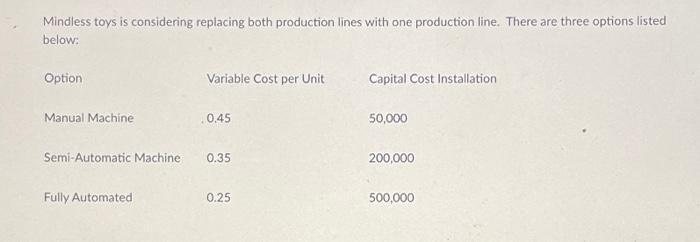
Mindless toys is considering replacing both production lines with one production line. There are three options...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
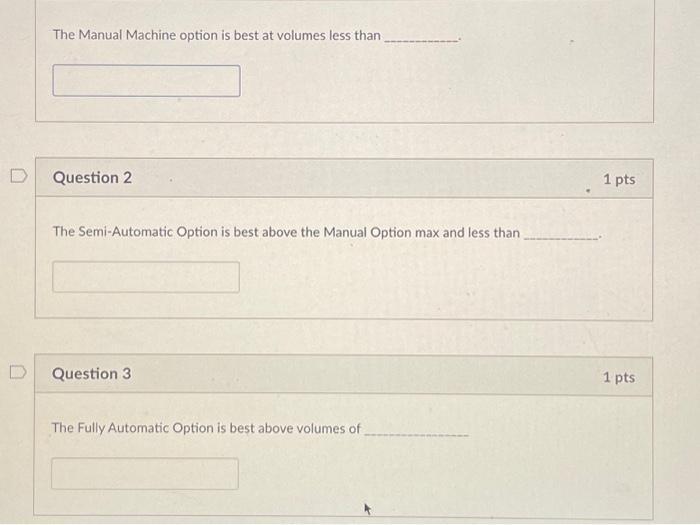
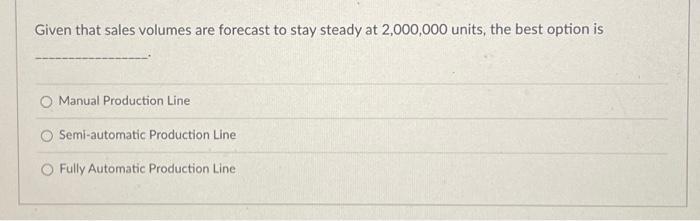
Mindless toys is considering replacing both production lines with one production line. There are three options listed below: Option Manual Machine Variable Cost per Unit Fully Automated 0.45 Semi-Automatic Machine 0.35 0.25 Capital Cost Installation 50,000 200,000 500,000 The Manual Machine option is best at volumes less than Question 2 The Semi-Automatic Option is best above the Manual Option max and less than Question 3 The Fully Automatic Option is best above volumes of 1 pts 1 pts Given that sales volumes are forecast to stay steady at 2,000,000 units, the best option is O Manual Production Line Semi-automatic Production Line Fully Automatic Production Line Mindless toys is considering replacing both production lines with one production line. There are three options listed below: Option Manual Machine Variable Cost per Unit Fully Automated 0.45 Semi-Automatic Machine 0.35 0.25 Capital Cost Installation 50,000 200,000 500,000 The Manual Machine option is best at volumes less than Question 2 The Semi-Automatic Option is best above the Manual Option max and less than Question 3 The Fully Automatic Option is best above volumes of 1 pts 1 pts Given that sales volumes are forecast to stay steady at 2,000,000 units, the best option is O Manual Production Line Semi-automatic Production Line Fully Automatic Production Line
Expert Answer:
Answer rating: 100% (QA)
ANS 1 To determine at what volume the Manual Machine option is best we need to compare the total costs of each option The Manual Machine has a variable cost per unit of 045 and a capital cost installa... View the full answer

