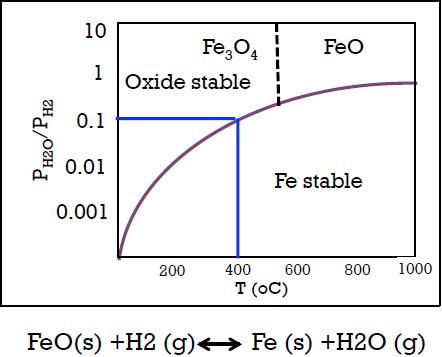
One hundred grams of iron oxide (FeO) is reacted with 1000 cm 3 of pure hydrogen(STP) in
Fantastic news! We've Found the answer you've been seeking!
Question:
One hundred grams of iron oxide (FeO) is reacted with 1000 cm3 of pure hydrogen(STP) in a closed system at one atmosphere using a temperature of 400 oC for an extended time. What is the weight of the pure Fe that would be formed? Assume that FeO is stable at 400 oC (WH2 = 2 gm/mol; WFe =56 gm/mole)
Related Book For 

Thermodynamics An Engineering Approach
ISBN: 978-0073398174
8th edition
Authors: Yunus A. Cengel, Michael A. Boles
Posted Date:




