Mr. John Hanson is an employee of a public company. He was given stock options from...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
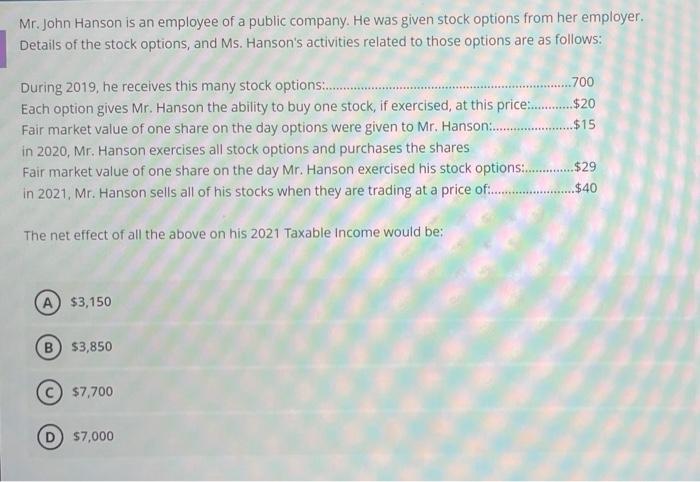
Mr. John Hanson is an employee of a public company. He was given stock options from her employer. Details of the stock options, and Ms. Hanson's activities related to those options are as follows: .700 During 2019, he receives this many stock options:. Each option gives Mr. Hanson the ability to buy one stock, if exercised, at this price: .$20 Fair market value of one share on the day options were given to Mr. Hanson .$15 in 2020, Mr. Hanson exercises all stock options and purchases the shares Fair market value of one share on the day Mr. Hanson exercised his stock options .$29 in 2021, Mr. Hanson sells all of his stocks when they are trading at a price of . ..$40 The net effect of all the above on his 2021 Taxable Income would be: A $3,150 $3,850 $7,700 57,000 Mr. John Hanson is an employee of a public company. He was given stock options from her employer. Details of the stock options, and Ms. Hanson's activities related to those options are as follows: .700 During 2019, he receives this many stock options:. Each option gives Mr. Hanson the ability to buy one stock, if exercised, at this price: .$20 Fair market value of one share on the day options were given to Mr. Hanson .$15 in 2020, Mr. Hanson exercises all stock options and purchases the shares Fair market value of one share on the day Mr. Hanson exercised his stock options .$29 in 2021, Mr. Hanson sells all of his stocks when they are trading at a price of . ..$40 The net effect of all the above on his 2021 Taxable Income would be: A $3,150 $3,850 $7,700 57,000
Expert Answer:
Answer rating: 100% (QA)
OPTION D Solution Employment Income Market value Exerc... View the full answer

Related Book For 

Managers and the Legal Environment Strategies for the 21st Century
ISBN: 978-1111530631
7th edition
Authors: Constance E. Bagley
Posted Date:
Students also viewed these accounting questions
-
In Year 1, Kayla, an employee of a public company, was granted an option to acquire 100 shares from the companys treasury at a price of $12 per share. At the date that the option was granted, the...
-
Is it ever ethical for an employee of a company to accept gifts from and individual or firm that does business or wishes to do business with that company? If so, under what circumstances?
-
In 2015, Maggy (34 years old) is an employee of YBU Corp. YBU provides a 401(k) plan for all its employees. According to the terms of the plan, YBU contributes 50 cents for every dollar the employee...
-
41.(6 pointa) In the following balanced reaction, magnesium metal (Mg) reacts with carbon dioxide gas (CO) o form solid magnesium exide (MgO) and solid carbon (C). How many grams of carbon dioxide...
-
Briannas Wedding Shops earned net income of $25,000, which included depreciation of $16,000. Briannas acquired a $116,000 building by borrowing $116,000 on a long-term note payable. Requirements 1....
-
Specify whether the labeled protons in each of the following structures would be expected to have the same or different chemical shifts. (a) (b) CH3 H,C CH3 HC C-CH CI Hb
-
Two key performance indicators for a company are profitability and solvency. Why are these so important? Is the value of accrual accounting more evident when centered on profitability or solvency?
-
Amalgamated General Corporation is a consulting firm that also offers financial services through its credit division. From time to time the company buys and sells securities. The following selected...
-
The Cole Corporation has provided the following account balances: Account Amount Cash $20,000 Short-term Investments 5,000 Accounts Receivable 6,000 Supplies 7,000 Long-term Notes Receivable 3,000...
-
Wedding Planners Limited (WP), owned by Anne and Francois Tremblay, provides wedding planning and related services. WP owns a building (the Pavilion) that has been custom-made for hosting weddings....
-
Read the "Software Laws, Hypotheses and Conjectures" on Static and dynamic testing and find at least two (2) of these laws, which are challenged or contradicted by the Agile Development Methodology.
-
What is true about the process of recycling different materials? Responses A It is different for different materials.It is different for different materials. B It is the same for all materials.It is...
-
A purchase of a vehicle for cash would have what affect on the accounting equation? Question 8 options: A) Total asset amount remains the same. B) Total owner's equity is overstated. C) Both A and B...
-
Morgan Freeman, an 86-year-old widower, lost his wife in 2020 and has remained single since. With no dependents, he currently resides by himself. What would be his standard deduction for the tax year...
-
Which among the air pollution control technologies can both remove particulate matter and gaseous emissions from stationary sources? Explain your answer.
-
3. The radioactive isotope Molybdenum-99 is produced at Chalk River Laboratories. Molybdenum-99 has a half-life of 66 h. a. What is the exponential function that would describe the relationship...
-
A data analyst uses a changelog while cleaning their data. What data modifications should they track in the changelog? O Errors, deletions, and notes O Additions, changes, and queries O Errors,...
-
Determine two different Hamilton circuits in each of the following graphs. A B F G
-
Are termination benefits specified in a pre-petition employment agreement entitled to priority as administrative expenses when the employee is terminated after rendering post-petition services?
-
Is Matthews correct that her termination constituted unlawful discrimination under Title VII?
-
Was either Jeffrey or Brittany engaged in protected concerted action under the National Labor Relations Act when they made their Facebook postings? May Goalpost terminate them? Are Goalpost's threats...
-
a. For the allowed energies of a particle in a box to be large, should the box be very big or very small? Explain. b. Which is likely to have larger values for the allowed energies: an atom in a...
-
The molecules in the rods and cones in the eye are tuned to absorb photons of particular energies. The retinal molecule, like many molecules, is a long chain. Electrons can freely move along one...
-
What was the approximate activity of the plutonium source at the start of the mission? A. \(2 \times 10^{21} \mathrm{~Bq}\) B. \(2 \times 10^{19} \mathrm{~Bq}\) C. \(2 \times 10^{17} \mathrm{~Bq}\)...

Study smarter with the SolutionInn App


