As a technician in a large pharmaceutical research firm, you need to produce 150. mL of...
Fantastic news! We've Found the answer you've been seeking!
Question:

Transcribed Image Text:
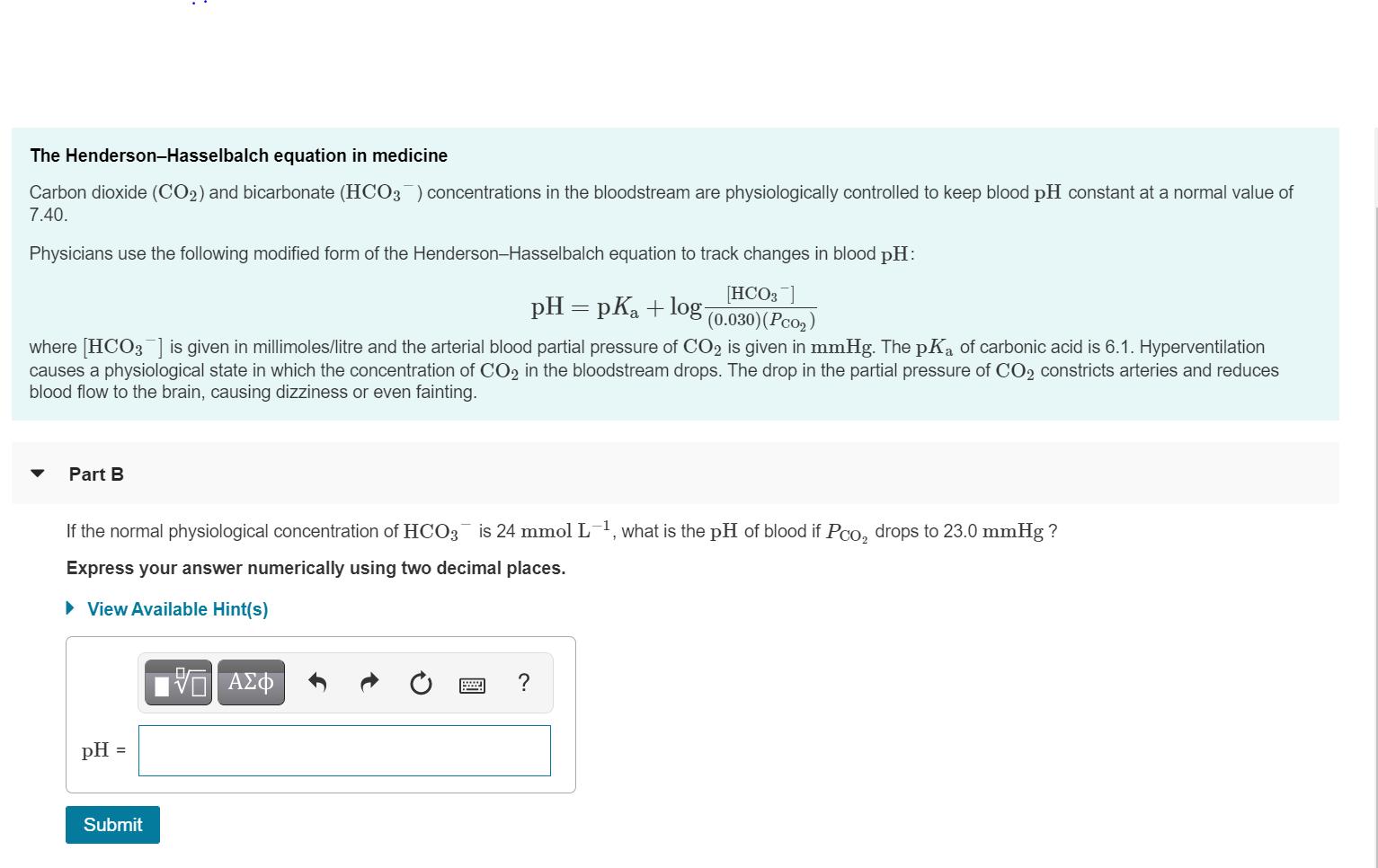
As a technician in a large pharmaceutical research firm, you need to produce 150. mL of 1.00 mol L potassium phosphate buffer solution of pH = 7.09. The pKa of H2PO4 is 7.21. You have the following supplies: 2.00 L of 1.00 mol L KH2PO4 stock solution, 1.50 L of 1.00 mol L K2HPO4 stock solution, and a carboy of pure distilled H20. How much 1.00 mol L KH3PO4 will you need to make this solution? Express your answer to three significant digits with the appropriate units. The Henderson-Hasselbalch equation in medicine Carbon dioxide (CO2) and bicarbonate (HCO3) concentrations in the bloodstream are physiologically controlled to keep blood pH constant at a normal value of 7.40. Physicians use the following modified form of the Henderson-Hasselbalch equation to track changes in blood pH: pH = pKa + log- [HCO3 ] (0.030)(Pco2 where [HCO3] is given in millimoles/litre and the arterial blood partial pressure of CO2 is given in mmHg. The pKa of carbonic acid is 6.1. Hyperventilation causes a physiological state in which the concentration of CO2 in the bloodstream drops. The drop in the partial pressure of CO2 constricts arteries and reduces blood flow to the brain, causing dizziness or even fainting. Part B If the normal physiological concentration of HC03 is 24 mmol L-1, what is the pH of blood if PCo, drops to 23.0 mmHg ? Express your answer numerically using two decimal places. • View Available Hint(s) ΑΣΦ ? pH = Submit As a technician in a large pharmaceutical research firm, you need to produce 150. mL of 1.00 mol L potassium phosphate buffer solution of pH = 7.09. The pKa of H2PO4 is 7.21. You have the following supplies: 2.00 L of 1.00 mol L KH2PO4 stock solution, 1.50 L of 1.00 mol L K2HPO4 stock solution, and a carboy of pure distilled H20. How much 1.00 mol L KH3PO4 will you need to make this solution? Express your answer to three significant digits with the appropriate units. The Henderson-Hasselbalch equation in medicine Carbon dioxide (CO2) and bicarbonate (HCO3) concentrations in the bloodstream are physiologically controlled to keep blood pH constant at a normal value of 7.40. Physicians use the following modified form of the Henderson-Hasselbalch equation to track changes in blood pH: pH = pKa + log- [HCO3 ] (0.030)(Pco2 where [HCO3] is given in millimoles/litre and the arterial blood partial pressure of CO2 is given in mmHg. The pKa of carbonic acid is 6.1. Hyperventilation causes a physiological state in which the concentration of CO2 in the bloodstream drops. The drop in the partial pressure of CO2 constricts arteries and reduces blood flow to the brain, causing dizziness or even fainting. Part B If the normal physiological concentration of HC03 is 24 mmol L-1, what is the pH of blood if PCo, drops to 23.0 mmHg ? Express your answer numerically using two decimal places. • View Available Hint(s) ΑΣΦ ? pH = Submit
Expert Answer:
Answer rating: 100% (QA)
part A The pH of a buffer can be calculated using the Henderson equation which is given below pH ... View the full answer

