INFO:: Used 3 ml styreNE 0.40 ml initiator tert butyl obtained 0.8660 g polystyrene polymerized 1.5512 g
Fantastic news! We've Found the answer you've been seeking!
Question:



INFO:: Used 3 ml styreNE
0.40 ml initiator tert butyl
obtained 0.8660 g polystyrene
polymerized 1.5512 g styrene
QUESTIONS:
1. A feature of polymers compared to “normal” molecules, which have fixed molecular weights, is that the same polymer can have a range of molecular weights depending on the conditions under which it was made. For the reaction we did, assume each initiator molecule starts one polystyrene chain and each chain is the same length, calculate the number of styrene molecules per chain and thus the weight of a chain of YOUR polymer.
2. Suggest a reason why methanol (the solvent used in this lab) is a better solvent for isolating polystyrene as a powder than acetone.
Transcribed Image Text:
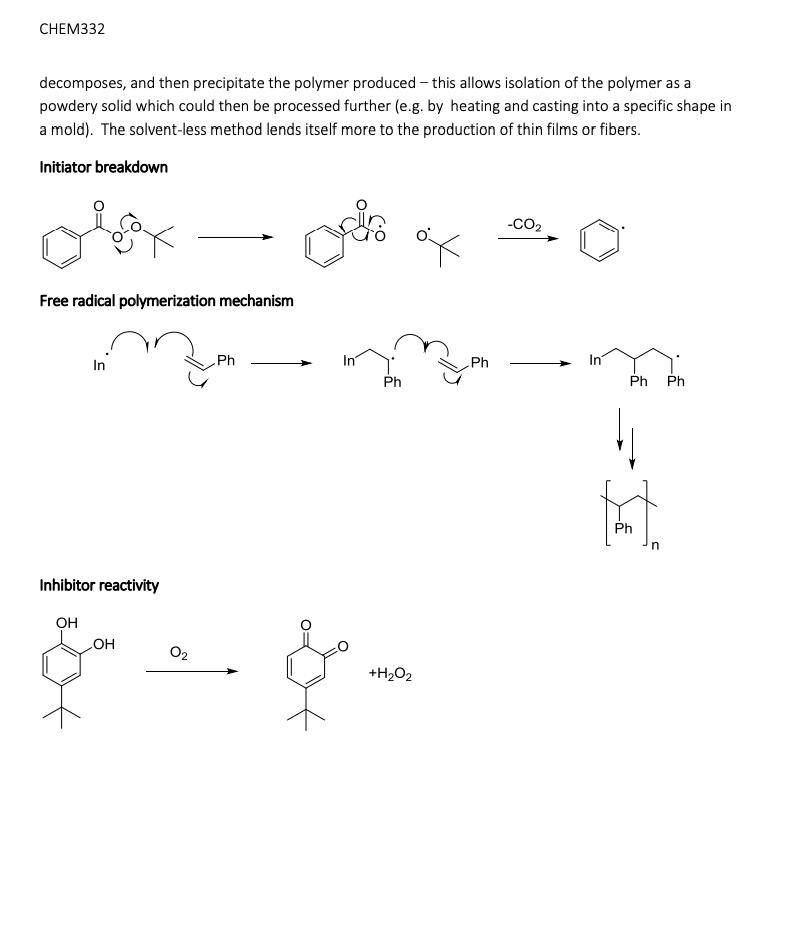
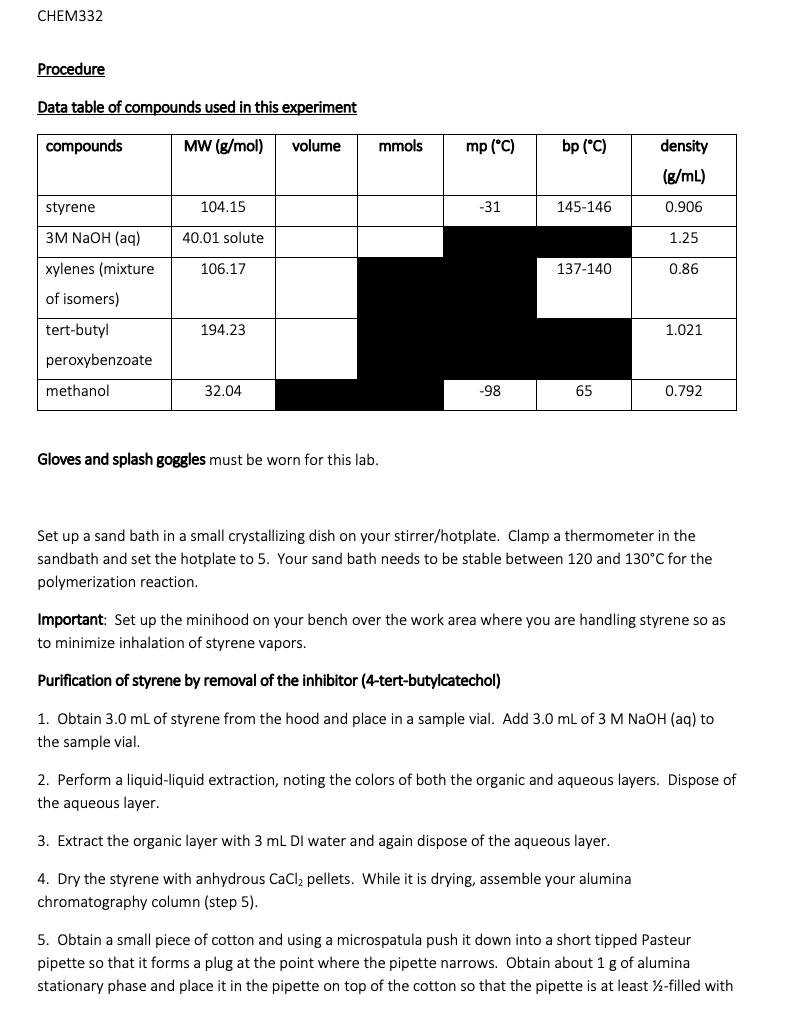
Simple alkenes are one of the most common types of monomer for polymerization reactions. Although they can be polymerized by ionic mechanisms, free radical polymerization is the most common method and you may have discussed the mechanism in CHEM331. In this lab we will polymerize styrene to form polystyrene, which is made on a huge scale industrially every year (millions of tonnes): Its primary uses are in packaging materials and in containers. One major issue associated with polystyrene is environmental - the material is very stable due to the strong, non-polar covalent bonds that comprise the alkane polymer chain and so is very slow to break down in the environment (biodegrade). This leads to build up of plastics in the ocean and other areas if it is not disposed of properly. Free radical polymerization requires an initiator - a compound that breaks apart easily to form free radicals that then begin the chain growth process. Initiators are usually compounds that contain a weak covalent bond that can be readily homolytically cleaved with either heat or light to form radicals: We will be using tert-butyl peroxybenzoate which contains a weak peroxy, or -0-0-, bond. OH Ph or tert-butyl peroxybenzoate OH 4-tert-butyl catechol styrene Many alkenes are so reactive towards this type of polymerization that the reaction may be initiated just by the presence of O₂ (itself a radical). To prevent this, the monomer must be stored long term at low temperatures and in the presence of an inhibitor, a compound which preferentially reacts with O₂ thus protecting the monomer. The amount of inhibitor is quite small, in the range 10-15 ppm. In this experiment, we will first remove the inhibitor from the styrene monomer before attempting the polymerization reaction. This is easily done as the inhibitor is 4-tert-butyl catechol, which contains weakly acidic phenol groups, and so reaction with a strong base and liquid-liquid extraction allows removal of the deprotonated form in the aqueous layer (see the acid-neutral separation lab from