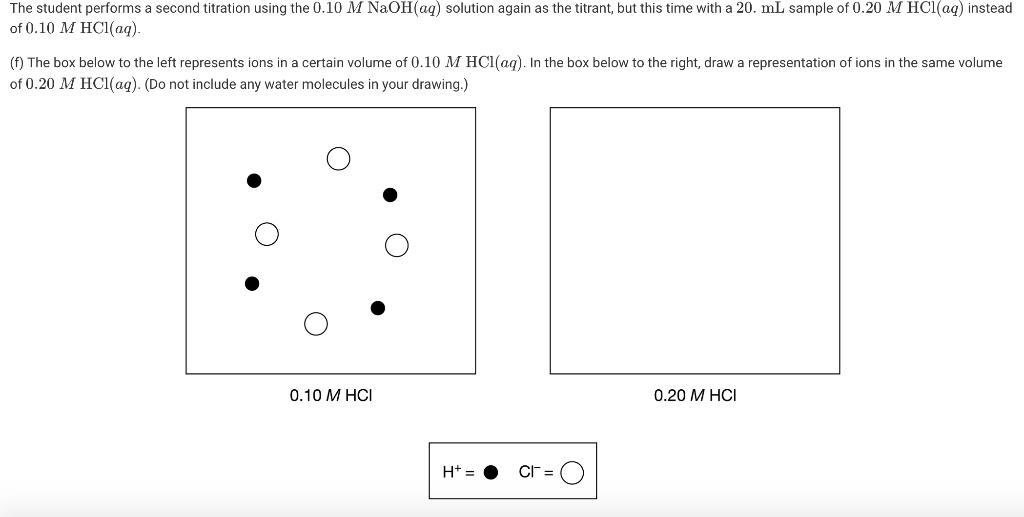
The student performs a second titration using the 0.10 M NAOH(aq) solution again as the titrant,...
Fantastic news! We've Found the answer you've been seeking!
Question:
![]()
Transcribed Image Text:
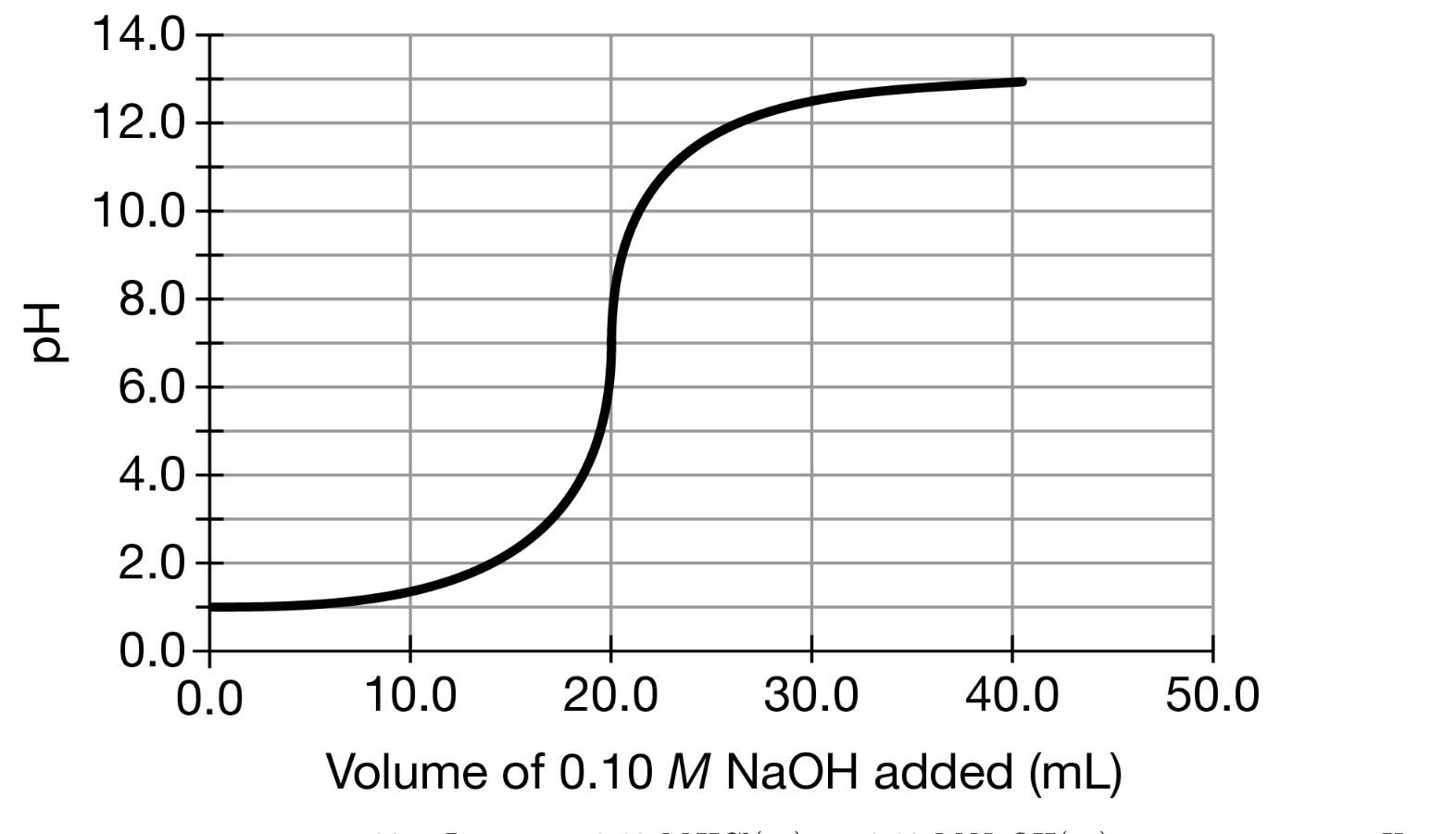
The student performs a second titration using the 0.10 M NAOH(aq) solution again as the titrant, but this time with a 20. mL sample of 0.20 M HCl(aq) instead of 0.10 M HC1(aq). (f) The box below to the left represents ions in a certain volume of 0.10 M HCl(aq). In the box below to the right, draw a representation of ions in the same volume of 0.20 M HC1(aq). (Do not include any water molecules in your drawing.) 0.10 M HCI 0.20 M HCI H+ = CF = (h) The student made observations related to the contents of the Erlenmeyer flask during the titration. Identify an observation that could have led the student to conclude that a chemical change took place during the titration. 14.0- 12.0- 10.0 8.0 6.0 4.0 2.0 0.0+ 0.0 10.0 20.0 30.0 40.0 50.0 Volume of 0.10 M NaOH added (mL) The student performs a second titration using the 0.10 M NAOH(aq) solution again as the titrant, but this time with a 20. mL sample of 0.20 M HCl(aq) instead of 0.10 M HC1(aq). (f) The box below to the left represents ions in a certain volume of 0.10 M HCl(aq). In the box below to the right, draw a representation of ions in the same volume of 0.20 M HC1(aq). (Do not include any water molecules in your drawing.) 0.10 M HCI 0.20 M HCI H+ = CF = (h) The student made observations related to the contents of the Erlenmeyer flask during the titration. Identify an observation that could have led the student to conclude that a chemical change took place during the titration. 14.0- 12.0- 10.0 8.0 6.0 4.0 2.0 0.0+ 0.0 10.0 20.0 30.0 40.0 50.0 Volume of 0.10 M NaOH added (mL)
Expert Answer:

Related Book For 

Posted Date:
Students also viewed these mathematics questions
-
The student performs a second titration using the 0.10M NaOH(aq) solution again as the titrant, but this time with a 20.mL sample of 0.20MHCl(aq) instead of 0.10MHCl(aq). 1. The box below to the left...
-
International mutual funds do not include any domestic assets whereas global mutual funds include both foreign and domestic assets. How might this difference affect their correlation with US equity...