Related Book For 

Fundamentals of biochemistry Life at the Molecular Level
ISBN: 978-0470547847
4th edition
Authors: Donald Voet, Judith G. Voet, Charlotte W. Pratt
Posted Date:
Students also viewed these physics questions
-
As a technician in a large pharmaceutical research firm, you need to produce 150. mL of 1.00 mol L potassium phosphate buffer solution of pH = 7.09. The pKa of H2PO4 is 7.21. You have the following...
-
You need to produce a set of cylindrical copper wires 350 m long that will have a resistance of 0.125 each. What will be the mass of each of these wires?
-
By mixing available fuel, you need to produce 3500 gallons of gasoline for your feet of trucks this month. You plan to mix 800 gallons of fuel A (80 octane), 1500 gallons of fuel B (92 octane) and...
-
for a manufacturing company product costs include all of the following except direct material overhead costs research and development costs direct labor costs
-
On April 2, Kelly borrowed $15 000 on a demand note with an interest rate of 9% per annum. Payments were made of $2800 on May 14 and $2400 on June 19. How much was the final payment made on August 3?
-
What are the strengths and limitations of scenario analysis?
-
What is the most accepted input power required for an ICP accelerometer?
-
Fausto Fabricators Inc. uses a standard cost system to account for its single product. The standards established for the product include the following: Materials . . . . . . . . . . . . . . . . . . ....
-
An incentive plan is useful for all companies, because it establishes an action protocol for those employees who have a margin for progression in the medium term. Although a fair salary is a sine qua...
-
Last August, Malcolm (who is single) moved out of his rented apartment in Toronto, Ontario, to move to Vancouver, British Columbia, where he now lives and works. Malcolm has two T4s, one from Ontario...
-
1. A polynomial fit to measurements of the total volume of a binary mixture of I and Y is v = 778.55 - 22.5749 x + 0.56892 x + 0.01023 x+ 0.00234 x, where v = V/cm3, ny/mol, and ny is the amount of Y...
-
To address his concern regarding the previous advisers asset allocation approach, Raye should assess the Laws portfolio using: A. a homogeneous and mutually exclusive asset classbased risk analysis....
-
Raye believes the previous advisers asset class specifications for equity and derivatives are inappropriate given that, for purposes of asset allocation, asset classes should be: A. diversifying. B....
-
The list of securities that a particular ETF wants to own, which is disclosed daily by all ETFs, is referred to as the: A. creation unit. B. creation basket. C. redemption basket.
-
Which of the following statements relating to capital gains in ETFs and mutual funds is correct? A. ETFs tend to distribute less in capital gains than mutual funds do. B. Mutual funds may elect not...
-
Investors buying ETFs: A. incur management fees that decrease with the length of the holding period. B. are assured of paying a price equal to the NAV if they purchase shares at the market close. C....
-
List three unique aspects of budgeting for volunteer fire departments.
-
Assume a simple Keynesian depression economy with a multiplier of 4 and an initial equilibrium income of $3,000. Saving and investment equal $400, and assume full employment income is $4,000. a. What...
-
The half-cell reduction potential is provided by the Nernst equation (Equation 14-8): (a) On the graph below, plot the reduction potentials for the FADH2/FAD half-cell (E°² = - 0.219 V) when...
-
Some mitochondria use a second codon, in addition to AUG, to specify Met. Which codon(s) is(are) most likely to be used this way?
-
Broken DNA can be repaired by DNA ligase (single-strand breaks) or nonhomologous end-joining (double-strand breaks). Explain why the cell's set of repair enzymes also includes tyrosyl-DNA...
-
Carry out an Internet search to find more examples of project success and failure. From your search, are there any common themes in each? What are the implications of success and failure in each case?
-
How successful are government contracting arrangements? How do these compare, for instance, with the arrangements BAA had with their suppliers in the construction of T5 at Heathrow (Project...
-
What is the role of brainstorming and how might it be used to greatest effect?

Study smarter with the SolutionInn App