A 50.0-mL volume of 0.15 mol L' HBr is titrated with 0.25 mol L KOH. Calculate...
Fantastic news! We've Found the answer you've been seeking!
Question:
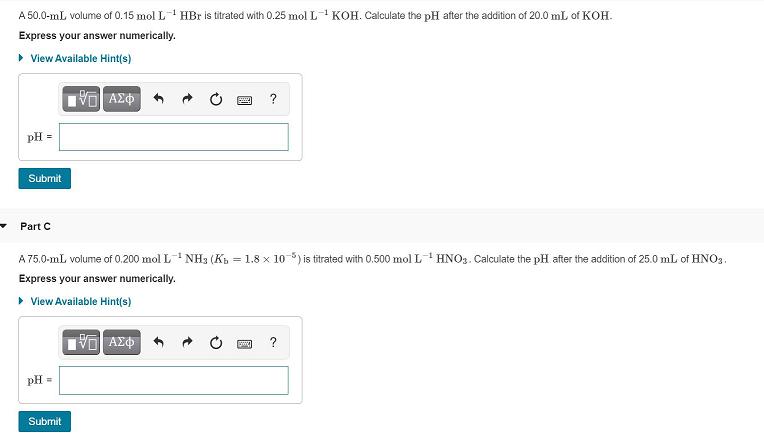
Transcribed Image Text:
A 50.0-mL volume of 0.15 mol L' HBr is titrated with 0.25 mol L KOH. Calculate the pH after the addition of 20.0 mL of KOH. Express your answer numerically. • View Available Hint(s) pH = Submit Part C A 75.0-mL volume of 0.200 mol L NH3 (Kb = 1.8 x 10 ) is titrated with 0.500 mol L HNO3. Calculate the pH after the addition of 25.00 mL of HNO3. Express your answer numerically. > View Available Hint(s) 阳AX中 ? pH = Submit Part D A 52.0-mL volume of 0.350 mol L CH3COOH (Ka = 1.8 x 10 5) is titrated with 0.400 mol L-1 NaOH. Calculate the pH after the addition of 27.0 mL of NaOH. Express your answer numerically. » View Available Hint(s) ΑΣΦ ? pH = Submit A 50.0-mL volume of 0.15 mol L' HBr is titrated with 0.25 mol L KOH. Calculate the pH after the addition of 20.0 mL of KOH. Express your answer numerically. • View Available Hint(s) pH = Submit Part C A 75.0-mL volume of 0.200 mol L NH3 (Kb = 1.8 x 10 ) is titrated with 0.500 mol L HNO3. Calculate the pH after the addition of 25.00 mL of HNO3. Express your answer numerically. > View Available Hint(s) 阳AX中 ? pH = Submit Part D A 52.0-mL volume of 0.350 mol L CH3COOH (Ka = 1.8 x 10 5) is titrated with 0.400 mol L-1 NaOH. Calculate the pH after the addition of 27.0 mL of NaOH. Express your answer numerically. » View Available Hint(s) ΑΣΦ ? pH = Submit
Expert Answer:

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
Use activity coefficients to calculate the pH after 10.0 mL of 0.100 M trimethylammonium bromide were titrated with 4.0 mL of 0.100 M NaOH.
-
The pH after the addition of 25 mL of 0.05 M NAOH to 20 ml of 0.05 M Malic acid equals (for Malic acid, Ka, = 3.84x10 Ka, 8.0sc10 Answer:
-
Dangerfield Pty Ltd would like to determine the variable rate for electricity per machine hour in order to estimate the electricity costs for the months of May and June. Information for the four...
-
In a small country near the Baltic Sea, there are only three commodities: potatoes, meatballs, and jam. Prices have been remarkably stable for the last 50 years or so. Potatoes cost 2 crowns per sack...
-
Government of Alberta 364-day T-bills with a face value of $1000000 were purchased on April 7 for $971 578. The T-bills were sold on May 16 for $983 500. (a) What was the market yield rate on April...
-
What are the main pitfalls of using Investment Comparables?
-
What type of signal is the output of a non-ICP accelerometer?
-
Excerpts from the 2012 financial statements for Goodyear are as follows (dollars in millions): REQUIRED: Assume that you have some capital to invest and that you are considering an equity investment...
-
What is the importance of employee-driven content? To appeal to adult learners, what do delivery methods need to include, and why?
-
The Stephanowicz Company's January 1 account balances are: During January, the following transactions were completed: (a) Materials purchased on account cost $92,000. (b) Miscellaneous factory...
-
Use the data in the table below to answer the following questions. (6 points) a. Calculate the standard deviation (sigma). (2 points) b. Identify the safety factor for a service level of 90% and...
-
Walker Patel is a portfolio manager at an investment management firm. After successfully implementing meanvariance optimization (MVO), he wants to apply reverse optimization to his portfolio. For...
-
An ETFs reported tracking error is typically measured as the: A. standard deviation of the difference in daily returns between an ETF and its benchmark. B. difference in annual return between an ETF...
-
Which of the following statements regarding applications of ETFs in portfolio management is correct? A. Equity ETFs tend to be more active than fixed-income ETFs. B. The range of risk exposures...
-
An ETFs tracking error, as traditionally reported, indicates to investors: A. whether the ETF is underperforming or outperforming its underlying index. B. the magnitude by which an ETFs returns...
-
Which of the following statements regarding exchange-traded funds (ETFs) is correct? ETFs: A. disclose their holdings on a quarterly basis. B. trade in both primary and secondary markets. C. offer a...
-
A taxpayer files their return and decides to treat it with what is later to be determined as negligence in their failing to accurately report their total income. What is the amount of the penalty...
-
How can a promoter avoid personal liability for pre-incorporation contracts?
-
Controlling the appearance of a graph. Figure 3-3 requires gridlines to read buret corrections. In this exercise, you will format a graph so that it looks like Figure 3-3. Follow the procedure in...
-
(a) What value of absorbance corresponds to 45.0% T? (b) If a 0.010 0 M solution exhibits 45.0% T at some wavelength, what will be the percent transmittance for a 0.020 0 M solution of the same...
-
For H2(g) + Br2(g) 2HBr(g) K = 72 10-4 at 1362K and H is positive. A vessel is charged with 48.0 PA HBr,1370 PaH2, and 3310 Pa Br2 at 1362 K. (a) Will the reaction proceed to the left or the right...
-
Aluminum is made by the electrolysis of bauxite. The embodied energy of virgin aluminium is approximately (all embodied energies are approximate) \(210 \mathrm{MJ} / \mathrm{kg}\). What else could...
-
Use the \(E-H_{p} ho\) chart of Fig. 14.7 to find the polymer with a modulus \(E\) greater than \(1 \mathrm{GPa}\) and the lowest embodied energy per unit volume. Data From Fig, 14.7 103 Young's...
-
A maker of polypropylene (PP) garden furniture is concerned that the competition is stealing market share by claiming that the 'traditional' material for garden furniture, cast iron, is less energy...
Fast Boat Nekkid An Escapade By Sea From Alaska To Mexico 1st Edition - ISBN: 0578979306 - Free Book

Study smarter with the SolutionInn App



