Now consider the shown modified cycle. An adiabatic heat exchanger is inserted into the cycle. The...
Fantastic news! We've Found the answer you've been seeking!
Question:
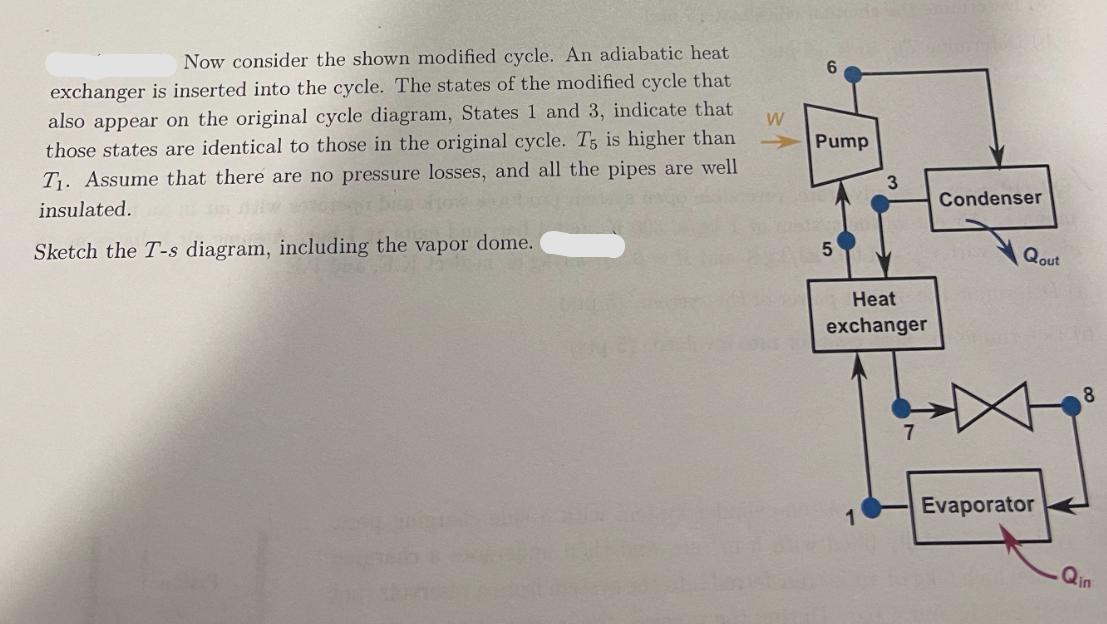
Transcribed Image Text:
Now consider the shown modified cycle. An adiabatic heat exchanger is inserted into the cycle. The states of the modified cycle that also appear on the original cycle diagram, States 1 and 3, indicate that those states are identical to those in the original cycle. T5 is higher than T₁. Assume that there are no pressure losses, and all the pipes are well insulated. Sketch the T-s diagram, including the vapor dome. W 6 Pump 5 3 Heat exchanger 1 Condenser Qout Evaporator 8 Qin Now consider the shown modified cycle. An adiabatic heat exchanger is inserted into the cycle. The states of the modified cycle that also appear on the original cycle diagram, States 1 and 3, indicate that those states are identical to those in the original cycle. T5 is higher than T₁. Assume that there are no pressure losses, and all the pipes are well insulated. Sketch the T-s diagram, including the vapor dome. W 6 Pump 5 3 Heat exchanger 1 Condenser Qout Evaporator 8 Qin
Expert Answer:

Related Book For 

Thermodynamics An Engineering Approach
ISBN: 978-0073398174
8th edition
Authors: Yunus A. Cengel, Michael A. Boles
Posted Date:
Students also viewed these mechanical engineering questions
-
How does a higher EPS potentially impact a company's stock price? 8. What are the limitations of using EPS as a standalone measure of a company's financial performance? 9. How can investors compare...
-
An adiabatic heat exchanger is used to heat cold water at 15C entering at a rate of 5 kg/s by hot air at 90C entering also at a rate of 5 kg/s. If the exit temperature of hot air is 20C, the exit...
-
An adiabatic heat exchanger is used to heat cold water at 15C entering at a rate of 5 kg/s by hot water at 90C entering at a rate of 4 kg/s. If the exit temperature of hot water is 50C, the exit...
-
- You have identified several risks on your project for which purchasing insurance is a possibility. The insurance company your firm uses has quoted reasonable rates, and your analysis shows that...
-
Suppose MidAmerican Energy is contemplating construction of a gas-fired power plant. The plant is likely to last 25 years and to have no salvage value . Depreciation allowances for tax purposes on...
-
Capricorn, Inc., is planning to go public by offering its common stock, which previously had been owned by only three shareholders. The company intends to limit the number of purchasers to...
-
Use i) Euler's Method and ii) the Midpoint Method to determine the given value of \(y\) for the following problems: a. \(\frac{d y}{d x}=2 y, y(0)=2\). Find \(y(1)\) with \(h=0.1\). b. \(\frac{d y}{d...
-
Baxter Inc., a developer of radiology equipment, has stock outstanding as follows: 18,000 shares of cumulative 2%, preferred stock of $75 par, and 40,000 shares of $10 par common. During its first...
-
The bank statement for Unique Fashion had an ending cash balance of $1,700 on March 31, 2019. On this date the cash balance in their general ledger was $2,891. After comparing the bank statement with...
-
Helix Company produces costumes used in the television and movie industries. Recently the company received an ongoing order for Samurai robes to be worn in an upcoming Japanese historical action...
-
Please explain what This quote is expressing and stating? Short snd straight to the point please and thank you! Mental Health "Some of the most well-documented consequences of sexualization relate to...
-
Arcon and Byrnco have both submitted bids for an equipment service contract. Their offers are: Arcon : $9,500 to be paid immediately $5,000 to be paid one year from now $5,000 to be paid two years...
-
Corey's annual salary is $86,800. He pays 23% in taxes. After taxes, he pays 4% into retirement. How much does Corey pay into retirement? 673.44 What is Corey's annual salary after taxes and...
-
The Alpine House, Incorporated, is a large retailer of snow skis. The company assembled the information shown below for the quarter ended March 31: Sales Selling price per pair of skis Variable...
-
Allison sued Marc for assault and battery in the U.S. District Court for the state of Loserville. Marc claimed that he acted in self-defense. The following testimony was offered at trial: Bryan...
-
On September 15, 2023, the Board of Directors of Cullumber Company declared a $1.15 per share cash dividend, payable on October 31 to shareholders of record as of October 1. On the date of...
-
I'm having some issues with the following task - It would be perfect if somebody could help me, please. I really would like to understand the details which means that I need really detailed...
-
The following information is available for Partin Company: Sales $598,000 Sales Returns and Allowances 20,000 Cost of Goods Sold 398,000 Selling Expense 69,000 Administrative Expense 25,000 Interest...
-
A refrigeration system uses water-cooled condenser for rejecting the waste heat. The system absorbs heat from a space at 25F at a rate of 24,000 Btu/h. Water enters the condenser at 65F at a rate of...
-
Determine the enthalpy of nitrogen, in kJ/kg, at 175 K and 8 MPa using (a) Data from the ideal-gas nitrogen table. (b) The generalized enthalpy departure chart. Compare your results to the actual...
-
Obtain a relation for the second-law efficiency of a heat engine that receives heat QH from a source at temperature TH and rejects heat QL to a sink at TL , which is higher than T0 (the temperature...
-
\(4 \times 13 \times 25\) Use properties of real numbers and mental math to calculate the expression.
-
\(13 \times 99\) Use properties of real numbers and mental math to calculate the expression.
-
\(23+98+75+12+77\) Use properties of real numbers and mental math to calculate the expression.

Study smarter with the SolutionInn App