CHEM331). Polymerization of alkenes can be done in the absence or presence of a solvent. In this lab we will do the polymerization of styrene in the solvent xylenes (dimethylbenzene) at reflux so that the initiator thermally Simple alkenes are one of the most common types of monomer for polymerization reactions. Although they can be polymerized by ionic mechanisms, free radical polymerization is the most common method and you may have discussed the mechanism in CHEM331. In this lab we will polymerize styrene to form polystyrene, which is made on a huge scale industrially every year (millions of tonnes): Its primary uses are in packaging materials and in containers. One major issue associated with polystyrene is environmental - the material is very stable due to the strong, non-polar covalent bonds that comprise the alkane polymer chain and so is very slow to break down in the environment (biodegrade). This leads to build up of plastics in the ocean and other areas if it is not disposed of properly. Free radical polymerization requires an initiator - a compound that breaks apart easily to form free radicals that then begin the chain growth process. Initiators are usually compounds that contain a weak covalent bond that can be readily homolytically cleaved with either heat or light to form radicals: We will be using tert-butyl peroxybenzoate which contains a weak peroxy, or -0-0-, bond. OH Ph or tert-butyl peroxybenzoate OH 4-tert-butyl catechol styrene Many alkenes are so reactive towards this type of polymerization that the reaction may be initiated just by the presence of O₂ (itself a radical). To prevent this, the monomer must be stored long term at low temperatures and in the presence of an inhibitor, a compound which preferentially reacts with O₂ thus protecting the monomer. The amount of inhibitor is quite small, in the range 10-15 ppm. In this experiment, we will first remove the inhibitor from the styrene monomer before attempting the polymerization reaction. This is easily done as the inhibitor is 4-tert-butyl catechol, which contains weakly acidic phenol groups, and so reaction with a strong base and liquid-liquid extraction allows removal of the deprotonated form in the aqueous layer (see the acid-neutral separation lab from CHEM331). Polymerization of alkenes can be done in the absence or presence of a solvent. In this lab we will do the polymerization of styrene in the solvent xylenes (dimethylbenzene) at reflux so that the initiator thermally
Expert Answer:
Answer rating: 100% (QA)
Detailed Explanation The experiment involves the use of tertbutyl peroxybenzoate as an initiator for free radical polymerization The process also includes the removal of the inhibitor 4tertbutyl catec... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemical engineering questions
-
A reality of communication is that the same sign often means different things to different people. The red ribbon, for example, means different things to different groups. Provide an example from...
-
Polystyrene can be made more rigid by copolymerizing styrene with divinylbenzene, What purpose does the divinylbenzene serve? Why is the copolymer more rigid? CH=CH2 CH=CH,
-
Polymer Molecular Weight Distribution the following data were obtained for polyethylene. Determine the average molecular weight and degree of polymerization.
-
Two pulses A and B are moving in opposite directions along a taut string with a speed of 2.00 cm/s. The amplitude of A is twice the amplitude of B. The pulses are shown in Figure P18.2 at t = 0....
-
Explain, in your own words, the difference between a multicurrency translation exposure report and a multicurrency transactions exposure report.
-
Discuss the challenges associated with maintaining consistency and consensus in distributed operating systems. What role do protocols like Paxos and Raft play in ensuring reliability and fault...
-
By finding the trace of the appropriate coherency matrix, show that the average intensity transmitted by a polarization analyzer set at \(+45^{\circ}\) to the \(X\)-axis can be expressed as \[...
-
A certain industrial firm desires an economic analysis to determine which of two different machines should be purchased. Each machine is capable of performing the same task in a given amount of time....
-
You are presented with the following trial balance of Golden City Bhd as at 31 December 2020: Debit RM'000 Credit RM'000 Intangible assets 50 Plant at cost 100 Plant, accumulated depreciation, at 1...
-
Who do you think the CEO should appoint as CIO? Why? The Suarez Effect Pat Talley stood and watched, with grudging admiration, as Carmelita Suarez worked the room. Sharp, charming, and armed with a...
-
Find all complex cube roots of z=-1-5i. Write each root in rectangular form rounding to two decimal places. The complex third root(s) of z is (are). (Round the final answer to two decimal places as...
-
3. Mr. and Mrs. Smith have just purchased a $600,000 house and have made a down payment of $120,000. They can amortize the balance at 4% for 30 years. a. calculate monthly payments. b. calculate...
-
Interest During Construction Matrix Inc. borrowed $1,100,000 at 8% to finance the construction of a new building for its own use. Construction began on January 1, 2019, and was completed on October...
-
Utilize the stock quote to answer the following questions about Burger King's stock. a. Find the current price per share when this report was generated on Dec 8 th . b. What was the price per share...
-
Compare three alternatives on the basis of their capitalized costs at /= 9% per year and select the best alternative. Alternative First Cost AOC, per Year E $-65,000 $-55.000 $16,000 F $-380,000...
-
(a) Let A = {1,2,3,4}, B = {3kk e Z-{-1,1}} and C = {ne Zn+n = 0}. Which one of the following statements is True? Explain your answer i. AnB = 0 ii. B-C=0 iii. BnC = 0 iv. AUC =0 (b) Let A and B be...
-
For the function y(x + 4)(x3 - 9x), at (-3, 0) find the following. (a) the slope of the tangent line (b) the instantaneous rate of change of the function Need Help? Read It Show My Work (Optional) (...
-
State whether each statement is true or false. If false, give a reason. {purple, green, yellow} = {green, pink, yellow}
-
The hydrolysis of the ester shown here is catalyzed by morpholine, a secondary amine. Propose a mechanism for this reaction. (The pKa of the conjugate acid of morpholine is 9.3, so morpholine is too...
-
The most stable form of glucose (blood sugar) is a six-membered ring in a chair conformation with its five substituents all in equatorial positions. Draw the most stable form of glucose by putting...
-
An aldol addition can be catalyzed by acids as well as by bases. Propose a mechanism for the acid-catalyzed aldol addition of propanal.
-
Which of Lees junior analysts three statements is correct? A. Statement 1 B. Statement 2 C. Statement 3 Elaine Lee is an analyst at an investment bank covering the energy sector. She and her junior...
-
Titian faces long-term risk from ____ due to potential regulatory changes in the developing economies. Theresa Blass manages the Toptier Balanced Fund (the Fund) and recently hired John Yorkton, a...
-
Which of the statements made by Yorkton on ESG factors in investment analysis is correct? A. Statement 1 B. Statement 2 C. Statement 3 Theresa Blass manages the Toptier Balanced Fund (the Fund) and...

Study smarter with the SolutionInn App


