hydrocarbon feed stream contains 45.4 kmol/h of propane, 136.1 kmol/h of isobutane, 226.8 kmol/h of n-butane, 181.4 kmol/h of isopentane, and 317.4 kmol/h of n-pentane. This stream is to be...
-
Carbon has the electron configuration 1s 2s2p. The two unpaired electrons in the n = 2 level suggests that carbon will form two bonds. We know, however, that carbon forms four bonds in most compounds...
-
Using Formula 71 on page 165, compute RF (risk-free rate). The real rate of return is 3 percent and the expected rate of inflation is 5 percent.
-
The Centers for Disease Control and Prevention reports that 25% of baby boys 68 months old in the United States weigh more than 20 pounds. A sample of 16 babies is studied. a. What is the probability...
-
The May cash records of Nielson, Inc., follow: Nielsons Cash account shows the balance of $6,171 on May 31. On May 31, Nielson, Inc., received the following bank statement: Additional data for the...
-
1. What are the communication errors Samuel made? 2. What should Angelique do when she gets the phone call from Penny asking her to come to Dallas to meet with Samuel? 3. Is there anything more...
-
A Product J has only two processes. Material is added at BEGINNING of the process X while at END of the process Y. Labor costs are added evenly, however, after 50% stage of completion in each process...
-
On January 1, 2023, Logan Limited had shares outstanding as follows: 6% cumulative preferred shares, $100 par value, 10,000 shares issued and outstanding $1,000,000 Common shares, 200,000 shares...
-
Determine which of the given lines are parallel to each other and which are perpendicular to each other. (a) 2 x 3 y + 4 = 0 (b) 3 x 2 y = 0 (c) 2 x 3 y = 7 (d) 2 x + 3 y + 2 = 0 (e) 2 x + 3 y + 9 =...
-
misks you colazib os ton vilesi mol yas What is the output of the following snippets of code? (15 points) a. int fees[] = {1, 2, 3, 4, 5, 6}; int fines[] = {10, 20, 30, 40}; int i = 4; for (i=0;i
-
For tax purpose, the company changes (1) depreciation method for building ($100,000,000) from double-declining to straight, and (2) salvage value from $25,000,000 to $868,800. Tax rate is 20% for all...
-
A bug zapper consists of two metal plates connected to a high-voltage power supply. The voltage between the plates is set to give an electric field slightly less than 1x10 V/m. When a bug flies...
-
Mrs. Munch later informs you and attorney Case that she will need back surgery due to this accident. Attorney Case is unsure if the surgery is related based upon the medical records. Should an expert...
-
Long-term liabilities Bonds payable $3,600,000 Less: Discount on bonds payable 81,000 $3,519,000 Oriole decides to redeem these bonds at 106 after paying annual interest. Prepare the journal entry to...
-
Office Equipment 5 10 4,987 1,175 800 Other Equipment 7 10 14,867 15,666 7,288 Vehicles 5 5 47,210 39,644 19,995 Office Furniture & Fixtures 7 10 5,477 10,400 4,800 Building 39 50 187,640 0 0 WTG...
-
Use a calculator to evaluate the expression. Round your result to the nearest thousandth. V (32 + #)
-
Derive expressions for (a) u. (b) h. (c) s for a gas whose equation of state is P(v - a) = RT for an isothermal process.
-
Describe the solar radiation properties of a window that is ideally suited for minimizing the air-conditioning load.
-
Reconsider the scram jet engine discussed in Prob. 17-41. Determine the speed of this engine in miles per hour corresponding to a Mach number of 7 in air at a temperature of 00F.
-
The \(\operatorname{arm} O A\) of an epicyclic gear train shown in Fig. 106 below revolves counter-clockwise about \(O\) with an angular velocity of \(4 \mathrm{rad} / \mathrm{s}\). Both gears are of...
-
The lengths of the links of a 4-bar linkage with revolute pairs only are \(p, q, r\) and \(s\) units. Given that \(p
-
The mechanism used in a shaping machine is (a) a closed 4-bar chain having 4 revolute pairs (b) a closed 6-bar chain having 6 revolute pairs (c) a closed 4-bar chain having 4 revolute and 2 sliding...

Study smarter with the SolutionInn App