-
Below are certain events that took place at Hazzard, Inc. , last year: a. Short-term investment securities were purchased. b. Equipment was purchased. c. Accounts payable increased. d. Deferred taxes...
-
Explain incremental cash flow, externality? Why is timing of cash flows important? What is scenario analysis? Would a project's NPV for a firm be higher or lower if the firm used accelerated rather...
-
Dry air at 25oC and atmospheric pressure blows over a 30-cm-square surface of ice at a velocity of 1.5 m/s. Estimate the amount of moisture evaporated per hour, assuming that the block of ice is...
-
Outline laboratory syntheses of each of the following compounds, starting with benzene and any other reagents. (The references to equations will assist you with nomenclature.) (a) p-dibromobenzene...
-
Identify the mode for each of the following sets of data: a. 6, 2, 7, 6, 6, 4 b. 13, 19, 12, 13, 7, 13, 20, 13, 15 c. 4, 2, 5, 1, 2, 2, 5, 5, 3, 1, 5, 2, 6, 3 d. 40, 10, 35, 30, 10, 25, 5, 10, 15,...
-
Whittington Company produces two types of leather purses: standard and handcrafted. Both purses use equipment for cutting and stitching. The equipment also has the capability of creating standard...
-
Reddy Company is a merchandising firm. Next month the company expects to sell 500 units. The following data describe the company's revenue and cost structure: Selling price per unit. $60 Sales...
-
Of all the price analysis methodologies that exist, which are the foundational methods. Longing & Shorting. Trading & Investing. Volume & sentiment. On-chain & Macroeconomic. Mean reversion &...
-
1. There is a box that contains 5 red balls and 3 white balls. a. If you take 2 balls randomly at the same time, what is the probability: - both balls are red - Both balls drawn are the same color -...
-
In this Individual Project (IP), you will examine and explain the history of industrial relations as they relate to labor, collective bargaining, and the role of human resources (HR), management,...
-
How to identify and document resources required to implement the sustainability policies? Answer the following questions: a. Why is it important to incorporate implementation and continuous...
-
1. Post at least one question regarding cohesion and coupling in software design. 2. Identify two methods in the GasBuddy system using the sequence diagram given in the GasBuddy documentation. For...
-
Your client, Ashkan Shirazi, a citizen of Iran, applied under the Federal Skilled Worker Class. Among other facts, the applicant's father lives in St. John's, Newfoundland with his wife. They are...
-
Answer the question using the table below. ACTIVITIES FOR A CONSTRUCTION PROJECT Activities Precedent activity Estimated Time Estimated Cost (weeks) (US$) A 4 10,000 B A 3 6,000 C A 2 4,000 D B 5...
-
What are the benefits of employing Secure Socket Layer (SSL) ? Why is the main purpose of using secure socket layer SSL )?
-
Design an experiment to demonstrate that RNA transcripts are synthesized in the nucleus of eukaryotes and are subsequently transported to the cytoplasm.
-
Write balanced half-reactions in which MnO - 4 acts as an oxidant at (a) pH = 0; (b) pH =10; (c) pH = 15.
-
Use the reaction quotient, Q, to explain why the fraction of dissociation of weak acid, HA, increases when the solution is diluted by a factor of 2.
-
Consider the electrophoresis of heparin in Figure 25-21. (a) Electrophoresis was carried out at pH 2.8, at which sulfate groups are negative. Why was reverse polarity (detector end positive) used?...
-
A blender does 5000 J of work on the food in its bowl. During the time the blender runs, 2000 J of heat is transferred from the warm food to the cooler environment. What is the change in the thermal...
-
Which system contains more atoms: 5 mol of helium (A = 4) or 1 mol of neon (A = 20)? A. Helium B. Neon C. They have the same number of atoms.
-
A sample of ideal gas is in a sealed container. The temperature of the gas and the volume of the container are both increased. What other properties of the gas necessarily change? (More than one...

Study smarter with the SolutionInn App


