Indicate the most and the least stable carbanion of the following: OH 0N -F NO ...
Fantastic news! We've Found the answer you've been seeking!
Question:
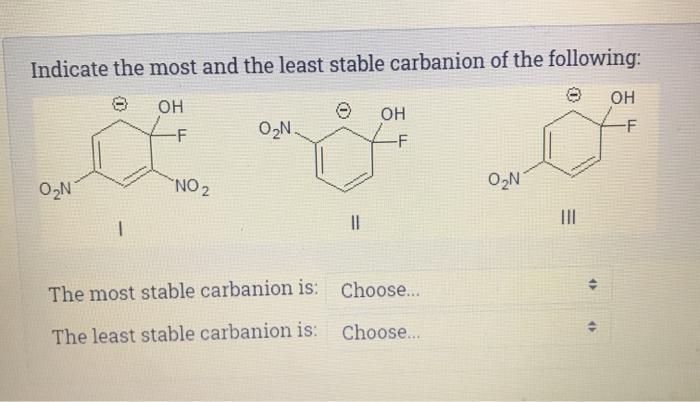
Transcribed Image Text:
Indicate the most and the least stable carbanion of the following: OH 0₂N -F NO ₂ O₂N. || OH -F The most stable carbanion is: Choose... The least stable carbanion is: Choose... O₂N ||| 46 OH -F Indicate the most and the least stable carbanion of the following: OH 0₂N -F NO ₂ O₂N. || OH -F The most stable carbanion is: Choose... The least stable carbanion is: Choose... O₂N ||| 46 OH -F
Expert Answer:
Answer rating: 100% (QA)
compound with more ternary carbon atoms is more sta... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
The most stable conformation of 5-hydroxy-1,3-dioxane has the OH group in an axial position, rather than an equatorial position. Provide an explanation for this observation.
-
Choose the more stable alkene in each of the following pairs. Explain your reasoning. (a) 1-Methylcyclohexene or 3-methylcyclohexene (b) Isopropenylcyclopentane or allylcyclopentane (c) (d)...
-
The resonance form shown is not the most stable one for the compound indicated. Write the most stable resonance form.
-
Litchfield Industries gathered the following information for the month ended June 31: The static budget volume is 5000 units: Overhead flexible budget: Number of units 8,000 9,000 10,000 Standard...
-
1. Suppose youve just started working in the office of the International Intellectual Property Alliance (IIPA; www.iipa.com). You learn that worldwide piracy of products is rampant. Your boss assigns...
-
Suppose that the acceleration of a car increases with time. Could we use the relationship v = v 0 + at in this situation? Explain
-
Ozone is a major component of air pollution in many cities. Atmospheric ozone levels are influenced by many factors, including weather. In one study, the mean percent relative humidity (x) and the...
-
On January 1, 2015, Global Manufacturing purchased a machine for $920,000 that it expected to have a useful life of five years. The company estimated that the residual value of the machine was...
-
Your firm has a British customer that is willing to place a $1 million order, but wants to pay in pounds instead of dollars. The spot exchange rate is $1.85 = 1.00 and the one-year forward rate is...
-
On December 31, Y3, Aylmer Industries Inc. purchased 85% of the outstanding shares of Belmont Inc. The purchase price is indicated on the excel file. The entry has been recorded on the books of...
-
uaicte Company sold 75,000 units of their only product during the year, while they produced 82,000 units. Their cost structure was: Variable Manufacturing Cost per unit: $25 Variable Selling and...
-
What should be the STP (Segmentation, Targeting, Positioning) based on boat industry in Canada?
-
Find Current ratio Sales Revenue $13,200 Add: Net Income 1,595 Cash $85,700 Total Revenue 13,200 Subtotal 1,595 Store supplies 250 Less: Dividends 1,500 Prepaid Insurance 5,500 Expenses: Retained...
-
2020 annual report for Cisco Ratios 2019 Workings 2020 Workings Formulae a Net profit margin 22.4% 11621/51904 22.7% 11214/49301 net income/net sales b Return on assets 11.9% 11621/97793 11.8%...
-
What deal(s) with the overall plan for deploying the resources that a firm possesses?
-
The income statement for Westbound Manufacturing is provided in the table below (all values are in million dollars): Gross sales less Cost of goods sold: Labour Raw materials Production overhead...
-
Quality Cabinets Inc. (Quality) manufactures custom kitchen cabinets from its location in Squamish, British Columbia. The cabinets are then sold to contractors who install them in kitchens. Bruce...
-
The bookkeeper for Riley, Inc., made the following errors: a. A cash purchase of supplies of $357 was recorded as a debit to Supplies for $375 and a credit to Cash of $375. b. A cash sale of $3,154...
-
Draw the resonance contributors for the carbocation intermediates obtained from electrophilic aromatic substitution at the 1-position and the 2-position of naphthalene. Use the resonance contributors...
-
Give two sets of alkyl bromide and alkene that could be used in a Heck reaction to prepare the following compound: CH3C CH= CH OCH3
-
Determine the structure of each of the following compounds based on its mass, IR, and 1H NMR spectra. a. b. 100 u 50 27 114 20 40 60 80 100 120 m/z 3 2 2 28 2.9 10 13 14 15 6 6 5 2 (ppm) frequency...
-
Jordan \& Stone, CPAs, audited the financial statements of Tech Co., a nonpublic entity, for the year ended December 31, 19X1, and expressed an unqualified opinion. For the year ended December 31,...
-
You have been engaged by the trustees of Roger Trust to audit the trust as of December 31, 19XX. The audit is for the purpose of assuring the trustees and beneficiaries that trust assets,...
-
The auditors' report below was drafted by a staff accountant of Baker and Baker, CPAs, at the completion of the audit of the comparative financial statements of Ocean Shore Partnership for the years...

Study smarter with the SolutionInn App