Related Book For 

Posted Date:
Students also viewed these general management questions
-
Managing Scope Changes Case Study Scope changes on a project can occur regardless of how well the project is planned or executed. Scope changes can be the result of something that was omitted during...
-
Study the information given below and answer the following questions: (25 Marks) 1.1 Calculate the Payback Period of both machines (answers expressed in years, months and days). (5 marks) 1.2 Which...
-
XYZ Co is a prominent company in the FMCG industry. It provides 400 different brands spanning 14 categories of home, personal care and foods products. XYZ is now one of the worlds biggest companies....
-
Why " Kodak " is unsuccessful in implementing a strategy. Can you prepare a critical examination of the strategy to address the following questions about Kodak. What was the strategy and why do you...
-
Determine the value of θ corresponding to the equilibrium position of the mechanism of Prob. 10.9 when 60 P = lb and Q = 75 lb. E
-
x = 52, n = 71, confidence level 97% In Exercises 1518, use the given data to construct a confidence interval of the requested level.
-
Communication between spouses to the other during a valid marriage is privileged. Just as with hearsay, however, there are notable exceptions. Investigate the California Evidence Code 980-987 and...
-
Morro Bay Surfboards is a California company that manufactures fibreglass surf boards. The standard cost of direct materials and direct manufacturing labour is $100 per board. This includes 20 pounds...
-
What did you learn about this week that grabbed your attention or caused you to think more deeply about it? As mentioned in the introduction, at the core of psychology in practice is the use of...
-
a. During February, $194,500 was paid to creditors on account, and purchases on account were $210,400. Assuming that the February 28 balance of Accounts Payable was $62,500, determine the account...
-
Your bank offers you a $500,000 25-year housing loan at a rate of 6% pa compounding monthly. After 5 years of making your regular monthly payments, you receive a pay rise and decide to increase your...
-
Recall that Q is closed under addition and multiplication. That is, if a, b e Q then a + b = Q and ab e Q. 1. Recall that I is the set of all irrational numbers, that is I (xER:*#Q) (a) Let a e Q and...
-
2.5: Lithium is the lightest metal, with a density of only 0.53g/cm3. What is the approximate mass of a cube made of lithium thatmeasures 2.0 m on all three sides?
-
The finance controller is seeking process improvement to reduce customer complaints due to late billings. The concerned departments include customer service who receives and processes orders,...
-
The Student Project tions S(z) which varied from 0 to 1 as z went from-co to +o. These functions, called compared several func- response functions, are used to model the growth of activity in a...
-
What roles have social factors ( beyond the sphere of health care orpublic health ) played in controlling or failing to control the HIV / AIDS epidemic?
-
What communication skills do you believe are essential for engaging in intercultural praxis for social justice, and how can they contribute to creating positive change? In what ways can we show...
-
The area of square PQRS is 100 ft2, and A, B, C, and D are the midpoints of the sides. Find the area of square ABCD. B A
-
(a) Determine the values of sample average and standard deviation of the data in Problem. (b) Determine the values 1 and 2 standard deviations from the mean. Of the 50 sample points, how many fall...
-
Convert the given interest rates in the left-hand column into the nominal rates listed in the right-hand column. (Assume 4 weeks/month.) Given Interest Rate Desired Interest Rate 1% per month...
-
During a period when the real estate market in Phoenix, Arizona, was undergoing a significant downturn, CSM Consulting Engineers made an agreement with a distressed seller to purchase an office...
-
Show that \[ \gamma_{i}=\frac{\bar{\phi}_{i}}{\phi_{i}} \]
-
Carbon dioxide occupies a tank at \(100^{\circ} \mathrm{C}\). If the volume of the tank is \(0.5 \mathrm{~m}^{3}\) and the pressure is \(500 \mathrm{kPa}\), determine the mass of the gas in the tank.
-
If a cylinder of volume \(0.1 \mathrm{~m}^{3}\) is filled with \(1.373 \mathrm{~kg}\) of ammonia at \(1.95 \mathrm{MPa}\), determine the temperature at which ammonia exists in the cylinder. Assume...

Study smarter with the SolutionInn App


